Abstract
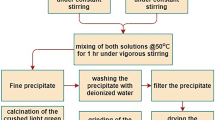
Neodymium dititanate, Nd2Ti2O7 (monoclinic structure, sp. gr. P21), has been prepared by solid-state reaction in air at temperatures from 1673 to 1773 K using the Nd2O3 and TiO2 oxides as starting materials. The high-temperature heat capacity of the resultant polycrystalline Nd2Ti2O7 samples has been determined by differential scanning calorimetry. The experimental Cp(T) data have been used to evaluate the thermodynamic functions of neodymium dititanate (enthalpy increment H°(T)–H°(320 K), entropy change S°(T)–S°(320 K), and reduced Gibbs energy Ф°(T)) in the temperature range 320–1053 K.
Similar content being viewed by others
References
Nanamatsu, S., Kimura, M., Doi, K., et al., A new ferroelectric: La2Ti2O7, Ferroelectrics, 1974, vol. 8, pp. 511–513.
Shcherbakova, L.G., Mamsurova, L.G., and Sikhanova, G.E., Rare-earth titanates, Usp. Khim., 1979, vol. 58, no. 3, pp. 423–447.
Hwang, D.W., Lee, J.S., Li, W., et al., Electronic band structure and photocatalytic activity of Ln2Ti2O7 (Ln = La, Pr, Nd), J. Phys. Chem. B, 2003, vol. 107, pp. 4963–4970.
Krishnankutty, K. and Dayas, K.R., Synthesis and characterization of monoclinic rare earth titanates, RE2Ti2O7 (RE = La, Pr, Nd), by a modified SHS method using inorganic activator, Bull. Mater. Sci., 2008, vol. 31, no. 6, pp. 907–918.
Komissarova, L.N., Shatskii, V.M., Pushkina, G.Ya., et al., Soedineniya redkozemel’nykh elementov. Karbonaty, oksalaty, nitraty, titanaty (Rare-Earth Compounds: Carbonates, Oxalates, Nitrates, and Titanates), Moscow: Nauka, 1984.
Vasil’eva, M.F., Gerasyuk, A.K., Goev, A.I., et al., High-quality optical coatings for the visible and near-IR spectral regions, based on new film-forming materials: gadolinium dititanate and lutetium dititanate, Prikl. Fiz., 2007, no. 5, pp. 91–98.
Milanova, M.M., Kakihana, M., Arima, M., et al., A simple solution route to the synthesis of pure La2Ti2O7 and Nd2Ti2O7 at 700–800 °C by polymerized complex method, J. Alloys Compd., 1996, vol. 242, pp. 6–10.
Balakrishnan, G., Petrenko, O.A., Lees, M.R., et al., Single crystal growth of rare-earth titanate pyrochlores, J. Phys.: Condens. Matter, 1998, vol. 10, pp. L723–L725.
Shlyakhtina, A.V., Morphotropy, isomorphism, and polymorphism of Ln2M2O7-Based (Ln = La–Lu, Y, Sc; M = Ti, Zr, Hf, Sn) oxides, Crystallogr. Rep., 2013, vol. 58, no. 4, pp. 548–562.
Scheunemann, K. and Müller-Buschbaum, H., Zur kristallstruktur von Nd2Ti2O7, J. Inorg. Nucl. Chem., 1975, vol. 37, no. 11, pp. 2261–2263.
Sloan, J. and Tilley, R.J.D., Phase formation in the Nd2Ti2O7–SrTiO3 system at 1350 °C in the presence of V2O5, CuV2O6, or SrCuO2, J. Solid State Chem., 1996, vol. 121, pp. 324–331.
Xing, H.X., Long, G., Guo, H., et al., Anisotropic paramagnetism of monoclinic Nd2Ti2O7 single crystals, J. Phys.: Condens. Matter, 2011, vol. 23, paper 21605.
Ishizawa, N., Ninomiya, K., Sakakura, T., et al., Redetermination of Nd2Ti2O7: a non-centrosymmetric structure with perovskite-type slabs, Acta Crystallogr., Sect. E: Struct. Rep. Online, 2013, vol. 69, paper i19.
Atuchin, V.V., Gavrilova, T.A., Crivel, J.-C., et al., Electronic structure of layered titanate Nd2Ti2O7, Surf. Sci., 2008, vol. 602, pp. 3095–3099.
Guo, H., Xing, H., Tong, J., et al., Possible spin frustration in Nd2Ti2O7 probed by muon spin relaxation, J. Phys.: Condens. Matter, 2014, vol. 28, paper 436 002.
