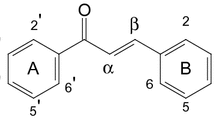
Abstract
Citral extracted from Cymbopogon citratus (Gramineae) was subjected to photochemical epoxidation with hydrogen peroxide to obtain a mixture of epoxy derivatives at the C2=C3 and C6=C7 double bonds. The thermal oxidation of citral with m-chloroperoxybenzoic acid at room temperature gave only the corresponding 6,7-epoxy derivative as a mixture of E and Z isomers with respect to the C2=C3 double bond. Photosensitized oxygenation of citral in the presence of tetraphenylporphyrin, Rose Bengal, or chlorophyll lead to a mixture of two isomeric hydroperoxides, (2E)-6-hydroperoxy-3,7-dimethylocta-2,7-dienal and (2E,5E)-7-hydroperoxy-3,7-dimethylocta-2,5-dienal. Epoxidation of pulegone isolated from Penny royal oil (Mentha pulegium, Lamiaceae) with hydrogen peroxide under irradiation with a sodium lamp lead to a mixture of cis- and trans-isomeric 2,2,6-trimethyl-1-oxaspiro[2.5]octan-4-ones, whereas under conditions of photosensitized oxygenation two hydroperoxide derivatives, 2-(1-hydroperoxy-1-methylethyl)-5-methylcyclohex-2-en-1-one and 2-hydroperoxy-5-methyl-2-(1-methylethenyl)cyclohexan-1-one, were also formed. Camphene reacted with hydrogen peroxide under irradiation to give a mixture of the corresponding endo- and exo-epoxy derivatives and camphor, while its thermal oxidation with m-chloroperoxybenzoic produced only the two former.
Similar content being viewed by others
References
Grayson, D.H., Nat. Prod. Rep., 1998, vol. 15, no. 5, p. 439.
Wagner, K.-H. and Elmadfa, I., Ann. Nutr. Metab., 2003, vol. 47, nos. 3–4, p. 95.
Crowell, P.L., J. Nutr., 1999, vol. 129, p. 775.
Mills, J.J., Chari, R.S., Boyer, I.J., Gould, M.N., and Jirtle, R.L., Cancer Res., 1995, vol. 55, p. 979.
Carnesecchi, S., Schneider, Y., Ceraline, J., Duranton, B., Gosse, F., Seller, N., and Raul, F., J. Pharm. Exp. Ther., 2001, vol. 298, p. 197.
Elgendy, E.M., Boll. Chim. Farm., 1997, vol. 136, p. 532.
Elgendy, E.M., Chim. Pharm J., 1998, vol. 50, p. 225.
Méou, A., Garcia, M.-A., and Brun, P., J. Mol. Catal. A: Chem., 1999, vol. 138, nos. 2–3, p. 221.
Elgendy, E.M., Boll. Chim. Farm., 2004, vol. 143, p. 337.
Geraghty, N.W.A., Photochemistry, Dunkin, I., Ed., London: Royal Soc. Chem., 2005, vol. 35, p. 116.
Richter, S., Gatto, B., Fabris, D., Takao, K.-i., Kobayashi, S., and Palumbo, S.M., Nucleic Acids Res., 2003, vol. 31, no. 17, p. 5149.
Yarovaya, O.I., Korchagina, D.V., Salomatina, O.V., Polovinka, M.P., and Barkhash, V.A., Mendeleev Commun., 2003, p. 27.
Yarovaya, O.I., Korchagina, D.V., Gatilov, Yu.V., and Barkhash, V.A., Russ. J. Org. Chem., 2002, vol. 38, no. 6, p. 810.
Asthana, A., Larson, R. A., Marley, K.A., and Tuveson. R.W., Photochem. Photobiol., 1992, vol. 56, no. 2, p. 211.
Tisserand, R.B. and Balacs, T., Essential oil Safety: A Guide for Health Care Professionals, Edinburgh: Churchill Livingstone, 1995, p. 147.
Schaneberg, B.T. and Khan, I.A., J. Agric. Food Chem., 2002, vol. 50, no. 6, p. 1345.
Reis-Vasco, E.M.C., Coelho, J.A.P., and Palavra, A.M.F., Flavor Fragrance J., 1999, vol. 14, no. 3, p. 156.
Barnes, J., Anderson, L.A., and Phillipson, J.D., Herbal Medicines, London: Pharmaceutical, 2002, 2nd ed., p. 207.
French, L.G., Chem. Educator, 2002, vol. 1, no. 5, p. 270.
Khojasteh-Bakht, S.C., Chen, W., Koenigs, L.L., Peter, R.M., and Nelson, S.D., Drug Metab. Dispos., 1999, vol. 27, no. 5, p. 574.
Ngo, K.-S., Cheung, K.-K., and Brown, G.D., J. Chem. Res., Synop., 1998, p. 80.
Epe, B., Häring, M., Ramaiah, D., Stopper, H., Abou-Elzahab, M.M., Adam, W., and Saha-Möller, C.R., Carcinogenesis, 1993, vol. 14, no. 11, p. 2271.
Elgendy, E.M., Chim. Pharm. J., 2000, vol. 52, p. 227.
Kochevar, I.E. and Dumn, D.A., Photochemistry and the Nucleic Acids, Morrison, H., Ed., (Bioorganic Photochemistry, vol. 1), New York: Wiley, 1990, p. 273.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Zhurnal Organicheskoi Khimii, 2008, Vol. 44, No. 6, pp. 825–833.
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Elgendy, E.M., Khayyat, S.A. Oxidation studies on some natural monoterpenes: Citral, pulegone, and camphene. Russ J Org Chem 44, 814–822 (2008). https://doi.org/10.1134/S1070428008060067
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428008060067