Abstract
A new method has been proposed for the green and efficient synthesis of xanthenediones and their corresponding open-chain analogs aryl-methylene bis(3-hydroxy-5,5’-dimethyl-2-cyclohexene-1-ones) using natural acid, Lemon juice, as the catalyst. This method involves the use of lemon juice as a surfactant-type bronsted acid, which makes it non-polluting and produces a high-purity product with excellent yield. Additionally, the work-up procedure is clean and straightforward. Dimedone and aldehydes are the starting materials for this process.
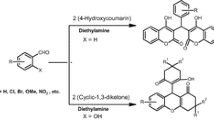
Graphical abstract





Similar content being viewed by others
References
L.A. Thompson, Curr. Opin. Chem. Biol. 4, 324 (2000)
A. Nefzi, J.M. Ostresh, R.A. Houghten, Chem. Rev. 97, 449 (1997)
N. Mizuno, M, Misono. Chem. Rev. 98, 199 (1998)
J.P. Poupelin, G. Saint-Rut, O. Fussard-Blanpin, G. Narcisse, G. Uchida-Ernouf, R. Lakroix, Eur. J. Med. Chem. 13, 67 (1978)
M.A. Bhat, A.M. Naglah, S. Akber Ansari, H.M. Al-Tuwajiria, A. Al-Dhfyan, Molecules 26(12), 3667 (2021)
K. Chibale, M. Visser, D.V. Schalkwyk, P.J. Smith, A. Saravanamuthu, A.H. Fairlamb, Tetrahedron 59, 2289 (2003)
A. Banerjee, A.K. Mukherjee, Stain Technol. 56, 83 (1981)
C.G. Knight, T. Stephens, Biochem. J. 258, 683 (1989)
M. Farzaneh, M. Feilizadeh, Chem. Methodol. 4, 647 (2020)
X. Fan, X. Hu, X. Zhang, J. Wang, Can. J. Chem. 83, 16 (2005)
B. Karami, S. Nejati, K. Eskandari, Curr. Chem. Lett. 4(4), 169 (2015)
G.K. Verma, K. Raghuvanshi, R.K. Verma, P. Dwivedi, M.S. Singh, Tetrahedron 67, 3698 (2011)
X.S. Fan, Y.Z. Li, X.Y. Zhang, X.Y. Hu, J.J. Wang, Chin. J. Org. Chem. 25, 1482 (2005)
J.J. Ma, J.C. Li, R.X. Tang, X. Zhou, Q.H. Wu, C. Wang, M.M. Zhang, Q. Li, Chin. J. Org. Chem. 27, 640 (2007)
F. Darviche, S. Balalaie, F. Chadegani, P. Salehi, Synth. Commun. 37, 1059 (2007)
P.B. Pawar, S.D. Jadhav, M.B. Deshmukh, S.S. Patil, Indian J. Chem. 52B, 1185 (2014)
B. Das, P. Thirupathi, K.R. Reddy, B. Ravikanth, L. Nagarapu, Catal. Commun. 8, 535 (2007)
G.I. Shakibaei, P. Mirzaei, A. Bazgir, Appl. Catal. 325, 188 (2007)
B. Das, P. Thirupathi, I. Mahender, V.S. Reddy, Y.K. Rao, J. Mol. Catal. A Chem. 247, 233 (2006)
G. Song, B. Wang, H. Luo, L. Yang, Catal. Commun. 8, 673 (2007)
M. Seyyedhamzeh, P. Mirzaei, A. Bazgir, Dyes Pigm. 76, 836 (2008)
A. John, P.J.P. Yadav, S. Palaniappan, J. Mol. Catal. A Chem. 248, 121 (2006)
S. Kantevari, R. Bantu, L. Nagarapu, J. Mol. Catal. A Chem. 269, 53 (2007)
Z.H. Zhang, Y.H. Liu, Catal. Commun. 9, 1715 (2008)
N. Mulakayala, P.V.N.S. Murthy, D. Rambabu, M. Aeluri, R. Adepu, G.R. Krishna, C.M. Reddy, K.R.S. Prasad, M. Chaitanya, C.S. Kumar, M.V.B. Rao, M. Pal, Bioorg. Med. Chem. Lett. 22, 2186 (2012)
T.S. Jin, J.S. Zhang, A.Q. Wang, T.S. Li, Synth. Commun. 35, 2339 (2005)
H. Tavakol, M. Firouzi, Front. Chem. 10, 1015830 (2022)
T.S. Jin, J.S. Zhang, J.C. Xiao, A.Q. Wang, T.S. Li Synlett 5, 866 (2004)
S. Kantevari, R. Bantu, L. Nagarapu Arkivoc 16(6), 136 (2006)
D.Q. Shi, Q.Y. Zhuang, J. Chen, X.S. Wang, S.J. Tu, H.W. Hu, Chin. J. Org. Chem. 23, 694 (2003)
T.S. Jin, J.S. Zhang, A.Q. Wang, F.S. Zhang, Chin. J. Org. Chem. 25, 335 (2005)
J.J. Ma, X. Zhou, X.H. Zhang, C. Wang, Z. Wang, J.C. Li, Q. Li Aus. J. Chem. 60, 146 (2007)
M. Salami, A. Ezabadi, Polycyclic Aromat. Compd. 42, 3377 (2022)
S. Kamalifar, H. Kiyani, Polycyclic Aromat. Compd. 42, 3675 (2022)
F. Darviche, S. Balalaie, F. Chadegani, P. Salehi. Synth. Commun. 37, 1059 (2007)
