Abstract
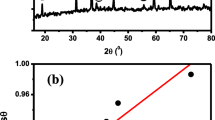
By using Co2+ and Co3+ salts, and freshly extracted ovalbumin, Co3O4 nanocrystals have been synthesized successfully. The pH of the solution was self-regulated for the hydrolysis of metal ions as the ovalbumin-water mixture was highly basic. Water soluble ovalbumin proteins served as a perfect matrix for entrapment of Co2+ and Co3+ ions thus forming a gel. Upon heat treatment, the dried gel precursor decomposed into nanocrystalline Co3O4. The crystallite size obtained by XRD line profile fitting was 45 ± 8 nm and particle size estimated from the SEM was in the range 20 nm-2 μm. EPR results show a very good fit to literature reports for nanocrystals in the size range of 8–17 nm. Even though the overall particle size is quite large and its distribution is quite wide EPR results confirm nanocrystalline nature of the particles obtained. Presented route is simple, cost effective, and environmentally friendly.
Similar content being viewed by others
References
Busca, G., Trifiro, F., and Vaccari, A., Characterization and Catalytic Activity of Cobalt-Chromium Mixed Oxides, Langmuir, 1990, vol. 6, no. 9, pp. 1440–1447.
Poizot, P., Laruelle, S., Grugeon, S., et al., Nano-Sized Transition-Metal Oxides As Negative-Electrode Materials for Lithium-Ion Batteries, Nature, 2000, vol. 407, no. 6803, pp. 496–499.
Yuan, Z., Huang, F., Feng, C., et al., Synthesis and Electrochemical Performance of Nanosized Co3O4, Mater. Chem. Phys., 2003, vol. 79, no. 1, pp. 1–4.
Kamata, K., Lu, Y., and Xia, Y., Synthesis and Characterization of Monodispersed Core-Shell Spherical Colloids with Movable Cores, J. Am. Chem. Soc., 2003, vol. 125, no. 9, pp. 2384–2385.
Wang, Y., Cai, L., and Xia, Y., Monodisperse Spherical Colloids of Pb and Their Use As Chemical Templates to Produce Hollow Particles, Adv. Mater., 2005, vol. 17, no. 4, pp. 473–477.
Mergler, Y.J., Hoebink, J., and Nieuwenhuys, B.E., CO Oxidation over a Pt/CoOx/SiO2 Catalyst: A Study Using Temporal Analysis of Products, J. Catal., 1997, vol. 167, no. 2, pp. 305–313.
Hamada, H., Kintaichi, Y., Inaba, M., et al., Role of Supported Metals in the Selective Reduction of Nitrogen Monoxide with Hydrocarbons over Metal/Alumina Catalysts, Catal. Today, 1996, vol. 29, nos. 1–4, pp. 53–57.
Sinha, A.S.K. and Shankar, V., Characterization and Activity of Cobalt Oxide Catalysts for Total Oxidation of Hydrocarbons, Chem. Eng. J., 1993, vol. 52, no. 3, pp. 115–120.
Mocuta, C., Barbier, A., and Renaud, G., CoO(111) Surface Study by Surface X-Ray Diffraction, Appl. Surf. Sci., 2002, vols. 162–163, pp. 56–61.
Xu, R. and Zeng, H.C., Mechanistic Investigation on Salt-Mediated Formation of Free-Standing Co3O4 Nanocubes at 95°C, J. Phys. Chem. B, 2003, vol. 107, no. 4, pp. 926–930.
Lakshmi, B.B., Patrissi, C.J., and Martin, C.R., Sol-Gel Template Synthesis of Semiconductor Oxide Micro- and Nanostructures, Chem. Mater., 1997, vol. 9, no. 11, pp. 2544–2550.
Svegl, F., Orel, B., Grabec-Svegl, I., et al., Characterization of Spinel Co3O4 and Li-Doped Co3O4 Thin Film Electrocatalysts Prepared by the Sol-Gel Route, Electrochim. Acta, 2000, vol. 45, nos. 25–26, pp. 4359–4371.
Baydi, M.E., Poillerat, G., Rehspringer, J.L., et al., A Sol-Gel Route for the Preparation of Co3O4 Catalyst for Oxygen Electrocatalysis in Alkaline Medium, J. Solid State Chem., 1994, vol. 109, no. 2, pp. 281–288.
Ni, Y.H., Ge, X.W., Zhang, Z.C., et al., A Simple Reduction-Oxidation Route to Prepare Co3O4 Nanocrystals, Mater. Res. Bull., 2001, vol. 36, nos. 13–14, pp. 2383–2387.
Sakamoto, S., Yoshinaka, M., Hirota, K., and Yamaguchi, O., Fabrication. Mechanical Properties, and Electrical Conductivity of Co3O4 Ceramics, J. Am. Ceram. Soc., 1997, vol. 80, no. 1, pp. 267–268.
