Abstract—
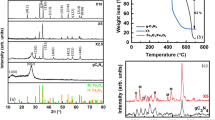
A composite material consisting of graphitic carbon nitride and iron(II,III) oxide (g-C3N4/Fe3O4) and having photocatalytic and magnetic properties has been synthesized via single-step thermal decomposition of melamine in the presence of Fe3O4 nanoparticles. It has been shown that, at synthesis temperatures in the range 400–500°C, Fe2+ ions do not oxidize, and the Fe3O4 particles retain their crystal structure and ferromagnetic properties, but the process involves the formation of crystalline g-C3N4 as well. The maximum in the intensity of photoluminescence bands of the g-C3N4/Fe3O4 composite material is shifted to lower photon energies compared to g-C3N4 prepared without Fe3O4 particles under the same synthesis conditions, which is due to a decrease in its band gap, formed by a system consisting of C–N π bonds with sp2 hybridization. The magnetic properties of g-C3N4/Fe3O4 composite particles allow them to be readily recovered from liquids for reuse.




Similar content being viewed by others
REFERENCES
Spasiano, D., Marotta, R., Malato, S., Fernandez-Ibanez, P., and Di Somma, I., Solar photocatalysis: materials, reactors, some commercial, and pre-industrialized applications. A comprehensive approach, Appl. Catal., B, 2015, vol. 170, pp. 90–123.
Wang, A., Wang, C., Fu, L., Wong-Ng, W., and Lan, Y., Recent advances of graphitic carbon nitride-based structures and applications in catalyst, sensing, imaging, and LEDs, Nano–Micro Lett., 2017, vol. 9, pp. 47–49.
Wen, J., Xie, J., Chen, X., and Li, X., A review on g‑C3N4-based photocatalysts, Appl. Surf. Sci., 2017, vol. 391, pp. 72–123.
Ong, W.-J., Tan, L.-L., Ng, Y.H., Yong, S.-T., and Chai, S.-P., Graphitic carbon nitride (g-C3N4)-based photocatalysts for artificial photosynthesis and environmental remediation: are we a step closer to achieving sustainability?, Chem. Rev., 2016, vol. 116, pp. 7159–7329.
Fina, F., Callear, S.K., Carins, G.M., and Irvine, J.T.S., Structural investigation of graphitic carbon nitride via XRD and neutron diffraction, Chem. Mater., 2015, vol. 27, pp. 2612–2618.
Thomas, A., Fischer, A., Goettmann, F., Antonietti, M., Müller, J.-O., Schlögl, R., and Carlsson, J.M., Graphitic carbon nitride materials: variation of structure and morphology and their use as metal-free catalysts, J. Mater. Chem., 2008, vol. 18, pp. 4893–4908.
Wu, W., Zhang, J., Fan, W., Li, Z., Wang, L., Li, X., Wang, Y., Wang, R., Zheng, J., Wu, M., and Zeng, H., Remedying defects in carbon nitride to improve both photooxidation and H2 generation efficiencies, ACS Catal., 2016, vol. 6, pp. 3365–3371.
Sudhaik, A., Raizada, P., Shandilya, P., Jeong, D.-Y., Lim, J.-H., and Singh, P., Review on fabrication of graphitic carbon nitride based efficient nanocomposites for photodegradation of aqueous phase organic pollutants, J. Ind. Eng. Chem., 2018, vol. 67, pp. 28–51.
Jia, X., Dai, R., Sun, Y., Song, H., and Wu, X., One-step hydrothermal synthesis of Fe3O4/g-C3N4 nanocomposites with improved photocatalytic activities, J. Mater. Sci.: Mater. Electron., 2016, vol. 27, pp. 3791–3798.
Lima, M.J., Sampaio, M.J., Silva, C.G., Silva, A.M.T., and Faria, J.L., Magnetically recoverable Fe3O4/g-C3N4 composite for photocatalytic production of benzaldehyde under UV-LED radiation, Catal. Today, 2019, vol. 328, pp. 293–299.
Denisov, N.M., Chubenko, E.B., Bondarenko, V.P., and Borisenko, V.E., Synthesis of oxygen-doped graphitic carbon nitride from thiourea, Tech. Phys Lett., 2019, vol. 45, pp. 108–110.
Chubenko, E.B., Baglov, A.V., Lisimova, E.S., and Borisenko, V.E., Synthesis of graphitic carbon nitride in porous silica glass, Int. J. Nanosci., 2019, vol. 18, paper 1940042.
Denisov, N.M., Chubenko, E.B., Bondarenko, V.P., and Borisenko, V.E., Photoluminescence of ZnO/C nanocomposites formed by the sol–gel method, J. Sol–Gel Sci. Technol., 2018, vol. 85, pp. 422–427.
Lotsch, B.V. and Schnick, W., New light on an old story: formation of melam during thermal condensation of melamine, Chem. – Eur. J., 2007, vol. 13, pp. 4956–4969.
Kumar, R., Barakat, M.A., and Alseroury, F.A., Oxidized Fe3O4/g-C3N4/polyaniline nanofiber composite for the selective removal of hexavalent chromium, Sci. Rep., 2017, vol. 7, paper 12850.
Geng, Z., Lin, Y., Yu, X., Shen, Q., Ma, L., Li, Z., Pan, N., and Wang, X., Highly efficient dye adsorption and removal: a functional hybrid of reduced graphene oxide–Fe3O4 nanoparticles as an easily regenerative adsorbent, J. Mater. Chem., 2012, vol. 22, pp. 3527–3535.
Wilson, D. and Langell, M.A., XPS analysis of oleylamine/oleic acid capped Fe3O4 nanoparticles as a function of temperature, Appl. Surf. Sci., 2014, vol. 303, pp. 6–13.
Jiang, Y., Sun, Z., Tang, C., Zhou, Y., Zeng, L., and Huang, L., Enhancement of photocatalytic hydrogen evolution activity of porous oxygen doped g-C3N4 with nitrogen defects induced by changing electron transition, Appl. Catal., B, 2019, vol. 240, pp. 30–38.
ACKNOWLEDGMENTS
We are grateful to Prof. V.V. Uglov for performing the X‑ray diffraction characterizations of the samples and to Prof. X.W. Sun and Dr. M. Marus for analyzing the experimental samples by X-ray photoelectron spectroscopy.
Funding
This work was supported by the Belarussian Academy of Sciences and the Ministry of Education of the Republic of Belarus through the Materials Physics, Novel Materials, and Advanced Technologies National Research Program (task nos. 1.15 and 1.56).
V.E. Borisenko acknowledges the partial support of the Russian Federation Ministry of Science and Higher Education as part of the program aimed at improving the competitiveness of the Moscow Engineering Physics Institute (National Nuclear Research University).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by O. Tsarev
Rights and permissions
About this article
Cite this article
Chubenko, E.B., Baglov, A.V., Fedotova, Y.A. et al. Synthesis of Graphitic Carbon Nitride on the Surface of Fe3O4 Nanoparticles. Inorg Mater 57, 136–141 (2021). https://doi.org/10.1134/S0020168521020059
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168521020059