Abstract
Under normal physiological conditions, the brain utilizes only a small number of carbon sources for energy. Recently, there is growing molecular and biochemical evidence that other carbon sources, including fructose, may play a role in neuroenergetics. Fructose is the number one commercial sweetener in Western civilization with large amounts of fructose being toxic, yet fructose metabolism remains relatively poorly characterized. Fructose is purportedly metabolizedvia either of two pathways, the fructose-1-phosphate pathway and/or the fructose-6-phosphate pathway. Many early metabolic studies could not clearly discriminate which of these two pathways predominates, nor could they distinguish which cell types in various tissues are capable of fructose metabolism. In addition, the lack of good physiological models, the diet-induced changes in gene expression in many tissues, the involvement of multiple genes in multiple pathways involved in fructose metabolism, and the lack of characterization of some genes involved in fructose metabolism have complicated our understanding of the physiological role of fructose in neuro-energetics. A recent neuro-metabolism study of the cerebellum demonstrated fructose metabolism and co-expression of the genes specific for the fructose 1-phosphate pathway, GLUT5 (glut5) and ketohexokinase (khk), in Purkinje cells suggesting this as an active pathway in specific neurons? Meanwhile, concern over the rapid increase in dietary fructose, particularly among children, has increased awareness about how fructose is metabolizedin vivo and what effects a high fructose diet might have. In this regard, establishment of cellular and molecular studies and physiological characterization of the important and/or deleterious roles fructose plays in the brain is critical. This review will discuss the status of fructose metabolism in the brain with special reference to the cerebellum and the physiological roles of the different pathways.
Similar content being viewed by others
References
Yudkin J, Kang SS, Bruckdorfer KR. Effects of high dietary sugar. Br Med J. 1980;281:1396.
Anderson TA. Recent trends in carbohydrate consumption. Annu Rev Nutr. 1982;2:113–32.
Cox TM. The genetic consequences of our sweet tooth. Nat Rev Genet. 2002;3:481–7.
Steinmann B, Gitzelmann R, Van denBerghe G. In: Scriver C, Beaudet A, Sly W, Valle D, editors. Disorders of fructose metabolism. The metabolic and molecular basis of inherited disease.. 8th ed, New York: McGraw-Hill, Inc, 2001. pp 1489–520.
Mayes PA. Intermediary metabolism of fructose. Am J Clin Nutr. 1993;58:754S-65S.
Gaby AR. Adverse effects of dietary fructose. Altern Med Rev. 2005;10:294–306.
Katzen HM, Schimke RT. Multiple forms of hexokinase in the rat: Tissue distribution, age dependency, and properties. Proc Natl Acad Sci USA. 1965;54:1218–25.
Froesch ER, Ginsberg JL. Fructose metabolism of adipose tissue. I. Comparison of fructose and glucose metabolism in epidiymal adipose tissues of normal rats. J Biol Chem. 1962;237:3317–23.
Beyer PL, Caviar EM, McCallum RW. Fructose intake at current levels in the United States may cause gastrointestinal distress in normal adults. J Am Diet Assoc. 2005;105: 1559–66.
Perheentupa J, Raivio K. Fructose-induced hyperuricaemia. Lancet. 1967;2:528–31.
Bergstrom J, Hultman E, Roch-Norlund AE. Lactic acid accumulation in connection with fructose infusion. Acta Med Scand. 1968;184:359–64.
Hers H. Misuses for Fructose. Nature. 1979;227:241.
Woods H, Alberti K. Dangers of intravenous fructose. Lancet. 1972;2:1354.
Sestoft L. Fructose-en advarsel. Ugeskr Laeger. 1972;134: 571.
Baker SS, Cochran WJ, Greer FR, Heyman MB, Jacobson MS, Jaksic T, et al. The use and misuse of fruit juice in pediatrics. Pediatrics. 2001;107:1210–13.
Gerrits PM, Tsalikian E. Diabetes and fructose metabolism. Am J Clin Nutr. 1993;58:796S-9S.
Silverman M. Structure and function of hexose transporters. Annu Rev Biochem. 1991;60:757–94.
Gould GW, Thomas HM, Jess TJ, Bell GI. Expression of human glucose transporters in Xenopus oocytes: Kinetic characterization and substrate specificities of the erythrocyte, liver, and brain isoforms. Biochemistry. 1991;30: 5139–45.
