Abstract
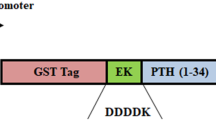
Opiorphin secreted into the human saliva is an endogenous regulator pentapeptide (QRFSR) showing a pain suppressant effect similar to morphine. We report here a strategy of producing tandemly repeated multimers of opiorphin peptide efficiently by expressing in Esherichia coli cells. For improving the expression level and increasing the yield of pentapeptide, 32-mer tandem sequence of opiorphin was generated by using partially complementary primers inserted into pGEX-2T expression vector, following fused with glutathione transferase (GST) gene. Recombinant tandem multimer was easily captured by GST column with a yield of 9.2 mg/L, and then 32-mer opiorphin polypeptide of 28.49 kDa size was cleaved into monomers by CNBr and endoproteinase Asp-N. Opiorphin polypeptides having a molecular weight of about 693 Da and containing 5 amino acids in length were then generated. The amount of cleaved and purified recombinant opiorphin monomers was calculated as 6.2 mg/L by the Bradford assay. Protein expression and cleavage of recombinant polypeptide were confirmed by SDS-PAGE and TLC. Results from this study clearly open a door to the mass production of the opiorphin peptide to use for laboratory research and industrial purposes. The overall gene construction and the direct cloning to an expression vector strategy using partially complementary primers with cleavable linker peptides could be applicable to many small peptides and their analogs for pharmaceutical and clinical application.




Similar content being viewed by others
REFERENCES
Wisner, A., Dufour, E., Messaoudi, M., Nejdi, A., Marcel, A., Ungeheuer, M.N., and Rougeot, C., Proc. Natl. Acad. Sci. U.S.A., 2006, vol. 103, no. 47, pp. 17979–17984.
Rougeot, C., Messaoudi, M., Hermitte, V., Rigault, A.G., Blisnick, T., Dugave, C., Desor, D., and Rougeon, F., Proc. Natl. Acad. Sci. U. S. A., 2003, vol. 100, no. 14, pp. 8549–8554.
Nishimura, K. and Hazato, T., Biochem. Biophys. Res. Commun., 1993, vol. 194, no. 2, pp. 713–719.
Tong, Y., Tar, M., Melman, A., and Davies, K., BJU Int., 2008, vol. 102, pp. 736–740.
Rougeot, C., Rosinski-Chupin, I., Njamkepo, E., and Rougeon, F., Eur. J. Biochem., 1994, vol. 219, pp. 765–773.
Rougeot, C., Robert, F., Menz, L., Bisson, J.F., and Messaoudi, M., J. Physiol. Pharmacol., 2010, vol. 6, pp. 483–490.
Tian, X., Chen, J., Xiong, W., He, T., and Chen, Q., Peptides, 2009, vol. 30, pp. 1348–1355.
Thanawala, V., Kadam, V.J., and Ghosh, R., Curr. Drug Targets, 2008, vol. 10, pp. 887–889.
Yang, Q.Z., Lu, S.S., Tian, A.M., Yang, A.M., Ge, W.W., and Chen, Q., Neurosci. Lett., 2011, vol. 489, pp. 131–135.
Andersson, L., Blomberg, L., Flegel, M., Lepsa, L., Nilsson, B., and Verlander, M., Biopolymers, 2000, vol. 55, no. 3, pp. 227–250.
Ingham, A.B., and Moore, R.J., Biotechnol. Appl. Biochem., 2007, vol. 47, pp. 1–9.
Li, Y., and Chen, Z., FEMS Microbiol. Lett., 2008, vol. 289, no. 2, pp. 126–129.
Li, Y., Biotechnol. Appl. Biochem., 2009, vol. 6, no. 54/1, pp. 1–9.
Wieles, B., Nagai, S., Wiker, H.G., Harboe, M., and Ottenhoff, T.H., Infect. Immun., 1995, vol. 63, no. 12, pp. 4946–4948.
