Abstract
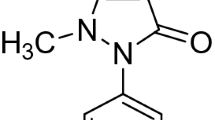
Complexes of sulfadoxine as a pharmaceutical ligand with Ca(II), Mg(II), Zn(II), Fe(III), and VO(II), were synthesized and characterized by microanalysis, conductance, infrared and thermogravimetric (TGA/DTG and DTA) measurements. The ligand can be coordinate as a bidentate feature via (C=N) and (S=O) groups. Sulfadoxine ligand as well as their complexes has been checked against some kinds of bacteria and fungi which gave a significant effect. The kinetic thermodynamic parameters such as: activation of E*, ΔH*, ΔS*, and ΔG* were estimated using Coats-Redfern as well as Horowitz-Metzger equations.
Similar content being viewed by others
References
Moulton, B., and Zaworotko, M.J., Chem. Rev., 2001, vol. 101, p. 1629.
Wu, C.D., Lu, C.Z., Zhuang, H.H., and Huang, J.S., J. Am. Chem. Soc., 2002, vol. 124, p. 3836.
Dybtsev, D.N., Chun, H., and Kim, K., Angew. Chem. Int. Ed., 2004, vol. 43, p. 5033.
Lpez-Gresa, M.P., Ortiz, R., Perell, L., Latorre, J., Liu-Gonzalez, M., Garcıa-Granda, S., Perez-Priede, M., and Cantn, E., J. Inorg. Biochem., 2002, vol. 92, p. 65.
Yuan, R.X., Xiong, R.G., Abrahams, B.F., Lee, G.H., Peng, S.M., Che, C.M., and You, X.Z., J. Chem. Soc. Dalton Trans, 2001, p. 2071.
Turel, I., Coord. Chem. Rev., 2002, vol. 232, p. 27.
Xiao, D.R., He, J.H., Sun, D.Z., Chen, H.Y., Yan, S.W., Wang, X., Yang, J., Yuan, R., and Wang, E.B., Eur. J. Inorg. Chem., 2012, p. 1783.
Drevensek, P., Zupancic, T., Pihlar, B., Jerala, R., Kolitsch, U., Plaper, A., and Turel, I., J. Inorg. Biochem., 2005, vol. 99, p. 432.
Xiao, D.R., Wang, E.B., An, H.Y., Su, Z.M., Li, Y.G., Gao, L., Sun, C.Y., and Xu, L., Chem. Eur. J., 2005, vol. 11, p. 6673.
He, J.H., Xiao, D.R., Chen, H.Y., Yan, S.W., Sun, D.Z., Wang, X., Yang, J., Yuan, R., and Wang, E.B., Inorg. Chim. Acta, 2012, vol. 385, p. 170.
Kathawate, L., Sproules, S., Pawar, O., Markad, G., Haram, S., Puranik, V., Salunke-Gawali, S., J. Mol. Struct., 2013, vol. 1048, p. 223.
Beerlev, W.N., Pelers, W., and Mager, K., Ann. Trop. Med. Parasite, 1960, vol. 26, p. 288.
Hoffman La Roche Co., Swiss Patent no. 416648, 1967.
Sharaby, C., Synth. React. Inorg. Met. Org. Chem., 2005, vol. 35, p. 133.
Basco, L.K., Tahar, R., and Ringwald, P., Antimicrob. Agents and Chemotherapy, 1998, vol. 42, p. 1811.
White, N.J., J. Clinical Investigation, 2004, vol. 113, p. 1084.
Liang, A.B., British Medical J., 1965, vol. 1, p. 905.
Liang, A.B., British Medical J., 1964, vol. 2, p. 1439.
Richards, W.H., Nature, 1966, vol. 212, p. 1494.
Harinasuta, T., Viravan, C., and Reid, H.A., Lancet, 1967, vol. 1, p. 1117.
Laing, A.B., Medical J. Malaya, 1968, vol. 23, p. 5.
Laing, A.B., Trans. Royal Soc. Trop. Med. Hygiene, 1970, vol. 64, p. 562.
Wernsdorfer, W.H., and Payne, D., Pharmacology & Therapeutics, 1991, vol. 50, p. 95.
WHO, Susceptibility of Plasmodium Falciparum to Antimalarial Drugs: Report on Global Monitoring, 1996–2004, WHO/HTM/MAL/2005.1103. Geneva: World Health Organization, 2005.
WHO, A Strategic Framework for Malaria Control and Prevention During Pregnancy in the African Region, WHO Regional Office for Africa, Brazzaville, 2004.
Peters, P.J., Thigpen, M.C., Parise, M.E., and Newman, R.D., Drug Safety: An Int. J. Med. Toxicol. Drug Exp., 2007, vol. 30, p. 481.
Wilson, E.O. and Gisvold, D., Advances in Malaria Chemotherapy, WHO Technical Report Series 71I, New York: Prentice Hall, 1984.
Obaleye, J.A., J. Pure App. Sci., 1996, vol. 13, p. 32.
Astier, H., Renard, C., Cheminel, V., Soares, O., Mounier, C., Peyron, F., and Chaulet, J.F., J. Chromatogr. B, 1997, vol. 698, p. 217.
Bergqvist, Y., Eckerbom, S., Larsson, H., and Malekzadeh, M., J. Chromatogr., 1991, vol. 571, p. 169.
Edstein, M.D., Lika, I.D., Chongsuphajaisiddhi, T., Sabchareon, A., and Webster, H.K., Ther. Drug Monit., 1991, vol. 13, p. 146.