Miyamoto K, Tatsumi S, Morimoto A, Minami H, Yamamoto H, Sone K, et al. Characterization of the rabbit intestinal fructose transporter (GLUT5). Biochem J. 1994;303:877–83.
Burant CF, Takeda J, Brot-Laroche E, Bell GI, Davidson NO. Fructose transporter in human spermatozoa and small intestine is GLUT5. J Biol Chem. 1992;267: 14523–6.
Kayano T, Burant CF, Fukumoto H, Gould GW, Fan YS, Eddy RL, et al. Human facilitative glucose transporters. Isolation, functional characterization, and gene localization of cDNAs encoding an isoform (GLUT5) expressed in small intestine, kidney, muscle, and adipose tissue and an unusual glucose transporter pseudogene-like sequence (GLUT6). J Biol Chem. 1990;265:13276–82.
Wood IS, Trayhurn P. Glucose transporters (GLUT and SGLT): Expanded families of sugar transport proteins. Br J Nutr. 2003;89:3–9.
Wood IS, Hunter L, Trayhurn P. Expression of Class III facilitative glucose transporter genes (GLUT-10 and GLUT-12) in mouse and human adipose tissues. Biochem Biophys Res Commun. 2003;308:43–9.
Joost HG, Thorens B. The extended GLUT-family of sugar/ polyol transport facilitators: Nomenclature, sequence characteristics, and potential function of its novel members (review). Mol Membr Biol. 2001;18:247–56.
Phay JE, Hussain HB, Moley JF. Cloning and expression analysis of a novel member of the facilitative glucose transporter family, SLC2A9 (GLUT9). Genomics. 2000;66: 217–20.
Johnson JH, Newgard CB, Milburn JL, Lodish HF, Thorens B. The high Km glucose transporter of islets of Langerhans is functionally similar to the low affinity transporter of liver and has an identical primary sequence. J Biol Chem. 1990;265:6548–51.
Hers H-G. Le métabolisme du fructose. Brussels: Editions Arsica, 1957.
Hers H, Kusaka T. Le Métabolisme du fructose 1-phosphate dans le foie. Biochim Biophys Acta. 1953;11: 427–32.
Jenkins BT, Hajra AK. Glycerol kinase and dihydroxyacetone kinase in rat brain. J Neurochem. 1976;26:377–85.
Grivell AR, Halls HJ, Berry MN. Role of mitochondria in hepatic fructose metabolism. Biochim Biophys Acta. 1991;1059:45–54.
Duran M, Beemer FA, Bruinvis L, Ketting D, Wadman SK. D-glyceric acidemia: An inborn error associated with fructose metabolism. Pediat Res. 1987;21:502–06.
Purich DL, Fromm HJ, Rudolph FB. The hexokinases: kinetic, physical, and regulatory properties. Adv Enzymol Relat Areas Mol Biol. 1973;39:249–326.
Cardenas ML, Rabajille E, Niemeyer H. Fructose is a good substrate for rat liver ‘glucokinase’ (hexokinase D). Biochem J. 1984;222:363–70.
Lebherz HG, Rutter WJ. Distribution of fructose diphosphate aldolase variants in biological systems. Biochemistry. 1969;8:109–21.
Penhoet EE, Kochman M, Rutter WJ. Isolation of fructose diphosphate aldolases A, B, and C. Biochemistry. 1969;8: 4391–95.
Malay AD, Procious SL, Tolan DR. The temperature dependence of activity and structure for the most prevalent mutant aldolase B associated with hereditary fructose intolerance. Arch Biochem Biophys. 2002;408:295–304.
Morris AJ, Tolan DR. Site-directed mutagenesis identifies aspartate 33 as a previously unidentified critical residue in the catalytic mechanism of rabbit aldolase A. J Biol Chem. 1993;268:1095–100.
Arakaki TL, Pezza JA, Cronin MA, Hopkins CE, Zimmer DB, Tolan DR, et al. Structure of human brain fructose 1,6-bisphosphate aldolase: Linking isozyme structure with function. Protein Sci. 2004;13:3077–84.
Penhoet EE, Kochman M, Rutter WJ. Molecular and catalytic properties of aldolase C. Biochemistry. 1969;8: 4396–402.
Owen OE, Morgan AP, Kemp HG, Sullivan JM, Herrera MG, Cahill GF, Jr. Brain metabolism during fasting. J Clin Invest. 1967;46:1589–95.