Burriel, M., Garcia, G., Santiso, J., et al., Growth Kinetics, Composition, and Morphology of Co3O4 Thin Films Prepared by Pulsed Liquid-Injection MOCVD, Chem. Vap. Deposition, 2005, vol. 11, no. 2, pp. 106–111.
Singh, R.N., Koenig, J.F., Poillerat, G., and Chartier, P., Electrochemical Studies on Protective Thin Co3O4 and NiCo2O4 Films Prepared on Titanium by Spray Pyrolysis for Oxygen Evolution, J. Electrochem. Soc., 1990, vol. 137, no. 5, pp. 1408–1413.
Mine, Y., Recent Advances in the Understanding of Egg White Protein Functionality, Trends Food Sci. Technol., 1995, vol. 6, no. 7, pp. 225–232.
Lyckfeldt, O., Brandt, J., and Lesca, S., Protein Forming—A Novel Shaping Technique for Ceramics, J. Eur. Ceram. Soc., 2000, vol. 20, nos. 14–15, pp. 2551–2559.
Dhara, S. and Bhargava, P., Egg White As an Environmentally Friendly Low-Cost Binder for Gelcasting of Ceramics, J. Am. Ceram. Soc., 2001, vol. 84, no. 12, pp. 3048–3050.
Dhara, S. and Bhargava, P., A Simple Direct Casting Route to Ceramic Foams, J. Am. Ceram. Soc., 2003, vol. 86, no. 10, pp. 1645–1650.
Maensiri, S., Masingboon, C., Boonchom, B., and Seraphin, S., A Simple Route to Synthesize Nickel Ferrite (NiFe2O4) Nanoparticles Using Egg White, Scr. Mater., 2007, vol. 56, no. 9, pp. 797–800.
Dhara, S. and Bhargava, P., Influence of Nature and Amount of Dispersant on Rheology of Aged Aqueous Alumina Gelcasting Slurries, J. Am. Ceram. Soc., 2005, vol. 88, no. 3, pp. 547–552.
Sri Devi Kumari, T., Kannan, R., and Prem Kumar, T., Synthesis of LiMn2O4 from a Gelled Ovalbumin Matrix, Ceram. Int., 2008, vol. 35, no. 4, pp. 1565–1568.
Durmus, Z., Baykal, A., Kavas, H., et al., Ovalbumin Mediated Synthesis of Mn3O4, Polyhedron, 2009, doi: 10.1016/j.poly.2009.03.026.
Wejrzanowski, T., Pielaszek, R., Opalinska, A., et al., Quantitative Methods for Nanopowders Characterization, Appl. Surf. Sci., 2006, vol. 253, no. 1, pp. 204–208.
Pielaszek, R., Analytical Expression for Diffraction Line Profile for Polydispersive Powders, Proc. XIX Conf. Appl. Crystallography, Krakow, 2003, vol. 43.
Kurtulus, F. and Güler, H., A Simple Microwave-Assisted Route to Prepare Black Cobalt Co3O4, Inorg. Mater, 2005, vol. 41, no. 5, pp. 564–565.
Ozkaya, T., Baykal, A., Koseolu, Y., and Kavas, H., Synthesis of Co3O4 Nanoparticles by Oxidation-Reduction Method and Its Magnetic Characterization, Cent. Eur. J. Chem., 2009, doi: 10.2478/s 11532-009-0012-4.
Ozkaya, T., Baykal, A., Toprak, M.S., et al., Reflux Synthesis of Co3O4 Nanoparticles and Its Magnetic Characterization, J. Magn. Magn. Mater., 2009, vol. 321, pp. 2145–2149.
Ozkaya, T., Synthesis and Characterization of M3O4 (M: Fe, Mn, Co) Magnetic Nanoparticles, M. Sci. Thesis, Istanbul: Fatih Univ., 2008.
Malecki, A., Tareen, J., Doumerc, J., et al., Kinetics of Thermal Decomposition of Co3O4 Powder and Single Crystals, J. Solid State Chem., 1985, vol. 56, no. 1, pp. 49–57.
Dutta, P., Seehra, M.S., Thota, S., and Kumar, J., A Comparative Study of the Magnetic Properties of Bulk and Nanocrystalline Co3O4, J. Phys.: Condens. Matter, 2008, vol. 20, no. 1, pp. 015218–015225.
Venkateswara Rao, K. and Sunandana, C.S., Co3O4 Nanoparticles by Chemical Combustion: Effect of Fuel to Oxidizer Ratio on Structure, Microstructure and EPR, Solid State Commun., 2008, vol. 148, nos. 1–2, pp. 32–37.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Ahmadov, T.O., Durmus, Z., Baykal, A. et al. A simple approach for the synthesis of Co3O4 nanocrystals. Inorg Mater 47, 426–430 (2011). https://doi.org/10.1134/S0020168511040017
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168511040017