Abstract
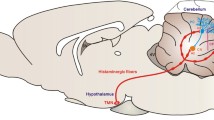
Morphological studies have described reciprocal cerebello-hypothalamic projections in various species. These connections provide evidence for the key role of the cerebellum and hypothalamus in physiological regulatory processes such as autonomic and endocrine homeostasis. Our recent study using horseradish peroxidase (HRP) retrograde axonal transport technique showed cerebellar connections with the posterior and the dorsomedial hypothalamic nuclei. Further, we have demonstrated regional differences of the connections of the dorsomedial hypothalamic nucleus in rat. The results of HRP labelling showed that afferent pathways originating from the anterior and posterior parts of dorsomedial hypothalamic nucleus indicate a number of differences in the projections. The posterior part of the dorsomedial hypothalamic nucleus and the posterior hypothalamic nucleus receives direct distinct projections from the cerebellum, whereas the anterior part of the dorsomedial hypothalamic nucleus does not. Moreover, the posterior part of the dorsomedial nucleus of the hypothalamus when compared to the posterior hypothalamic nucleus has more intense connections with the cerebellum. These observations bring a new perspective on the question of how the cerebellum is involved in the regulation visceromotor functions.
Similar content being viewed by others
References
Albe-Fessard D, Berkley KJ, Kruger L, Ralston HJ, Willis WD. Diencephalic mechanisms of pain sensation. Brain Res Rev 1985; 9: 217–296.
Gilbert PFC. A theory of memory that explains the function and structure of the cerebellum. Brain Res 1974; 70: 1–18.
Gonzalo-Ruiz A, Leichnetz GR. Connections of the caudal cerebellar interpositus complex in a new world monkey (Cebus apella). Brain Res Bull 1990; 25: 919–917.
Middleton FA, Strick PL. Basal ganglia and cerebellar loops: motor and cognitive circuits. Brain Res Rev 2000; 31: 236–250.
Nixon PD, Passingham RE. The cerebellum and cognition: cerebellar lesions do not impair spatial working memory or visual associative learning in monkeys. Eur J Neurosci 1999; 11: 4070–4080.
Schmahmann JD, Pandya DN. The cerebro-cerebellar system. Int Rev Neurobiol 1997; 41: 31–60.
Teune TM, Van Der Burg J, Van Der Moer J, Voogd J, Ruigrok TJ. Topography of cerebellar nuclear projections to the brain stem in the rat. Prog Brain Res 2000; 124: 141–172.
Holderfer RN, Miller LE, Chen LJ, Houk JC. Functional connectivity between cerebellum and primary motor cortex in the awake monkey. J Neurophysiol 2000; 84: 585–590.
Tamada T, Miyauchi S, Imamizu H, Yoshioka T, Kawato M. Cerebro-cerebellar functional connectivity revealed by the laterality index in tool-use learning. Neuro Report 1999; 10: 325–331.
Leichnetz GR, Smith DJ, Spencer RF. Cortical projections of the paramedian tegmental and basilar pons in the monkey. J Comp Neurol 1984; 228: 388–408.
SchMahmann JD, Pandya DN. Prelunate occipitotemporal and parahipocampal projections to the basis pontis in the rhesus monkey. J Comp Neurol 1993; 337: 94–112.
SchMahmann JD, Pandya DN. Prefrontal cortex projections to the basilar pons in the rhesus monkey. Implications for the cerebellar contributions to higher functions. Neurosci Lett 1995; 199: 175–178.
Asanuma C, Thach WT, Jones EG. Distribution of cerebellar terminations in the ventral lateral thalamic region of the monkey. Brain Res Rev 1983; 5: 237–265.
Yamamoto T, Yoshida K, Kishimoto Y, Oka H. The medial dorsal nucleus is one of the thalamic relays of the cerebellocerebral response to the frontal association cortex in the monkey: horseradish peroxidase and fluorescent dye double staining study. Brain Res 1992; 579: 315–320.
Middleton FA, Strick PL. Anatomical evidence for cerebellar and basal ganglia involvement in higher cognitive functions. Science 1994; 266: 458–461.
Person RJ, Andrezik JA, Dormer KJ, Foreman RD. Fastigial nucleus projections in the midbrain and thalamus. Neurosci 1986; 18: 105–120.
