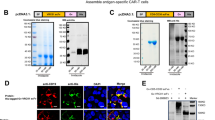
Abstract
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is the only curative therapy for hematopoietic malignancies. The graft-derived donor lymphocytes are capable of eliminating the residual recipient malignant cells in the course of allogeneic immune response, thus decreasing the chances of a relapse of the disease. Foreign peptides of the recipient presented by the MHC molecules are able to elicit the immune response immunologically. These polymorphic peptides are known as minor histocompatibility antigens (MiHAs). MiHAs occur due to the nonsynonymous single nucleotide polymorphisms in human genome. Transfusion of T cells specific to MiHAs presented predominantly in the cells of hematopoietic origin will allow the targeted elimination of residual malignant clones avoiding undesirable damage to healthy tissues. To induce the immune response, the donor must be homozygous by the MiHA allele and the recipient must either be homozygous or heterozygous by the alternative MiHA allele. The therapeutic mismatch occurs in 25% of cases under the optimal frequency of allelic variants. Minor antigen ACC-1Y originates from polymorphism in the BCL-2A1 gene; its immunogenic mismatch occurrence approaches the theoretical maximum. In addition, BCL2A1 is overexpressed in cells of various lymphomas. ACC-1Y is presented on allele HLA-A*24:02, which is relatively frequent in the Russian population. Combination of these factors makes the minor antigen ACC-1Y a promising target for immunotherapy. Transfusion of donor CD8+ lymphocytes modified with transgenic MiHA-specific TCR is one of the promising methods of posttransplant leukemia therapy and relapse prophylaxis. We obtained a sequence of high-affinity ACC-1Y-specific TCR after the antigen-specific expansion of T cells derived from a healthy ACC-1Y–/– donor. We cloned this sequence into the lentiviral vector and obtained the assembled viral particles. Further, we transduced the CD8+ lymphocyte culture and demonstrated its antigen-specific cytotoxic activity. It is suggested that CD8+ lymphocytes modified by the described method could be potentially transferred to recipients as a therapy against relapse after allo-HSCT.




Similar content being viewed by others
REFERENCES
Barrett A.J., Battiwalla M. 2010. Relapse after allogeneic stem cell transplantation. Expert. Rev. Hematol. 3 (4), 429‒441.
Goulmy E.L.S., Schipper R., Pool J., Blokland E., Falkenburg F., Vossen J., Gratwohl A., Vogelsang G.B., van Houwelingen H.C., Van Rood J.J. 1996. Mismatches of minor histocompatibility antigens between HLA-identical donors and recipients and the development of graft-versus-host disease after bone marrow transplantation. N. Engl. J. Med. 334 (5), 281–285.
Wallny H.J., Rammensee H.G. 1990. Identification of classical minor histocompatibility antigen as cell-derived peptide. Nature. 343 (6255), 275.
Efimov G.A., Vdovin A.S., Grigor’ev A.A., Fil’kin S.Yu., Bykova N.A., Savchenko V.G. 2015. Immunobiology of acute graft-versus-host reaction. Med. Immunol. 17 (6), 499‒516.
Heemskerk M.H., Hoogeboom M., Hagedoorn R., Kester M.G., Willemze R., Falkenburg J.F. 2004. Reprogramming of virus-specific T cells into leukemia-reactive T cells using T cell receptor gene transfer. J. Exp. Med. 199 (7), 885–894.
Granados D.P., Rodenbrock A., Laverdure J.P., Côté C., Caron-Lizotte O., Carli C., Pearson H., Janelle V., Durette C., Bonneil E., Roy D.C., Delisle J-S., Lemieux S., Thibault P. 2016. Proteogenomic-based discovery of minor histocompatibility antigens with suitable features for immunotherapy of hematologic cancers. Leukemia. 30 (6), 1344.
Bykova N.A., Malko D.B., Efimov, G.A. 2018. In silico analysis of minor histocompatibility antigen landscape based on 1000 Genomes project. Front. Immunol. 9, 1819.
Nishida T., Akatsuka Y., Morishima Y., Hamajima N., Tsujimura K., Kuzushima K., Kodera Y., Takahashi T. 2004. Clinical relevance of a newly identified HLA-A*24:02-restricted minor histocompatibility antigen epitope derived from BCL2A1, ACC-1, in patients receiving HLA genotypically matched unrelated bone marrow transplant. Br. J. Haematol. 124 (5), 629–635.