Hawkins RA, Williamson DH, Krebs HA. Ketone-body utilization by adult and suckling rat brain in vivo. Biochem J. 1971;122:13–18.
Magistretti P. In: Squire LR, Bloom FE, McConnell SK, Roberts JL, Spitzer NC, editors. Brain energy metabolism. Fundamental neuroscience. 2nd ed, New York: Academic Press, 2003. pp 339–360.
Weber M, deOliveira K, Valle S, Schweigert I, Rotta L, Fagundes I, et al. Study of developmental changes on hexoses metabolism in rat cerebral cortex. Neurochem Res. 2001;26:161–6.
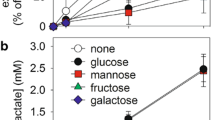
Wada H, Okada Y, Uzuo T, Nakamura H. The effects of glucose, mannose, fructose and lactate on the preservation of neural activity in the hippocampal slices from the guinea pig. Brain Res. 1998;788:144–50.
Saitoh M, Okada Y, Nabetani M. Effect of mannose, fructose and lactate on the preservation of synaptic potentials in hippocampal slices. Neurosci Lett. 1994;171: 125–8.
Allen L, Anderson S, Wender R, Meakin P, Ransom BR, Ray DE, et al. Fructose supports energy metabolism of some, but not all, axons in adult mouse optic nerve. J Neurophysiol. 2006;95:1917–25.
Pellerin L, Magistretti P. Neuroenergetics: Calling upon astrocytes to satisfy hungry neurons. Neuroscientist. 2004;l0:53–62.
Wiesinger H, Thiess U, Hamprecht B. Sorbitol pathway activity and utilization of polyols in astroglia-rich primary cultures. Glia. 1990;3:277–82.
Bergbauer K, Dringen R, Verleysdonk S, Gebhardt R, Hamprecht B, Wiesinger H. Studies on fructose metabolism in cultured astroglial cells and control hepatocytes: lack of fructokinase activity and immunoreactivity in astrocytes. Dev Neurosci. 1996;18:371–9.
Allen NJ, Karadottir R, Attwell D. A preferential role for glycolysis in preventing the anoxic depolarization of rat hippocampal area CA1 pyramidal cells. J Neurosci. 2005;25: 848–59.
Chain EB, Rose SP, Masi I, Pocchiari F. Metabolism of hexoses in rat cerebral cortex slices. J Neurochem. 1969;16: 93–100.
Bais R, James HM, Rofe AM, Conyers RA. The purification and properties of human liver ketohexokinase. A role for ketohexokinase and fructose-bisphosphate aldolase in the metabolic production of oxalate from xylitol. Biochem J. 1985;230:53–60.
Haradahira T, Tanaka A, Maeda M, Kanazawa Y, Ichiya YI, Masuda K. Radiosynthesis, rodent biodistribution, and metabolism of l-deoxy-l-[18F]fluoro-D-fructose. Nucl Med Biol. 1995;22:719–25.
Watanabe M, Shimono R, Kihara T. The distribution of [U-14] Fructose in the mice studied by whole-body autoradiography. Acta Histochem Cytochem. 1981;14:153–62.
Mantych GJ, James DE, Devaskar SU. Jejunal/kidney glucose transporter isoform (Glut-5) is expressed in the human blood-brain barrier. Endocrinol. 1993;132:35–40.
Weiser MM, Quill H. Estimation of fructokinase (ketohexokinase) in crude tissue preparations. Methods Enzymol. 1975;41:61–3.
Adelman RC, Ballard FJ, Weinhouse S. Purification and properties of rat liver fructokinase. J Biol Chem. 1967;242: 3360–65.
Hayward BE, Bonthron DT. Structure and alternative splicing of the ketohexokinase gene. Eur J Biochem. 1998;257:85–91.
Funari VA, Herrera VLM, Freeman D, Tolan DR. Genes required for fructose metabolism are expressed in purkinje cells in the cerebellum. Molec Brain Res. 2005;142:115–22.
McKenna MC. Introduction: Metabolic trafficking comes of age in the decade of the brain. Dev Neurosci. 1996;18: 333–5.
Pellerin L, Magistretti PJ. Excitatory amino acids stimulate aerobic glycolysis in astrocytes via an activation of the Na+/ K+ATPase. Dev Neurosci. 1996;18:336–42.
Kishi K, Tanaka T, Igawa M, Takase S, Goda T. Sucraseisomaltase and hexose transporter gene expressions are coordinately enhanced by dietary fructose in rat jejunum. J Nutr. 1999;129:953–6.