J.E. Thai, M.C. Roach, M. M. Reynolds Front Chem. 11, 1259835 (2023)
M. Karegar, M.M. Khodaei, Res. Chem. Intermed. 47, 4537 (2021)
S.A. Shaikh, V.S. Kamble, S.T. Salunkhe, S.K. Patil, B.D. Aghav, Org. Prep. Proced. Int. 55, 393 (2023)
S. Kohli, N. Sharma, R. Chandra, Appl. Organomet. Chem. 37, e7049 (2023)
Y. Merroun, S. Chehab, A.E. Hallaoui, T. Guedira, S. Boukhris, R. Ghailane, A. Souizi, J. Mol. Struct. 1294, 136383 (2023)
P.G. Kargar, G. Bagherzade, H. Eshghi, RSC Adv. 11(8), 4339 (2021)
F. Mohamadpour, Org. Prep. Proced. Int. 53, 59 (2021)
G. Rathee, S. Kohli, N. Singh, A. Awasthi, R. Chandra, ACS Omega 5, 15673 (2020)
F.G. Kahangi, M. Mehrdad, M.M. Heravi, Sci. Rep. 10, 15285 (2020)
P. Bansal, G. Chaudhary, N. Kaur, S.K. Mehta, RSC Adv. 5, 8205 (2015)
F. Taghavi, M. Gholizadeh, A.S. Saljooghi, M. Ramezanib, RSC Adv. 6, 87082 (2016)
Z.G. Kohi, K.R. Moghadum, S.T. Roudsari, Mole. Catal. 548, 113417 (2023)
B. Maleki, S. Barzegar, Z. Sepehr, J. Iran. Chem. Soc. 9, 757 (2012)
E. Hataminejad, A. Ezabadi, Res. Chem. Intermed. 48, 2535 (2022)
M. Shiri, M.A. Zolfigol, Tetrahedron 65, 587 (2009)
H. Firouzabadi, N. Iranpoor, A. Garzan, Adv. Synth. Catal. 347, 1925 (2005)
K.L. Penniston, S.Y. Nakada, R.P. Holmes, D.G. Assimos, J. Endourol. 22(3), 567 (2008)
M.O.H. Helali, M. Ibrahim, M.Z. Shafique, M.M. Rahman, S.K. Biswas, M.S. Islam, J. bio. sci. 16, 125 (2008)
S. Ranganna, V.S. Govindarajan, K.V. Ramana, Crit. Rev. Food Sci. Nutr. 18(4), 313 (1983)
S. Ranganna, V.S. Govindarajan, K.V. Ramana, Crit. Rev. Food Sci. Nutr. 19(1), 1 (1983)
M.B. Deshmukh, S.S. Patil, S.D. Jadhav, P.B. Pawar, Synth. Commun. 42, 1177 (2012)
S. Patil, S. Jadhav, M.B. Deshmukh, J. Chem. Sci. 125, 851 (2013)
S.S. Patil, S.D. Jadhav, U.P. Patil, Arch. Appl. Sci. Res. 4, 1074 (2012)
S.D. Jadhav, R.C. Patil, A.A. Jagdale, S.S. Patil, Res. Chem. Intermed. 48, 593 (2022)
S.S. Patil, S.D. Jadhav, M.B. Deshmukh, Ind. J. Chem. Sec. B 52B, 1172 (2013)
S.T. Morbale, S.D. Jadhav, M.B. Deshmukh, S.S. Patil, RSC Adv. 5, 84610 (2015)
S.T. Morbale, S.K. Shinde, S.D. Jadhav, M.B. Deshmukh, S.S. Patil, Der Pharm. Lett. 7, 169 (2015)
B.M. Patil, S.K. Shinde, A.A. Jagdale, S.D. Jadhav, S.S. Patil, Res. Chem. Intermed. 47, 4369 (2021)
M. Bustamante, N. Duran, M.C. Diez, Soil Sci. Plant Nutr. 12, 667 (2012)
E. Fuguet, C. Ràfols, M. Rosés, E. Bosch, Anal. Chim. Acta 548, 95 (2005)
K. Hanna, R. Denoyel, I. Beurroies, J.P. Dubès, Coll. Surf. A Physicochem. Eng. Asp. 254, 231 (2005)
D.A. Edwards, R.G. Luthy, Z. Liu, Environ. Sci. Technol. 25, 127 (1991)
D.J.L. Prak, P.H. Pritchard, Water Res. 36, 3463 (2002)
L.A. Moreira, A. Firoozabadi, Langmuir 25, 12101 (2009)
Acknowledgements
The authors would like to express their thankfulness to the Indian Institute of Chemical Technology (IICT), Hyderabad, for the NMR analysis.
Author information
Authors and Affiliations
Contributions
SDJ and SSP contributed equally to the experiment design and direction, with SDJ and BMP performing the laboratory work. The authors jointly prepared the draft manuscript, and finalized the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jadhav, S.D., Patil, B.M. & Patil, S.S. An eco-friendly approach to synthesize arylmethylene bis(3-hydroxy-5,5’-dimethyl-2-cyclohexene-1-ones) and 1,8-dioxo-octahydroxanthenes. Res Chem Intermed (2024). https://doi.org/10.1007/s11164-024-05274-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11164-024-05274-w