Warren E.H., Fujii N., Akatsuka Y., Chaney C.N., Mito J.K., Loeb K.R., Gooley T.A., Brown M.L., Koo K.K.W., Rosinski K.V., Ogawa S., Matsubara A., Appelbaum F.R., Riddell S.R. 2010. Therapy of relapsed leukemia after allogeneic hematopoietic cell transplant with T cells specific for minor histocompatibility antigens. Blood. 115 (19), 3869–3878.
Vdovin A.S., Bykova N.A., Efimov G.A. 2017. T Lymphocytes with modified specificity in the therapy of malignant diseases. Mol. Biol. (Moscow). 51 (6), 874–886.
Dossa R.G., Cunningham T., Sommermeyer D., Medina-Rodriguez I., Biernacki M.A., Foster K., Bleakley M. 2018. Development of T-cell immunotherapy for hematopoietic stem cell transplantation recipients at risk of leukemia relapse. Blood. 131 (1), 108–120.
White M., Whittaker R., Gandara C., Stoll E.A. 2017. A guide to approaching regulatory considerations for lentiviral-mediated gene therapies. Hum. Gene Ther. Methods. 28 (4), 163–176.
Romaniuk D., Postovskaya A., Khmelevskaya A., Malko D., Efimov G.A. 2018. Rapid multiplex genotyping of 20 HLA-A*02:01 restricted minor histocompatibility antigens. Front. Immunol. (in press).
Wölfl M., Greenberg P.D. 2014. Antigen-specific activation and cytokine-facilitated expansion of naive, human CD8+ T cells. Nat. Protoc. 9 (4), 950.
Vdovin A.S., Filkin S.Yu., Efimova P.R., Shitikov S.A., Kapranov N.M., Davydova Yu.O., Egorov E.S., Khamaganova E.G., Drokov M.Yu., Kuzmina L.A., Parovichnikova E.N., Efimov G.A., Savchenko V.G. 2016. Recombinant MHC tetramers for isolation of virus-specific CD8+ cells from healthy donors: Potential approach for cell therapy of posttransplant cytomegalovirus infection. Biochemistry (Moscow). 81 (11), 1371–1383.
Toebes M., Rodenko B., Ovaa H., Schumacher T.N. 2009). Generation of peptide MHC class I monomers and multimers through ligand exchange. Curr. Protoc. Immunol. 87 (1), 18–16.
Mamedov I.Z., Britanova O.V., Zvyagin I.V., Turchaninova M.A., Bolotin D.A., Putintseva E.V., Lebedev Y.B., Chudakov D.M. 2013. Preparing unbiased T-cell receptor and antibody cDNA libraries for the deep next generation sequencing profiling. Front. Immunol. 4, 456.
Donnelly M.L., Luke G., Mehrotra A., Li X., Hughes L.E., Gani D., Ryan M.D. 2001. Analysis of the aphthovirus 2A/2B polyprotein ‘cleavage’ mechanism indicates not a proteolytic reaction, but a novel translational effect: A putative ribosomal ‘skip’. J. Gen. Virol. 82 (5), 1013–1025.
Janelle V., Carli C., Taillefer J., Orio J., Delisle J.S. 2015. Defining novel parameters for the optimal priming and expansion of minor histocompatibility antigen-specific T cells in culture. J. Transl. Med. 13 (1), 123.
Oostvogels R., Lokhorst H.M., Mutis T. 2016. Minor histocompatibility Ags: Identification strategies, clinical results and translational perspectives. Bone Marrow Transplant. 51 (2), 163.
van Loenen M.M., de Boer R., Amir A.L., Hagedoorn R.S., Volbeda G.L., Willemze R., van Rood J.J., Falkenburg J.H.F., Heemskerk M.H. 2010. Mixed T cell receptor dimers harbor potentially harmful neoreactivity. Proc. Natl. Acad. Sci. U. S. A. 107 (24), 10972–10977.
Roth T.L., Puig-Saus C., Yu R., Shifrut E., Carnevale J., Li P.J., Hiatt J., Saco J., Krystofinski P., Li H., Tobin V., Nguyen D.N., Lee M.R., Putnam A.L., Ferris A.L., et al. 2018. Reprogramming human T cell function and specificity with non-viral genome targeting. Nature. 559 (7714), 405.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by I. Shipounova
Rights and permissions
About this article
Cite this article
Pilunov, A.M., Kuchmiy, A.A., Sheetikov, S.A. et al. Modification of Cytotoxic Lymphocytes with T Cell Receptor Specific for Minor Histocompatibility Antigen ACC-1Y. Mol Biol 53, 402–410 (2019). https://doi.org/10.1134/S0026893319030142
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893319030142