Winfield, G., Azough, F., and Freer, R., Neodymium titanate (Nd2Ti2O7) ceramics, Ferroelectrics, 1992, vol. 133, pp. 181–186.
Zagorodnyuk, A.V., Sadkovskaya, L.V., Shamrai, G.V., et al., System Nd2O3–TiO2, Russ. J. Inorg. Chem., 1986, vol. 31, no. 9, pp. 1377–1379.
Huang, Z.K., Lin, Z.X., Yen, T.S., et al., J. Chin. Silicate Soc., 1979, vol. 7, no. 1, pp. 1–10.
Gong, W. and Zhang, R., Phase relationship in the TiO2–Nd2O3 pseudo-binary system, J. Alloys Compd., 2013, vol. 548, pp. 216–221.
Denisova, L.T., Chumilina, L.G., Denisov, V.M., et al., High-temperature heat capacity of samarium and erbium titanates with pyrochlore structure, Phys. Solid State, 2017, vol. 59, no. 12, pp. 2321–2324.
Denisov, V.M., Denisova, L.T., Irtyugo, L.A., and Biront, V.S., Thermal physical properties of Bi4Ge3O12 single crystals, Phys. Solid State, 2010, vol. 52, no. 7, pp. 1274–1277.
Denisova, L.T., Irtyugo, L.A., Kargin, Yu.F., et al., High-temperature heat capacity and thermodynamic properties of Tb2Sn2O7, Inorg. Mater., 2017, vol. 53, no. 1, pp. 67–69.
Rahimi-Nasrabadi, M., Mahdavi, S., and Adib, K., Photocatalytically active La2Ti2O7 nanostructures, synthesis and characterization, J. Mater. Sci.: Mater. Electron., 2017, vol. 28, no. 17, pp. 12 564–12 571.
Orum, A., Takatori, K., Hori, S., et al., Atomic force microscopy surface analysis of layered perovskite La2Ti2O7 particles, Jpn. J. Appl. Phys., 2016, vol. 55, paper 08NB08.
Gao, Z., Liu, L., Han, X., et al., Cerium titanate (Ce2Ti2O7): a ferroelectric ceramic with perovskitelike layered structure (PLS), J. Am. Ceram. Soc., 2015, vol. 98, no. 12, pp. 3930–3934.
Koz’min, P.A., Zakharov, N.A., and Surazhskaya, M.D., Crystal structure of Pr2Ti2O7, Inorg. Mater, 1997, vol. 33, no. 8, pp. 850–852.
Patwe, S.J., Katari, V., Salke, N.P., et al., Structural and electrical properties of layered perovskite type Pr2Ti2O7: experimental and theoretical investigations, J. Mater. Chem., 2015, vol. 3, pp. 4570–4584.
Kesari, S., Salke, N.P., Patwe, S.J., et al., Structural stability and anharmonicity of Pr2Ti2O7: Raman spectroscopic and XRD studies, Inorg. Chem., 2016, vol. 22, pp. 11791–11800.
Shannon, R.D., Revised effective ionic radii and systematic studies interatomic distances in halides and chalcogenides, Acta Crystallogr., Sect. A: Cryst. Phys., Diffr., Theor. Gen. Crystallogr., 1976, vol. 32, no. 5, pp. 751–767.
Reznitskii, L.A., Kalorimetriya tverdogo tela (Calorimetry of Solids), Moscow: Mosk. Gos. Univ., 1981.
Pet’kov, V.I., Asabina, E.A., Sukhanov, M.V., et al., Standard thermodynamic functions of CaNi0.5Zr1.5(PO4)3 crystalline phosphate in the range of T → 0 to 640 K, Russ. J. Phys. Chem. A, 2013, vol. 87, no. 12, pp. 1960–1968.
Saton, H., Takagi, M., Kinukawa, K.-I., et al., Heat capacity of LaMnO3, Thermochim. Acta, 1997, vol. 299, pp. 123–126.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © L.T. Denisova, Yu.F. Kargin, L.G. Chumilina, V.V. Ryabov, N.V. Belousova, V.M. Denisov, 2018, published in Neorganicheskie Materialy, 2018, Vol. 54, No. 8.
Rights and permissions
About this article
Cite this article
Denisova, L.T., Kargin, Y.F., Chumilina, L.G. et al. Synthesis and High-Temperature Heat Capacity of Neodymium Titanate. Inorg Mater 54, 815–819 (2018). https://doi.org/10.1134/S0020168518080046
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168518080046