Liu Fa-yi, Qiao JT, Dafny N. Cerebellar stimulation modulates thalamic noxious-evoked reponses. Brain Res Bull 1993; 30: 529–534.
Lynch JC, Hoover JE, Strick PL. Input to the primate frontal eye field from the substantia nigra, superior coliculus, and dentate nucleus demonstrated by transneuronal transport. Exp Brain Res 1994; 100: 181–186.
Rouiller EM, Liang F, Babalian A, Moret V, Wiesendanger M. Cerebellothalamocortical and pallidothalamocortical projections to the primary and supplementary motor cortical areas: a multiple tracing study in the macaque monkeys. J Comp Neurol 1994; 345: 185–213.
Hendry SH, Jones EG, Graham J. Thalamic relay nuclei for cerebellar and certain related fiber systems in the cat. J Comp Neurol 1979; 185: 679–713.
Stepniewska L, Preuss TM, Kass JH. Thalamic connections of the primary motor cortex (MI) of owl monkeys. J Comp Neurol 1994; 349: 558–582.
Hua SE, Houk JC. Cerebellar guidance of premotor network development and sensorimotor learning. Learning Memory 1997; 4: 63–76.
Çavdar S, Onat F, Yananh HR, Şehirli Ü, Tulay C, Saka E, Gürdal E. Cerebellar connections to the rostral reticular nucleus of the thalamus in the rat. J Anat 2002; 201: 485–491.
Crick F. Function of the thalamic reticular complex: the searchlight hypothesis. Proceedings of the National Academy of Sciences USA 1984; 81: 4586–4590.
Mitrofanis J, DeFonseka R. Organization of connections between the zona inserta and the interposed nucleus. Anat Embryol (Berl) 2001; 204: 153–159.
Newman PP, Rezaa H. Functional relationship between the hippocampus and the cerebellum: an electrophysiological study of the cat. J Physiol 1979; 287: 405–426.
Ruigrok TJ, Voogd J. Cerebellar nucleo-olivary projections in the rat: an anterograde tracing study with phaseolus vulgaris-leucoagglutinin (PHA-L). J Comp Neurol 1990; 298: 315–333.
Kawamura S, Hattori S, Higo S, Matsuyama T. The cerebellar projections to the superior colliculus and pretectum in the cat: an autoradiographic and horseradish peroxidase study. Neurosci 1982; 7: 1673–1689.
Wallenberg A. Sekundäre Bahnen aus dem frontalen sensiblen Trigeminuskerne des Kaninchens. Anat Anz 1905; 26: 145–155.
Whiteside JA, Snider RS. Relation to cerebelllar to upper brain stem. J Neurophysiol 1953; 16: 397–413.
Jacobs VL The cerebellofugal system in the tarsius (Tarsiidae carbonarius) and the marmoset (Oedipomidas oedipus). Doctoral dissertation University of Kansas, Lawrence, Kansas, 1965.
Martin GF, King JS, Dom R. The projections of the deep cerebellar nuclei of the opossum, Didelphis marsupialis virginiana. J Hirnforsch 1974; 15: 545–573.
Zheng ZH, Dietrichs E, Walberg F. Cerebellar afferent fibres from the dorsal vagal nucleus in the cat. Neurosci Lett 1982; 32: 113–118.
Chida K, Iadecola C, Underwood MD, Reis DJ. A novel vasodepressor response elicited from the rat cerebellar fastigial nucleus: the fastigial depressor response. Brain Res 1986; 370: 378–382.
Bernardis LL, Bellinger LL. The dorsomedial hypothalamic nucleus revisited: 1998 update. Proc Soc Exp Biol Med 1998; 218: 284–306.
Dalton LD, Carpenter RG, Grossman SP. Ingestive behavior in adult rat with dorsomedial hypothalamic lesion. Physiol Behav 1981; 26: 117–123.
Gören Z, Asian N, Berkman K, Oktay S, Onat F. The role of amygdala and hypothalamus in GABAa antagonist bicucullineinduced cardiovascular responses in conscious rats. Brain Res 1996; 722: 118–124.