Sheibani, N., Prep. Biochem. Biotechnol., 1999, vol. 29, no. 1, pp. 77–90.
Sambrook, J.F. and Russell, D.W., Molecular Cloning: A Laboratory Manual, 3rd ed., New York: Cold Spring Harbor Laboratory Press, Academic, 2001.
http://gentle.magnusmanske.de/.
https://www.dnastar.com/.
Kalendar, R., Lee, D., and Schulman, A.H., Genes Genomes Genomics, 2009, vol. 3, pp. 1–14.
Schechter, I., and Berger, A., Biochem. Biophys. Res. Commun., 1967, vol. 20, no. 27/2, pp. 157–162.
Laemli, U.K., Nature, 1970, vol. 227, pp. 680–685.
Bradford, M.M., Anal. Biochem., 1976, vol. 72, pp. 248–254.
Villa, S., De Fazio, G., and Canosi, U., Analytical Biochemistry, 1989, vol. 177, pp. 161–164.
Tarentino, A.L., in Handbook of Proteolytic Enzymes, 2nd ed., Barret, A.J., Rawlings, N.D., and Woessner, J.F., Eds., London: Elsevier, Academic, 2004.
Bhusan, R., Mahesh, V.K., and Mallikharjun, P.V., Biomed. Chromatogram., 1989, vol. 3, no. 3, pp. 95–104.
Choi, H.J., Seo, M.J., Lee, J.C., Cheigh, C.I., Park, H., Ahn, C., and Pyun, Y.R., J. Microbiol. Biotechnol., 2005, vol. 15, no. 2, pp. 330–336.
Park, E., Chae, Y.K., Lee, JY., Lee, B., and Kim, Y., J. Microbiol. Biotechnol., 2006, vol. 16, pp. 1429–1433.
Rao, X., Hu, J., Li, S., Jin, X., Zhang, C., Cong, Y., Hu, X., Tan, Y., Huang, J., Chen, Z., Zhu, J., and Hu, F., Peptides, 2005, vol. 26, no. 5, pp. 721–729.
Lee, J.H., Minn, I., Park, C.B., and Kim, S.C., Protein Expr. Purif., 1998, vol. 12, pp. 53–60.
Park, C.J., Lee, J.H., Hong, S.S., Lee, H.S., and Kim, S., Appl. Microbiol. Biotechnol., 1998, vol. 50, pp. 71–76.
Lee, S.J., Lee, J.H., Jin, H.J., Lee, J.H., Ryu, H.Y., Kim, Y., Kong, I.S., and Kim, K.W., Mol. Cells, 2000, vol. 30, no. 10/2, pp. 236–240.
Wang, F., Song, H., Wang, X., Zhang, W., Wang, B., Zho, J., and Hu, Z.B., Appl. Biochem. Biotechnol., 2012, vol. 166, no. 3, pp. 612–619.
Jeong, D.W., Shin, D.S., Ahn, C.W., Song, I.S., and Lee, H.J., J. Microbiol. Biotechnol., 2007, vol. 17, no. 6, pp. 952–959.
Shin, J.R., Lim, K.J., Kim, D.J., Cho, J.H., and Kim, S.C., PLoS One, 2013, vol. 8, no. 3. e58997.
Stahl, E. and Ashworth, M.R.F., Quart. Rev. Biol., 1970, vol. 45, no. 3, pp. 316–317.
ACKNOWLEDGMENTS
We would like to thank Karadeniz Technical University Research Foundation (grant no. 5636), Turkey for their financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
No conflict of interest is declared. This article does not contain any studies involving animals or human participants performed by the author.
Rights and permissions
About this article
Cite this article
Guler, H.I. Recombinant Production of Opiorphin Pentapeptide as Tandem Multimers Through Rational Design of Primers. Appl Biochem Microbiol 56, 141–148 (2020). https://doi.org/10.1134/S0003683820020167
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0003683820020167