Abstract
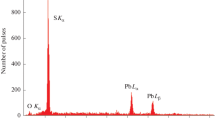
Nanoparticles of lead sulphide have been stabilized in the presence of excess Pb2+ in aqueous basic medium by a simple chemical route of synthesis. These PbS nanoparticles were synthesized very conveniently at room temperature using hexametaphosphate as stabilizer. These nanoparticles have an absorption extending into the NIR region. A significant quantum confinement effect made the bandgap of lead sulphide nanoparticles produce a blue shift from 0.41 eV to about 1.5 eV. The size and morphology of the particles were studied by TEM. Particles were relatively small sized (about 6 nm) having narrow size distribution. XRD data analysis indicate that the product is a mixture of PbS, PbO and Pb(OH)2. Both XRD pattern and HRTEM images confirm the crystalline structure of lead sulphide crystals. IR spectroscopy indicates the formation of PbS. PbS nanoparticles were fairly stable and could be stored for about three weeks at room temperature and for about two months at 5°C without agglomeration. These particles were photoactive and sensitized the reaction of aniline by light leading to the formation of azobenzene.
Similar content being viewed by others
References
Tanaka, K., Science, 1997, vol. 277, p. 1786.
Mitchell, K. and Ibers, J.A., Chem. Rev., 2002, vol. 102, p. 1929.
Kohn, S.E., Yu, P.Y., Petroff, Y., Shen, Y.R., Tsang, Y., and Cohen, M.I., Phys. Rev. B., 1973, vol. 8, p. 1477.
Machol, J.L., Wise, F.W., Patel, R.C., and Tanner, D.B., Phys. Rev. B, 1993, vol. 48, p. 2819.
Nair, P.K., Gomezdaza, O., and Nair, M.T.S., Adv. Mater. Opt. Electron., 1992, vol. 1, p. 139.
Gadenne, P., Yagil, Y., and Deutscher, G., J. Appl. Phys., 1989, vol. 66, p. 3019.
Hirata, H. and Higashiyama, K., Bull. Chem. Soc. Jpn., 1971, vol. 44, p. 2420.
Chaudhuri, T.K. and Chatterjes, S., Proceedings of the International Conference on Thermoelectron (Arlington, USA), 1992, vol. 11, p. 40.
Colvin, V.L., Schlamp, M.C., and Alivisatos, A.P., Nature, 1994, vol. 370, p. 354.
Kane, R.S., Cohen, R.E., and Silbey, R., J. Phys. Chem. B, 1996, vol. 100, p. 7928.
Nenadovice, M.T., Comor, M.I., Vasic, V., and Micic, O.I., J. Phys. Chem., 1990, vol. 94, p. 6390.
Trindade, T., Brien, P.O., Zhang, X., and Motevalli, M., J. Mater. Chem., 1997, vol. 7, p. 1011.
Kaito, C., Saito, Y., and Fujita, K., Jpn. J. Appl. Phys., 1987, vol. 26, p. 1973.
Wang, S.H. and Yang, S.H., Langmuir, 2000, vol. 16, p. 389.
Wang, Y., Suna, A., Mahler, W., and Kasowski, R., J. Chem. Phys., 1987, vol. 87, p. 7315.
Wang, Y. and Herron, N., J. Phys. Chem., 1987, vol. 91, p. 257.
Kane, R.S., Cohen, R.E., and Silbey, R., Chem. Mater., 1996, vol. 8, p. 1919.
Liveri, V.T., Rossi, M.D., Arrigo, G., Manno, D., and Micocci, G., Appl. Phys. A, 1999, vol. 69, p. 369.
Yang, S.B., Qadri, B.R., and Ratna, J. Phys. Chem., 1996, vol. 100, p. 17255.
Qiao, Z., Xie, Y., Xu, J., Zhu, Y., and Qian, Y., J. Colloid Interf. Sci. 1999. V. 214. P. 459.
Ni, Y.H., Wang, F., Liu, H.J., Yin, G., Hong, J.M., Ma, X., and Xu, Z., J. Crystal Growth., 2004, vol. 262, p. 399.
Yang, J.P. and Fendler, J.H., J. Phys. Chem., 1995, vol. 99, p. 5505.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Gupta, S.M. Synthesis and photophysics of leadsulphide nanocrystallites. High Energy Chem 47, 130–134 (2013). https://doi.org/10.1134/S0018143913030053
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0018143913030053