Gouyon F, Caillaud L, Carriere V, Klein C, Dalet V, Citadelle D, et al. Simple-sugar meals target GLUT2 at enterocyte apical membranes to improve sugar absorption: A study in GLUT2-null mice. J Physiol. 2003; 552:823–32.
Payne J, Maher F, Simpson I, Mattice L, Davies P. Glucose transporter Glut5 expression in microglial cells. Glia. 1997;21:327–31.
Nualart F, Godoy A, Reinicke K. Expression of the hexose transporters GLUT1 and GLUT2 during the early development of the human brain. Brain Res. 1999;824:97–104.
Hundal HS, Darakhshan F, Kristiansen S, Blakemore SJ, Richter EA. GLUT5 expression and fructose transport in human skeletal muscle. Adv Exp Med Biol. 1998;441: 35–45.
Lal S, Szwergold BS, Kappler F, Brown T. Detection of fructose-3-phosphokinase activity in intact mammalian lenses by 31P NMR spectroscopy. J Biol Chem. 1993;268: 7763–7.
Cui XL, Jiang L, Ferraris RP. Regulation of rat intestinal GLUT2 mRNA abundance by luminal and systemic factors. Biochim Biophys Acta. 2003;1612:178–85.
Mesonero J, Matosin M, Cambier D, Rodriguez-Yoldi MJ, Brot-Laroche E. Sugar-dependent expression of the fructose transporter GLUT5 in Caco-2 cells. Biochem J. 1995;312 (Pt. 3):757–62.
Maher F, Vannucci SJ, Simpson IA. Glucose transport proteins in brain. FASEB J. 1994;8:1003–11.
Pellerin L, Magistretti PJ. How to balance the brain energy budget while spending glucose differently. J Physiol. 2003;546:325.
Boado RJ, Black KL, Pardridge WM. Gene expression of GLUT3 and GLUT1 glucose transporters in human brain tumors. Brain Res Mol Brain Res. 1994;27:51–57.
Vannucci SJ, Maher F, Simpson IA. Glucose transporter proteins in brain: delivery of glucose to neurons and glia. Glia. 1997;21:2–21.
Maher F. Immunolocalization of GLUT1 and GLUT3 glucose transporters in primary cultured neurons and glia. J Neurosci Res. 1995;42:459–469.
Horikoshi Y, Sasaki A, Taguchi N, Maeda M, Tsukagoshi H, Sato K, et al. Human GLUT5 immunolabeling is useful for evaluating microglial status in neuropathological study using paraffin sections. Acta Neuropathol (Berl). 2003; 105:157–62.
Choeiri C, Staines W, Messier C. Immunohistochemical localization and quantification of glucose transporters in the mouse brain. Neurosci. 2002; 111:19–34.
Klein JR, Hurwitz R, Olsen NS. Distribution of intravenously injected fructose and glucose between blood and brain. J Biol Chem. 1946;164:509–12.
Thurston JH, Levy CA, Warren SK, Jones EM. Permeability of the blood-brain barrier to fructose and the anaerobic use of fructose in the brains of young mice. J Neurochem. 1972;19:1685–96.
Maher F, Vannucci SJ, Simpson IA. Glucose transporter isoforms in brain: Absence of GLUT3 from the blood-brain barrier. J Cereb Blood Flow Metab. 1993;13:342–5.
Arluison M, Quignon M, Nguyen P, Thorens B, Leloup C, Penicaud L. Distribution and anatomical localization of the glucose transporter 2 (GLUT2) in the adult rat brain — an immunohistochemical study. J Chem Neuroanat. 2004;28: 117–36.
Takata K. Glucose transporters in the transepithelial transport of glucose. J Elec Microscopy (Tokyo). 1996;45: 275–84.
Oldendorf WH. Brain uptake of radiolabeled amino acids, amines, and hexoses after arterial injection. Am J Physiol. 1971;221:1629–39.
Popovici T, Berwald-Netter Y, Vibert M, Kahn A, Skala H. Localization of aldolase C mRNA in brain cells. FEBS Lett. 1990;268:189–93.
Walther EU, Dichgans M, Maricich SM, Romito RR, Yang F, Dziennis S, et al. Genomic sequences of aldolase C (Zebrin II) direct lacZ expression exclusively in nonneuronal cells of transgenic mice. Proc Natl Acad Sci USA. 1998;95:2615–20.
