Abstract
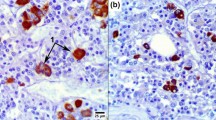
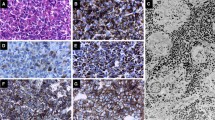
In order to elucidate the effects of hypothalamic regulation on the morphology of GH cells, light and electron microscopic immunocytochemical examinations were carried out comparing GH cells in the anterior pituitary gland of anencephalic fetus with those of normal fetuses. Three types of GH cells were identifted in the anterior pituitary gland of anencephalic fetus as well as in the normal fetus. Type1 is a small, round cell containing a few small secretory granules. Type-III is a large, polygonal cell with numerous large secretory granules. Type-II is a polygonal cell with medium-sized secretory granules. The Type-II GH cell was predominant in both anencephalic and normal fetuses. The most striking difference between anencephalic and normal fetuses was the presence of atypical forms of the Type II cell. These were polygonal cells containing secretory granules, which were either immunopositive or imrnunonegative to anti-human GH (anti-hGH) serum. Furthermore, two other types of GH cells were identified. The somatomammotroph (SM cell) contained GH and PRL. in different granules within the same cell. Also. a different type of the GH cell was noted containing two varieties of secretory granules; one was immunolabeled only with anti-hGH and the other was not irnmunolabeled to either anti-hGH or anti-human PRL (anti-hPRL). From these results, we suggest that an absence of hypothalamic regulation in the anencehpalic does not seriously modify GH cell morphology but induces an altered GH storage pattern in some of the cells.
Similar content being viewed by others
Reference
Guillemin R, Brazeau P, Bohlen P et al.: Growth hormone-releasing factor from a human pancreatic tumor that caused acromegaly. Science 218: 585–587, 1982.
Rivier J, Spiess J, Thorner M et al.: Characterization of a growth hormone-releasing factor from a human pancreatic islet tumour. Nature 300: 276–278, 1982.
Fukata J, Diamond DJ and Martin JB: Effects of rat growth hormone (rGH)-releasing factor and somatostatin on the release and synthesis of rGH in dispersed pituitary cells. Endocrinology 117: 457–467, 1985.
Barinaga M, Yamonoto G, Rivier C et al.: Transcriptional regulation of growth hormone gene expression by growth hormone-releasing factor. Nature 306: 84–85, 1983.
Barinaga M, Bilezikjian LM, Vale WW et al.: Independent effects of growth hormone releasing factor on growth hormone release and gene transcription. Nature 314: 279–281, 1985.
Billestrup N, Swanson LW and Vale W: Growth hormone-releasing factor stimulates proliferation of somatotrophs in vitro. Proc. Natl. Acad. Sci. USA 83: 6854–6857, 1986.
Bileziknjian LM and Vale WW: Stimulation of adenosine 3′, 5′ monophosphateproduction by growth hormone-releasing factor and its inhibition by somatostatin in anterior pituitary cells in vitro. Endocrinology 113: 1726–1731, 1983.
Koch BD and Schonbrunn A: The somatostatin receptor is directly coupled to adenylate cyclase in GH4Cl 1 pituitary cell membranes. Endocrinology 114: 1784–1790, 1984.
Kaplan SL, Grumbach MM and Shepard TH: The ontogenesis of human fetalhormones: I. Growth hormone and insulin. J. Clin. Invest. 51: 3080–3093, 1972.
Kaplan SL, Grumbach MM and Aubert ML: The ontogenesis of pituitary hormones and hypothalamic factors in the human fetus: Maturation of central nervous system regulation of anterior pituitary function. Recent Progress in Hormone Research 32: 161–235, 1976.
Cavallo L, Altomare M, Palmieri P et al.: Endocrine function in four anencephalic infants. Hormone Res. 15: 159–166, 1981.
Tachibana T, Ito T and Kwon OH (Gon G): Immunoelectron microscopic study of the GH cell in the anterior pituitary gland of normal human fetus. Anat. Rec. 239: 177–184, 1994.
Mollenhauer HH: Plastic embedding mixtures for use in electron micrcscopy. Stain Technol. 39: 111–114, 1964.
Aubert ML, Grumbach MM and Kaplan SL The ontogenesis of human fetal hormones. IV. Somatostatin, luteinizing hormone releasing factor, and thyrotropin releasing factor in hypothalamus and cerebral cortex of human fetuses 10–22 weeks of age. J. Clin. Endocrinol. Metab. 44: 1130–1141, 1977.
Bugnon C, Fellmann D and Bloch B: Immunocytochemical study of the ontogenesis of the hypothalamic somatostatin-containing neurons in the human fetus. Metabolism (Suppl. 1) 27: 1161–1165, 1978.
Bresson JL, Clavequin MC, Fellmann D et al.: Ontogeny of the neuroglandular system revealed with HPGRF 44 antibodies in human hypothalamus. Neuroendocrinology 39: 68–73, 1984.
Hayek A, Driscoll SG and Warshaw JB: Endocrine shdies in anencephaly. J. Clin. Invest. 52: 1636–1641, 1973.
Furuhashi N, Fukaya T, Kono H et al.: Cord serum growth hormone in the human fetus. Sex difference and a negative correlation with birth weight. Gynecol. Obstet. Invest. 16: 119–124, 1983.
Arosio M, Cortelazzl D, Persam L et al.: Circulating levels of growth hormone, insulin-like growth factor-1 and prolactin in normal, growth retarded and anencephalic human fetuses. J. Endocrinol. Invest. 18: 346–353, 1995.
