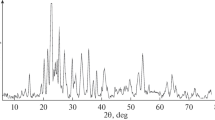
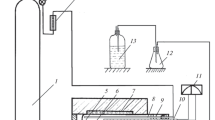
Abstract
We have investigated the preparation of molybdenum powders by reducing the oxide compounds MoO3, MgMoO4, and CaMoO4 with magnesium vapor at residual argon pressures in the range 5–20 kPa and temperatures in the range 700–800°C. Using the MgMoO4 and CaMoO4 compounds as precursors, we have obtained molybdenum powders with specific surface areas of up to 20 m2/g. The powders have a mesoporous structure. The reduction of the molybdenum compounds under such conditions was accompanied by separation of the reaction products due to the removal of magnesium oxide from the reaction zone.
Similar content being viewed by others
References
Kalamazov, R.U., Tsvetkov, Yu.V., and Kal’kov, A.A., Vysokodispersnye poroshki vol’frama i molibdena (Fine Tungsten and Molybdenum Powders), Moscow: Metallurgiya, 1988.
Dang, J., Zhang, G., Chou, K., et al., Kinetics and mechanism of hydrogen reduction of MoO3 to MoO2 applications, Int. J. Refract. Met. Hard Mater., 2013, vol. 41, no. 11, pp. 216–223.
Dang, J., Zhang, G., and Chou, K., Study on kinetics of hydrogen reduction of MoO2, Int. J. Refract. Met. Hard Mater., 2013, vol. 41, no. 11, pp. 356–362.
Nersisyan, H.H., Lee, J.H., and Won, C.W., The synthesis of nanostructured molybdenum under self-propagating high-temperature synthesis mode, Mater. Chem. Phys., 2005, vol. 89, nos. 2–3, pp. 283–288.
Aydinyan, S.V., Gumruyan, Zh., Manukyan, Kh.V., and Kharatyan, S.L., Self-sustaining reduction of MoO3 by the Mg–C mixture, Mater. Sci. Eng., B, 2010, vol. 172, no. 3, pp. 267–271.
Liu, L., Gu, H., and Chen, Q., Preparation of nanosized Mo powder by microwave plasma chemical vapor deposition method, Mater. Chem. Phys., 1999, vol. 59, no. 3, pp. 204–209.
Karelin, V.A. and Kovalev, S.V., Electrolytic synthesis of high-purity molybdenum powder from fluoride melts, Izv. Tomsk. Politekh. Univ., 2005, vol. 308, no. 3, pp. 97–100.
Kolosov, V.N., Miroshnichenko, M.N., and Orlov, V.M., RF Patent 2 596 513, Byull. Izobret., 2016, no.25.
Kim, H., Oh, J., Suh, C., et al., US Patent 8 979 975, 2015.
Zhong, H., Jianghao, L., and Xiangong, D., Low temperature molten salt preparation of molybdenum nanoparticles, Int. J. Refract. Met. Hard Mater., 2016, vol. 54, no. 1, pp. 315–321.
Orlov, V.M., Kryzhanov, M.V., and Kalinnikov, V.T., Magnesium reduction of tantalum oxide compounds, Dokl. Chem., 2014, vol. 457, no. 2, pp. 160–163.
Orlov, V.M., Kryzhanov, M.V., and Kalinnikov, V.T., Magnesium-vapor reduction of niobium oxide compounds, Dokl. Chem., 2015, vol. 465, no. 1, pp. 257–260.
Kolosov, V.N., Miroshnichenko, M.N., and Orlov, V.M., Influence of the chemical composition of precursors and reduction conditions on the properties of magnesiothermic tungsten powders, Inorg. Mater., 2016, vol. 52, no. 8, pp. 783–790.
Tablitsy fizicheskikh velichin (Tables of Physical Quantities), Kikoin, I.K., Ed., Moscow: Atomizdat, 1976.
Kulikov, I.S., Termicheskaya dissotsiatsiya soedinenii (Thermal Dissociation of Compounds), Moscow: Metallurgiya, 1969, 2nd ed.
Kazenas, E.K., Termodinamika ispareniya dvoinykh oksidov (Thermodynamics of Vaporization of Binary Oxides), Moscow: Nauka, 2004.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.N. Kolosov, M.N. Miroshnichenko, V.M. Orlov, 2017, published in Neorganicheskie Materialy, 2017, Vol. 53, No. 10, pp. 1081–1086.
Rights and permissions
About this article
Cite this article
Kolosov, V.N., Miroshnichenko, M.N. & Orlov, V.M. Influence of the composition of precursors and reduction conditions on the properties of magnesiothermic molybdenum powders. Inorg Mater 53, 1058–1063 (2017). https://doi.org/10.1134/S0020168517100119
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168517100119