Abstract
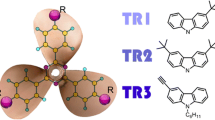
Singlet–singlet (S–S) and triplet–triplet (T–T) absorption spectra of multi-chromophoric molecules containing polyphenyl fragments are studied computationally by quantum chemical DFT/TDDFT methods. Patterns observed previously in experimental absorption spectra of the parent chromophore and its composite analogs are interpreted based on the localization of molecular orbitals (MO) involved in the optically active transitions. In the singlet ground state, the tilt between the phenyl fragments in the parent molecule and in the respective fragments of its composite analog is similar. The composite molecules exhibit symmetric geometry; therefore, MO localized on different ‘parent’ moieties are quasi-degenerate. Optically active transitions are delocalized over these moieties, and the position of the intense S–S absorption band is not altered. By contrast, the equilibrium geometry of the triplet ground state of the parent molecule is planar. In the multi-chromophoric analog, only one of the ‘parent’ polyphenyl moieties becomes planar. This property results in two specific features: (i) the character of the lowest triplet state is similar in the parent chromophore and its composite; therefore, no shift in their phosphorescence spectrum is observed; (ii) a change in the symmetry of the composite analog gives rise to intramolecular charge-transfer (CT) bands in its T–T absorption spectrum, which is not observed in the spectrum of the parent chromophore. An increase in the conjugation length of the parent chromophore leads to a decrease in the energy difference between the local and intramolecular CT bands in the spectrum of the composite species. These features are reproduced at the DFT/TDDFT level of theory with a good agreement with experimental data.








Similar content being viewed by others
REFERENCES
Ashkenasy, G., Hermans, T.M., Otto, S., and Taylor, A.F., Chem. Soc. Rev., 2017, vol. 46, p. 2543.
Alfimov, M.V., Batekha, I.G., Shek, Yu.B., and Gerko, V.I., Spectrochim. Acta, Part A, 1971, vol. 27, p. 329.
Gerko, V.I., Alfimov, M.V., Popov, L.S., and Kovalenko, N.P., Dokl. Akad. Nauk SSSR, 1976, vol. 230, no. 1, p. 125.
Sheck, Yu.B., Kovalenko, N.P., and Alfimov, M.V., J. Lumin., 1977, vol. 15, p. 157.
Chibisov, A.K., Russ. Chem. Rev., 1970, vol. 39, no. 10, p. 891.
Labhart, H. and Heinzelmann, W., Organic Molecular Photophysics, Birks, J.B., Ed., New York: Wiley, 1973, vol. 1, p. 297.
Carmichael, I. and Hug, G.L., J. Phys. Chem. Ref. Data, 1986, vol. 15, no. 1, p. 1.
Kasha, M., Rawls, H.R., and El-Bayoumi, M.A., Pure Appl. Chem., 1965, vol. 11, nos. 3–4, p. 371.
Kreibich, U.T. and Wild, U.P., J. Mol. Spectrosc., 1973, vol. 47, p. 189.
Tahara, T., Hamaguchi, H., and Tasumi, M., J. Phys. Chem., 1990, vol. 94, no. 1, p. 170.
Nguyen, K.A. and Kennel, J., J. Chem. Phys., 2002, vol. 117, no. 15, p. 7128.
Cronstrand, P., Rinkevicius, Z., Luo, Y., and Agren, H., J. Chem. Phys., 2005, vol. 122, no. 22, p. 224104.
Krysko, I.D., Freidzon, A.Y., and Bagaturyants, A.A., J. Phys. Chem. C, 2019, vol. 123, p. 11171.
Osad’ko, I.S., Opt. Spectrosc., 2019, vol. 127, no. 1, p. 1.
Andrzejak, M., Skora, T., and Petelenz, P., J. Phys. Chem. C, 2019, vol. 123, no. 1, p. 91.
Tatikolov, A.S. and Pronkin, P.G., J. Appl. Spectrosc., 2019, vol. 85, p. 991.
Fedotova, T.V., Gutrov, V.N., Zakharova, G.V., Chibisov, A.K., and Alfimov, M.V., High Energy Chem., 2019, vol. 53, no. 1, p. 22.
Arbeloa, E.M., Previtali, C.M., and Bertolotti, S.G., ChemPhysChem, 2018, vol. 19, no. 8, p. 934.
Militello, M.P., Hernandez-Ramirez, R.E., Lijanova, I.V., Previtali, C.M., Bertolotti, S.G., and Arbeloa, E.M., J. Photochem. Photobiol., A, 2018, vol. 353, p. 71.
Jacquemin, D., Perpete, E.A., Scalmani, G., Frisch, M.J., Assfeld, X., Ciofini, I., and Adamo, C., J. Chem. Phys., 2006, vol. 125, p. 164324.
Schmidt, M.W., Baldridge, K.K., Boatz, J.A., Elbert, S.T., Gordon, M.S., Jensen, J.H., Koseki, S., Matsunaga, N., Nguyen, K.A., Su, S., Windus, T.L., Dupuis, M., and Montgomery, J.A., J. Comput. Chem., 1993, vol. 14, p. 1347.
Gordon, M.S. and Schmidt, M.W., Theory and Applications of Computational Chemistry: The First Forty Years, Dykstra, C.E., Frenking, G., Kim, K.S., and Scuseria, G.E., Eds., Amsterdam: Elsevier, 2005, p. 1167.
Granovsky, A.A., Firefly, Version 8.2.0, 2017. https://classic.chem.msu.su/gran/firefly/index.html
ACKNOWLEDGMENTS
This work was supported by the Ministry of Science and Higher Education within the State assignment FSRC “Crystallography and Photonics” RAS. The authors are grateful to dr. K.G. Komarova and prof. A.K. Chibisov for many useful comments and suggestions related to the problem considered in this paper.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Alfimov, M.V., Anger, I.A., Dubinets, N.O. et al. Specific Features in the Triplet–Triplet Absorption Spectra of Multi-Chromophoric Molecules: a Computational Study. High Energy Chem 56, 391–398 (2022). https://doi.org/10.1134/S0018143922060029
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0018143922060029