Eljaschewitsch, J., Padberg, J., Schurmann, D., and Ruf, B., Ther. Drug Monit., 1996, vol. 18, p. 592.
Green, M.D., Mount, D.L., and Nettey, H., J. Chromatogr. B, 2002, vol. 767, p. 159.
Sinnaeve, B.A., Decaestecker, T.N., Risha, P.G., Remon, J.P., Verva, C., and Bocxlaer, J.F., J. Chromatogr. A, 2005, vol. 1076, p. 97.
Bhoir, S.I., Bhoir, I.C., Bhagwat, A.M., and Sundaresan, M., J. Chromatogr. B, 2001, vol. 757, p. 39.
Dua, V.K., Sarin, R., and Sharma, V.P., J. Pharm. Biomed. Anal., 1994, vol. 12, p. 1317.
Bustos, D.G., Lazaro, J.E., Gay, F., Pottier, A., Laracas, C.J., Traore, B., and Diquet, B., Trop. Med. Int. Health, 2002, vol. 7, p. 584.
Dzinjalamala, F.K., Macheso, A., Kublin, J.G., Taylor, T.E., Barnes, K.I., Molyneux, M.E., Plowe, C.V., and Smith, P.J., Antimicrob. Agents Chemother., 2005, vol. 49, p. 3601.
Mohamed, G.G., Soliman, A.A., El-Mawgood, M.A., Spectrochim. Acta Part A, 2005, vol. 62, p. 1095.
Obaleye, J.A., Amolegbe, S.A., and Gbotoso, G.O., Proc. of the Second Conference on Science and Mational Development, 10–13 october, 2006.
Abd El-Wahed, M.G., Refat, M.S., and El-Megharbel, S.M., Bull. Mater. Sci., 2009, vol. 32(2), p. 205.
Sekhon, B.S., and Leena G., Int. J. Chem. Tech. Res., 2010, vol. 2(1), p. 286.
Refat, M.S., and Mohamed, S.F., Spectrochim. Acta A, 2011, vol. 82, p. 108.
El-Nawawy, M.A., Farag, R.S., Sabbah, I.A., and Abu-Yamin, A.M., IJPSR, 2011, vol. 2(12), p. 3143.
Sartaj, T., Al-Asbahy, W.M., Afzal, M., Shamsi, M., and Arjmand, F., J. Luminescence, 2012, vol. 132, p. 3058.
Ogunniran, K.O., Ajani, O.O., Ehi-Eromosele, C.O., Obaleye, J.A., Adekoya, J.A., and Ajanaku, C.O., Int. J. Phy. Sci., 2012, vol. 7(13), p. 1998.
Alaghaz Abdel-Nasser M.A., Bayoumi, H.A., Ammar, Y.A., and Aldhlmani, S.A., J. Mol. Struct., 2013, no. 1035, pp. 383–399.
Ebrahimi, H., Hadi, J.S., and Al-Ansari, H.S., J. Mol. Struct., 2013, vol. 1039, p. 37.
Gupta, R., Saxena, R.K., Chatarvedi, P., and Virdi, J.S., J. Appl. Bacteriol., 1995, vol. 78, p. 378.
Geary, W.J., Coord. Chem. Rev., 1971, vol. 7, p. 81.
Kobelnik, M., Cassimiro, D.L., Santos, D.D., Ribeiro, C.A., and Crespi, M.S., Chinese J. Chem., 2011, vol. 29(11), p. 2271.
Obaleye, J.A., Adeyemi, O.G., and Balogun, E.A., Int. J. Chem., 2001, vol. 11, p. 101.
Vogel, T., Vogel Textbook of Practical Organic Chemistry. 4 ed., John Wiley Inc., 1989, pp: 133–325.
Iqbal, S.A., Sibi, J., and Jacob, G., Oriental J. Chem., 2011, vol. 27, no. 2, p. 731.
Nakamoto, K., Infrared Spectra of Inorganic and Coordination Compounds, New York: Wiley Interscience, 1970.
Bhattacharyya, S., Mukhopadhyay, S., Samanta, S., Weakley, T.J.R., and Chaudhury, M., Inorg. Chem., 2002, vol. 41, p. 2433.
Al-Saif, F.A., and Refat, M.S., Russ. J. Gen. Chem., 2014, vol. 84, no. 1, p. 143.
Sharma, S., Iqbal, S.A., and Bhattacharya, M., Oriental J. Chem., 2009, vol. 25, p. 1101.
Abd El-Wahed, M.G., El-Megharbel, S.M., El-Sayed, M.Y., Zahran, Y.M., and Refat, M.S., Russ. J. Gen. Chem., 2013, vol. 83, no. 12, p. 2438.
Coats, A.W. and Redfern, J.P., Nature, 1964, vol. 201, p. 68.
Horowitz, H.W. and Metzger, G., Anal. Chem., 1963, vol. 35, p. 1464.
Refat, M.S., Mohamed, G.G., Ibrahim, M.Y., Killa, H.M.A., and Fetooh, H., Russ. J. Gen. Chem., 2013, vol. 83, no. 12, p. 2479.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Zaky, M., El-Sayed, M.Y., El-Megharbel, S.M. et al. Synthesis, chemical structure elucidation, and biological studies on the effect of some vital metal ions on sulfadoxine. Russ J Gen Chem 85, 176–184 (2015). https://doi.org/10.1134/S1070363215010314
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363215010314