Thompson RJ, Kynoch PA, Willson VJ. Cellular localization of aldolase C subunits in human brain. Brain Res. 1982;232: 489–93.
LeFevre P, Marshall J. Conformational specificity in a biological sugar transport system. Am J Physiol. 1958;194: 333–7.
Joost HG, Bell GI, Best JD, Birnbaum MJ, Charron MJ, Chen YT, et al. Nomenclature of the GLUT/SLC2A family of sugar/polyol transport facilitators. Am J Physiol Endocrinol Metab. 2002;282:E974–976.
Doege H, Schurmann A, Bahrenberg G, Brauers A, Joost HG. GLUT8, a novel member of the sugar transport facilitator family with glucose transport activity. J Biol Chem. 2000;275:16275–80.
Doege H, Bocianski A, Scheepers A, Axer H, Eckel J, Joost HG, et al. Characterization of human glucose transporter (GLUT) 11 (encoded by SLC2A11), a novel sugar-transport facilitator specifically expressed in heart and skeletal muscle. Biochem J. 2001;359:443–9.
Doege H, Bocianski A, Joost HG, Schurmann A. Activity and genomic organization of human glucose transporter 9 (GLUT9), a novel member of the family of sugar-transport facilitators predominantly expressed in brain and leucocytes. Biochem J. 2000;350(Pt 3):771–6.
Augustin R, Carayannopoulos MO, Dowd LO, Phay JE, Moley JF, Moley KH. Identification and characterization of human glucose transporter-like protein-9 (GLUT9): Alternative splicing alters trafficking. J Biol Chem. 2004;279:16229–36.
Li Q, Manolescu A, Ritzel M, Yao S, Slugoski M, Young JD, et al. Cloning and functional characterization of the human GLUT7 isoform SLC2A7 from the small intestine. Am J Physiol Gastrointest Liver Physiol. 2004;287:G236–42.
Wu X, Li W, Sharma V, Godzik A, Freeze HH. Cloning and characterization of glucose transporter 11, a novel sugar transporter that is alternatively spliced in various tissues. Mol Genet Metab. 2002;76:37–45.
Manolescu A, Salas-Burgos AM, Fischbarg J, Cheeseman CI. Identification of a hydrophobic residue as a key determinant of fructose transport by the facilitative hexose transporter SLC2A7 (GLUT7). J Biol Chem. 2005;280:42978–83.
Lisinski I, Schurmann A, Joost HG, Cushman SW, AlHasani H. Targeting of GLUT6 (formerly GLUT9) and GLUT8 in rat adipose cells. Biochem J. 2001;358:517–22.
Raushel FM, Cleland WW. Bovine liver fructokinase: Purification and kinetic properties. Biochemistry. 1977;16: 2169–75.
Adelman RC, Morris HP, Weinhouse S. Fructokinase, triokinase, and aldolases in liver tumors of the rat. Cancer Res. 1967;27:2408–13.
Su AI, Wiltshire T, Batalov S, Lapp H, Ching KA, Block D, et al. A gene atlas of the mouse and human protein-encoding transcriptomes. Proc Natl Acad Sci USA. 2004;101: 6062–7.
Funari VA.Novel Computational and Classical Molecular Biological Approaches to Discovering Alternative Sites of Fructose Metabolism in Mice and Man, Boston University: PhD Dissertation, 2001.
Baron CB, Ozaki S, Watanabe Y, Hirata M, LaBelle EF, Coburn RF. Inositol 1,4,5-trisphosphate binding to porcine tracheal smooth muscle aldolase. J Biol Chem. 1995;270: 20459–65.
Sarna JR, Larouche M, Marzban H, Sillitoe RV, Rancourt DE, Hawkes R. Patterned Purkinje cell degeneration in mouse models of Niemann-Pick type C disease. J Comp Neurol. 2003;456:279–91.
Hawkes R, Herrup K. Aldolase C/zebrin II and the regionalization of the cerebellum. J Mol Neurosci. 1995;6:147–58.
Ahn AH, Dziennis S, Hawkes R, Herrup K. The cloning of zebrin II reveals its identity with aldolase C. Development. 1994;120:2081–90.
Voogd J, Ruigrok TJ. Transverse and longitudinal patterns in the mammalian cerebellum. Prog Brain Res. 1997;114: 21–37.
Brochu G, Maler L, Hawkes R. Zebrin II: A polypeptide antigen expressed selectively by Purkinje cells reveals compartments in rat and fish cerebellum. J Comp Neurol. 1990;291:538–52.