Track NS, Creutzfeldt C, Litzenberger J et al.: Appearance of gastrin and somatostatin in the human fetal stomach, duodenumand pancreas. Digestion 19 292–306, 1979.
Rahier J, Wallon J and Henquin JC: Abundance of somatostatin cells in the human neonatal pancreas. Diabetologia 18: 251–254, 1980.
Bryant MG, Buchan AM, Gregor M et al.: Development of intestinal regulatory peptides in the human fetus. Gastroenterology 83: 47–54, 1982.
Itzev D, Lolova I and Davidoff M: Immunocytochemical and electron microscopical data on the differentiation of somatostatin-containing endocrine cells in human large intestine. Anat Anz. 166: 77–85, 1988.
Dubois PM, Paulin C, Assan R et al.: Human fetal pancreas evidence for immunoreactive somatostatin in the endocrine cells. Nature 256: 731–732, 1975.
Dubois PM and Paulin C: Gastrointestinal somatostatin cells in the human fetus. Cell Tiss. Res. 166: 179–184, 1976.
Kraicer J and Chow AE: Release of growth hormone from purified somatotrophs: use of perifusion system to elucidate interrelations among Ca++, adenosine3′, 5′-monophosphate, and somatostatin. Endocrinology 111: 1173–1180, 1982.
Lin SC, Lin CR, Gukovsky I et al.: Molecular basis of the little mouse phenotype and implications for cell type-specific growth. Nature 364: 208–213, 1993.
Mayo KE, Hammer RE, Swanson LW et al.: Dramatic pituitary hyperplasia in transgenic mice expressing a human growth hormone-releasing factor gene. Mol. Endocrinol. 2: 606–612, 1988.
Lloyd RV, Gikas PW and Chandler WF: Prolactin and growth hormone-producing pituitary adenomas. An immunohistochemical and ultrastructural study. Am. J. Surg. Pathol. 7: 251–260, 1983.
Felix IA, Horvath E, Kovacs K et al.: Somatomammotroph adenoma of the pituitary associated with gigantism and hyperprolactinemia. A morphological study including immunoelectron microscopy. Acta Neuropathol. 71: 76–82, 1986.
Beckers A, Courtoy R, Stevenaert A et al.: Somatomammotropes in human pituitary adenomas as revealed by electron microscopic double gold immunostaining method. Acta Endocrino. 118: 503–512, 1988.
Robert F, Pelletier G, Serri O et al.: Mixed growth hormone and prolactin-secreting human pituitary adenomas: a pathologic, immunocytochemical, ultrastructural, and immunoelectron microscopic study. Hum. Pathol. 19: 1327–1334, 1988.
Reznik M and Hennen G: Somatomammotropes in human pituitary adenomas electron microscopic double gold immunostaining method. Acta Endocrinol. 118: 503–512, 1988.
Lloyd RV, Cano M, Chandler WF et al.: Human growth hormone and prolactin secreting pituitary adenomas analyzed by in situ hybridization. Am. J. Pathol. 134: 605–613, 1989.
Ishikawa H, Nogami H, Kamio M et al.: Single secretory granules contain both GH and Prolactin in pituitary mixed type of adenoma. Virchows Arch. 399: 221–226, 1983.
Mulchahey JJ and Jaffe RB: Detection of a potential progenitor cell in the human fetal pituitary that secretes both growth hormone and prolactin. J. Clin. Endocrinol. Metab. 66: 24–32, 1988.
Leong DA, Lau SK, Sinha YN et al.: Enumeration of lactotropes and somatotropes among male and female pituitary cells in culture: evidence in favor of a somatomammotrope subpopulation in the rat. Endocrinology 116: 1371–1378, 1985.
Hoeffler JP, Boockfor FR and Frawley LS: Ontogeny of prolactin cells in neonatal rats: initial prolactin secretors also release growth hormone. Endocrinology 117: 187–195, 1985.
Asa SL, Kovacs K, Horvath E et al.: Human fetal adenohypophysis: Electron microscopic and ultrastructural immunocytochemical analysis. Neuroendocrinology 48: 423–431, 1988.
Chihara K, Iwasaki J, Minamitani N et al.: Effect of vasoactive intestinal polypeptide on growth hormone secretion in perifused acromegalic pituitary adenoma tissues. J. Clin. Endocrino1.Metab. 54: 773–779, 1982.
Steel JH, Gon G, O’Halloran DJ et al.: Galanin and vasoactive intestinal polypeptide are colocalized with classical pituitary hormones and show plasticity of expression. Histochemistry 93: 183–189, 1989.
Brown ER, Roth KA, Krause JE: Sexually dimorphic distribution of substance P in specific anterior pituitary cell populations. Proc. Natl. Acad. Sci. USA 88: 1222–1226, 1991.
Sako T, Mera F, Shimada T et al.: Glucagon-like material in the human and bovine pituitaries as revealed by immunocytochemistry. Jikeikai Med. J. 36: 21–27, 1989.
Yamamoto T, Katsumata N, Tachibana K et al.: Distribution of a novel peptide in the anterior pituitary, gastric pyloric gland, and pancreatic islets of rat. J. Histochem. Cytochem. 40: 221–229, 1992.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tachibana, T., Ito, T. Immunocytochemical study of the GH cells in the anterior pituitary gland of human fetus II. Anencephalic fetus. Hum Cell 16, 205–215 (2003). https://doi.org/10.1111/j.1749-0774.2003.tb00155.x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1111/j.1749-0774.2003.tb00155.x