Abstract
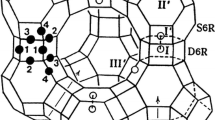
The structures of chabazite, laumontite, and wairakite are analyzed using literature data with the aim of revealing their similar and dissimilar features. The framework of the three zeolites contain chains made up of alternating six-and four-membered rings. A distinguishing feature of chabazite is a network of oxygen-hydrogen bonds between intraframework oxygens (which link silicon (aluminum) atoms) and water molecules and between water molecules. A characteristic feature of laumontite is the ordered arrangement of the silicon and aluminum sites in the framework in the form of alternating aluminum and silicon layers. A distinguishing feature of wairakite is that there are no oxygen-hydrogen bonds between water molecules. The transition from the chabazite framework to the laumontite framework can be interpreted in terms of the ordering and displacement of aluminum and silicon atoms. In addition to the differences between the frameworks of laumontite and wairakite, which are attributable to the fact that these zeolites were formed at different temperatures and pressures, they differ in the relative arrangement of the water molecules and calcium atoms, which cannot be accounted for by different formation conditions.
Similar content being viewed by others
References
Bosc d’Antic, L., Mémoire sur la Chabazite, J. Hist. Nat., 1792, vol. 2, pp. 181–184.
Jameson, R., System of Mineralogy, Edinburgh: Bell and Bradfute, 1805, p. 539.
Haüy, R.J., Tableau comparative des résultats de cristallographie et de l’analyse chimique relativement à la classification des minéraux, Paris: Coureier, 1809.
von Leonhard, K.C., Handbuch der Oryktognosie, Heidelberg: Monh & Winter, 1821, p. 448.
Haüy, R.J., Traité de minéralogy, Paris: Chez Louis, 1801, vol. 3, pp. 410–412.
Steiner, A., Wairakite, the Calcium Analogue of Analcime, a New Zeolite Mineral, Mineral. Mag., 1955, vol. 30, pp. 691–698.
Coombs, D.S., X-ray Observations on Wairakite and Non-cubic Analcime, Mineral. Mag., 1955, vol. 30, pp. 699–708.
Combs, D.S., Alberti, A., Armbruster, T., et al., Recommended Nomenclature for Zeolite Minerals: Report of the International Mineralogical Association, Commission on New Minerals and Minerals Names, Can. Mineral., 1997, vol. 35, pp. 1571–1606.
Smith, J.V., Rinaldi, R., and Dent Glasser, L.S., Structures with a Chabazite Framework: II. Hydrated Ca-Chabazite at Room Temperature, Acta Crystallogr., 1963, vol. 16, pp. 45–53.
Yakubovich, O.V. and Simonov, M.A., Refined Crystal Structure of the Zeolite Laumontite, Ca(H2O)2.8[Al2Si4O12] · 0.5H2O, Kristallografiya, 1985, vol. 30, no. 6, pp. 1072–1076.
Ståhl, K. and Artioli, G.A., Neutron Powder Diffraction Study of Fully Deuterated Laumontite, Eur. J. Mineral., 1993, vol. 5, pp. 851–856.
Fridriksson, T., Bish, D.L., and Bird, D.K., Hydrogen-Bonded Water in Laumontite: I. X-ray Powder Diffraction Study of Water Site Occupancy and Structural Changes in Laumontite during Room-temperature Isothermal Hydration/Dehydration, Am. Mineral., 2003, vol. 88, pp. 277–287.
Takeuchi, Y., Mazzi, F., Haga, N., et al., The Crystal Structure of Wairakite, Am. Mineral., 1979, vol. 64, pp. 993–1001.
Author information
Authors and Affiliations
Additional information
Original Russian Text © T.N. Kol’tsova, 2007, published in Neorganicheskie Materialy, 2007, Vol. 43, No. 2, pp. 218–226.
Rights and permissions
About this article
Cite this article
Kol’tsova, T.N. Crystal structures of zeolites with the general formula CaAl2Si4O12 · nH2O. Inorg Mater 43, 176–184 (2007). https://doi.org/10.1134/S002016850702015X
Received:
Issue Date:
DOI: https://doi.org/10.1134/S002016850702015X