Staugaitis SM, Zerlin M, Hawkes R, Levine JM, Goldman JE. Aldolase C/zebrin II expression in the neonatal rat forebrain reveals cellular heterogeneity within the subventricular zone and early astrocyte differentiation. J Neurosci. 2001;21:6195–205.
Beutler E, Guinto E. Dihydroxyacetone metabolism by human erythrocytes: Demonstration of triokinase activity and its characterization. Blood. 1973;41:559–68.
Miwa I, Kito Y, Okuda J. Purification and characterization of triokinase from porcine kidney. Prep Biochem. 1994;24: 203–23.
Akhtar N, Blomberg A, Adler L. Osmoregulation and protein expression in a pbs2delta mutant of Saccharomyces cerevisiae during adaptation to hypersaline stress. FEBS Lett. 1997;403:173–80.
Blomberg A. Osmoresponsive proteins and functional assessment strategies in Saccharomyces cerevisiae. Electrophor. 1997;18:1429–40.
Molin M, Norbeck J, Blomberg A. Dihydroxyacetone kinases in Saccharomyces cerevisiae are involved in detoxification of dihydroxyacetone. J Biol Chem. 2003;278: 1415–23.
Hagopian K, Ramsey JJ, Weindruch R. Fructose metabolizing enzymes from mouse liver: influence of age and caloric restriction. Biochim Biophys Acta. 2005;1721: 37–43.
Welsh JP, Yuen G, Placantonakis DG, Vu TQ, Haiss F, O’Hearn E, et al. Why do Purkinje cells die so easily after global brain ischemia? Aldolase C, EAAT4, and the cerebellar contribution to posthypoxic myoclonus. Adv Neurol. 2002;89:331–59.
Jordan J, Simandle S, Tulbert C, Busija D, Miller A. Fructose-fed rats are protected against ischemia/reperfusion injury. J Pharmacol Exp Ther. 2003;307:1007–11.
Joyeux-Faure M, Rossini E, Ribuot C, Faure P. Fructosefed rat hearts are protected against ischemia-reperfusion injury. Exp Biol Med (Maywood). 2006;231:456–62.
Rosenthal M, Sick TJ. Glycolytic and oxidative metabolic contributions to potassium ion transport in rat cerebral cortex. Can J Physiol Pharmacol. 1992;70(Suppl):S165–9.
Kelleher JA, Chan PH, Chan TY, Gregory GA. Energy metabolism in hypoxic astrocytes: Protective mechanism of fructose-l,6-bisphosphate. Neurochem Res. 1995;20: 785–92.
Sola A, Berrios M, Sheldon RA, Ferriero DM, Gregory GA. Fructose-1,6-bisphosphate after hypoxic ischemic injury is protective to the neonatal rat brain. Brain Res. 1996;741: 294–9.
Lazzarino G, Tavazzi B, Di Pierro D, Giardina B. Ischemia and reperfusion: Effect of fructose-l,6-bisphosphate. Free Radic Res Commun. 1992;16:325–39.
Giordano FJ. Oxygen, oxidative stress, hypoxia, and heart failure. J Clin Invest. 2005;l15:500–08.
Kehrer JP. Concepts related to the study of reactive oxygen and cardiac reperfusion injury. Free Radic Res Commun. 1989;5:305–14.
Woods HF, Eggleston LV, Krebs HA. The cause of hepatic accumulation of fructose 1-phosphate on fructose loading. Biochem J. 1970;l19:501–10.
Farah V, Elased KM, Chen Y, Key MP, Cunha TS, Irigoyen MC, et al. Nocturnal hypertension in mice consuming a high fructose diet. Auton Neurosci. 2006;130:41–50.
Shu HJ, Isenberg K, Cormier RJ, Benz A, Zorumski CF. Expression of fructose sensitive glucose transporter in the brains of fructose-fed rats. Neurosci. 2006;140: 889–95.
Doyle SA, Tolan DR. Characterization of recombinant human aldolase B and purification by metal chelate chromatography. Biochem Biophys Res Commun. 1995;206:902–08.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Funari, V.A., Crandall, J.E. & Tolan, D.R. Fructose metabolism in the cerebellum. Cerebellum 6, 130–140 (2007). https://doi.org/10.1080/14734220601064759
Issue Date:
DOI: https://doi.org/10.1080/14734220601064759