Inglefield JR, Schwarzkopf SB, Kellog CK. Alterations in behavioral responses to Stressors following excitotoxin lesion of dorsomedial hypothalamic region. Brain Res 1994; 633: 151–161.
Dietrichs E. Cerebellar autonomic function: direct hypothalamocerebellar pathway. Science 1984; 223: 591–593.
Dietrichs E, Zheng H. Are hypothalamo-cerebellar fibers collaterals from the hypothalamo-spinal projections? Brain Res 1984; 296: 225–231.
Dietrichs E, Haines DE, Qvist H. Indirect hypothalamo-cerebellar pathway? Demonstration of hypothalamic efferents to the lateral reticular nucleus. Exp Brain Res 1985; 60: 483–491.
Dietrichs E, Walberg F, Haines DE. Cerebellar nuclear afferents from feline hypothalamus demonstrated by retrograde transport after implantation of crystallin wheat germ agglutinin-horseradish peroxidase complex. Neurosci Lett 1985; 54: 129–133.
Haines DE, Dietrichs E. Hypothalamo-cerebellar projections in the squirrel monkey (Saimiri sciureus): an HRP study. Soc Neurosci Abstr 1983; 9: 870.
Ter Horst GJ, Luiten PGM. The projections of the dorsomedial hypothalamic nucleus in the rat. Brain Res Bull 1986; 16: 231–248.
Dietrichs E, Haines DE. Demonstration of hypothalamo-cerebellar and cerebello-hypothalamic fibers in a prosimian primate (Galago crassicaudatus) Anat Embryol 1984; 170: 313–318.
Wang T, Yu Qx, Wang JJ. Effects of stimulating lateral hypothalamic area and ventromedial nucleus of hypothalamus on cerebellar cortical neuronal activity in the cat. Chin J Physiol 1994; 10: 17–25.
Dietrichs E, Roste GK, Roste LS, Qvist HL, Haines DE. The hypothalamocerebellar projection in the cat: branching and nuclear termination. Arch Ital Biol 1990; 132: 25–38.
Haines DE, May PJ, Dietrichs E. Neuronal connections between the cerebellar nuclei and hypothalamus in Macaca fascicularis: cerebello-visceral circuit. J Comp Neurol 1990; 299: 106–122.
Haines DE, Sowa TE, Dietrichs E. Connections between the cerebellum and hypothalamus in the tree shrew (Tupaia glis). Brain Res 1985; 328: 367–373.
Gören Z, Akici A, Berkman K, Onat F. Cardiovascular responses to NMDA injected into nuclei of hypothalamus or amygdala in conscious rats. Pharmacology 2000; 61: 257–264.
Dampney RAL. Functional organization of central pathways regulating the cardiovascular system. Physiol Rev 1994; 74: 323–364.
De Novellis V, Stotz-Potter EH, Morin SM, Rossi F, Di Micco JA. Hypothalamic sites mediating cardiovascular effects of microinjected bicuculline and EAAs in rats. Am J Physiol 1995; 269: 131–140.
Yoshimatus H, Niijima A, Oomura Y, Yamabe K, Katafuchi T. Effects of hypothalamic lesions on pancreatic autonomic nerve activity in the rat. Brain Res 1984; 303: 147–152.
Eferakeya JE, Bunag RD. Adrenomedullary pressor responses during posterior hypothalamic stimulation. Am J Physiol 1974; 227: 114.
Haines DE, Dietrichs E, Mihailoff GA, McDonald EF. The cerebellar-hypothalamic axis: Basic circuits and clinical observations. Int Rev Neurobiol 1997; 41P: 83–107.
Çavdar S, Onat F, Aker R, Şehirli Ü, Şan T, Yananli HR. The afferent connections of the posterior hypothalamic nucleus in the rat using horseradish peroxidase. J Anat 2001; 198: 463–472.
Çavdar S, Şan T, Aker R, Şehirli Ü, Onat F. Cerebellar connections to the dorsomedial and posterior nuclei of the hypothalamus in the rat. J Anat 2001; 198: 37–45.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Onat, F., Çavdar, S. Cerebellar connections: hypothalamus. Cerebellum 2, 263–269 (2003). https://doi.org/10.1080/14734220310016187
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1080/14734220310016187