Abstract
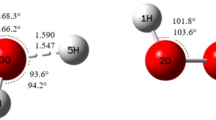
The geometric structures, vibration frequencies, and energies of the reactants, products, and transition states in the decomposition of C5H4O were evaluated by quantum chemical calculations using the CCSD(T)-F12/vtz-f12B method. The calculated energy barriers for the two most probable pyrolysis pathways of C5H4O, equal to 96.3 and 96.5 kcal/mol, respectively, are evidence that the pyrolysis proceeds at a high temperature, and the most likely reaction products are vinylacetylene and carbon monoxide. It is shown that the formation of products such as cyclobutadiene, acetylene, and propadienal can be explained by the occurrence of an energetically favorable pathway.
Similar content being viewed by others
References
M. Frenklach, “Reaction Mechanism of Soot Formation in Flames,” Proc. Natl Acad. Sci. USA, No. 4, 2028–2037 (2002).
C. A. Taatjes, D. L. Osborn, T. M. Selby, et al., “Products of the Benzene + O(3P) Reaction,” J. Phys. Chem. A 9 (114), 3355–3370 (2010).
H. Richter and J. B. Howard, “Formation of Polycyclic Aromatic Hydrocarbons and their Growth to Soot—A Review of Chemical Reaction Pathways,” Prog. Energy Combust. Sci. 4 (26), 565–608 (2000).
A. M. Starik, N. S. Titova, and S. A. Torokhov, “Kinetics of Oxidation and Combustion of Complex Hydrocarbon Fuels: Aviation Kerosene,” Fiz. Goreniya Varyva 49 (4), 12–30 (2013) [Combust. Expl. Shock Waves 49 (4), 392–408 (2013)].
S. Takamasa, N. Masakazu, and M. Akira, “High-Temperature Reactions of OH Radicals with Benzene and Toluene,” J. Phys. Chem. A, No. 110, 5081–5090 (2006).
H. X. Zhang, S. I. Ahonkhai, and M. H. Back, “Rate Constants for Abstraction of Hydrogen from Benzene, Toluene, and Cyclopentane by Methyl and Ethyl Radicals over the Temperature Range 650–770 K,” Can. J. Chem. 67 (10), 1541–1549 (1989).
I. V. Tokmakov, G.-S. Kim, V. V. Kislov, et al., “The Reaction of Phenyl Radical with Molecular Oxygen: AG2M Study of the Potential Energy Surface,” J. Phys. Chem. A 109 (27), 6114–6127 (2005).
D. S. N. Parker, R. I. Kaiser, T. P. Troy, et al., “Toward the Oxidation of the Phenyl Radical and Prevention of PAH Formation in Combustion Systems,” J. Phys. Chem. A 119 (28), 7145–7154 (2014).
W. M. Baird, “Carcinogenic Polycyclic Aromatic Hydrocarbon-dna Adducts and Mechanism of Action,” Environ. Mol. Mutagen. 45 (2-3), 106–114 (2005).
B. J. Finlayson-Pittsa and J. N. Pitts, “Tropospheric Air Pollution: Ozone, Airborne Toxics, Polycyclic Aromatic Hydrocarbons, and Particles,” Science 276, 1045–1052 (1997).
H. Richter and J. B. Howard, “Formation and Consumption of Single-Ring Aromatic Hydrocarbons and Their Precursors in Premixed Acetylene, Ethylene and Benzene Flames,” Phys. Chem. 4, 2038–2055 (2002).
D. S. N. Parker, F. Zhang, Y. S. Kim, et al., “Low Temperature Formation of Naphthalene and Its Role in the Synthesis of Pahs (Polycyclic Aromatic Hydrocarbons) in the Interstellar Medium,” Proc. Natl Acad. Sci. USA 109 (1), 53–58 (2012).
C. Venkat, K. Brezinsky, and I. Glassman, “High Temperature Oxidation of Aromatic Hydrocarbons,” Proc. Combust. Inst. 19, 143–152 (1982).
A. R. Ghildina, A. D. Oleinikov, A. M. Mebel, and V. N. Azyazov, “Products of Reaction C5H4O + H: Quantum-Chemical Studies,” in Nonequilibrium Processes in Physics and Chemistry (Torus Press, Moscow, 2016), pp. 50–56.
D. J. Robichaud, A. M. Scheer, C. Mukarakate, et al., “Unimolecular Thermal Decomposition of Dimethoxybenzenes,” J. Chem. Phys. 140 (23), 234302 (2014).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, et al., Gaussian 09, Rev. B.01 (Gaussian, Inc.,Wallingford, 2010).
H.-J. Werner, P. J. Knowles, R. Lindh, et al. MOLPRO, Ver. 2012.1. A Package of ab initio Programs (Univ. Coll. Cardiff Consult., Cardiff, 2012), p. 555.
G. E. Scuseria, C. L. Janssen, and H. F. Schaefer, “An Efficient Reformulation of the Cclosed-Shell Coupled Cluster Single and Double Excitation (CCSD) Equations,” J. Chem. Phys. 89, 7382 (1988).
G. E. Scuseria and H. F. Schaefer, “Is Coupled Cluster Singles and Doubles (CCSD) More Computationally Intensive than Guadratic Configuration Interaction (QCISD)?” J. Chem. Phys. 90, 3700 (1989).
T. B. Adler, G. Knizia, and H.-J. Werner, “A Simple and Efficient CCSD(T)-F12 Approximation,” J. Chem. Phys. 127, 221106 (2007).
M. Y. Zhang, C. Wesdemiotis, M. Marchetti, et al., “Characterization of Four C4H4 Molecules and Cations by Neutralization-Reionization Mass Spectrometry,” J. Amer. Chem. Soc. 111, (22), 8341–8346 (1989).
H. Wang and K. Brezinsky, “Computational Study on the Thermochemistry of Cyclopentadiene Derivatives and Kinetics of Cyclopentadienone Thermal Decomposition,” J. Phys. Chem. A 102 (9), 1530–1541 (1998).
A. M. Mebel, V. V. Kislov, and R. I. Kaiser, “Ab Initio/Rice–Ramsperger–Kassel–Marcus study of the Singlet C4H4 Potential Energy Surface and of the Reactions of C2(X1S+ g ) with C4H4(X1A+ 1g) and C(1D) with C3H4 (Allene and Methylacetylene),” J. Chem. Phys. 125 (13), 133113 (2006).
A. M. Scheer, C. Mukarakate, D. J. Robichaud, et al., “Thermal Decomposition Mechanisms of the Methoxyphenols: Formation of Phenol, Cyclopentadienone, Vinylacetylene and Acetylene,” J. Phys. Chem. A 115, 11381–11389 (2011).
Y. Georgievskii, J. A. Miller, M. P. Burke, and S. J. Klippenstein, “Reformulation and Solution of the Master Equation for Multiple-Well Chemical Reactions,” J. Phys. Chem. A, No. 117, 12146 (2013).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.R. Ghildina, A.M. Mebel, I.A. Medvedkov, V.N. Azyazov.
Rights and permissions
About this article
Cite this article
Ghildina, A.R., Mebel, A.M., Medvedkov, I.A. et al. Quantum-Chemical Calculations of the Primary Reactions of Thermal Decomposition of Cyclopentadienone. Combust Explos Shock Waves 54, 9–15 (2018). https://doi.org/10.1134/S0010508218010021
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0010508218010021