Summary
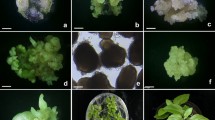
In vitro regeneration of plants via somatic embryogenesis through cell suspension culture was achieved in horsegram. Embryogenic calluses were induced on leaf segments on solid Murashige and Skoog (MS) medium with 9.0 μM 2,4-dichlorophenoxyacetic acid (2,4-D). Differentiation of somatic embryos occurred when the embryogenic calluses were transferred to liquid MS medium containing 2,4-D. Maximum frequency (33.2%) of somatic embryos was observed on MS medium supplemented with 7.9 μM 2,4-D. Cotyledonary-torpedo-shaped embryos were transferred to liquid MS medium without growth regulators for maturation and germination. About 5% of the embryos germinated into plants, which grew further on solid MS medium. The plants were hardened and established in soil. Effects of various auxins, cytokinins, carbohydrates, amino acids, and other additives on induction and germination of somatic embryos were also studied. A medium supplemented with 7.9 μM 2,4-D, 3.0% sucrose, 40 mg l−1L-glutamine, and 1.0 μM abscisic acid was effective to achieve a high frequency of somatic embryo induction, maturation, and further development.
Similar content being viewed by others
References
Ammirato, P. V. The regulation of somatic embryo development in plant cell cultures: suspension culture techniques and hormone requirements. Biotechnology 1:68–74; 1983.
Anbazhagan, V. R.; Ganapathi, A. Somatic embryogenesis in cell suspensions of pigeonpea (Cajanus cajan L.). Plant Cell Tiss. Organ Cult. 56:179–184; 1999.
Christou, P. Biotechnology applied to grain legumes. Field Crops Res. 53:83–97; 1997.
Eapen, S.; George, L. Ortogeny of somatic embryos of Vigna aconitifolia, Vigna mungo and Vigna radiata. Ann. Bot. 66:219–226; 1990.
Eapen, S.; George, L. Somatic embryogenesis in peanut: influence of growth regulators and sugars. Plant. Cell Tiss. Organ Cult., 35:151–156; 1993.
Finer, J. J.; Kriebal, H. W.; Becwar, M. R. Initiation of embryogenic callus and suspension cultures of eastern white pine (Pinus strobus L.). Plant Cell Rep. 8:203–206; 1989.
Finer, J. J.; McMullen, D. Transformation of soybean via particle bombardment of embryogenic suspension culture tissue. In Vitro Cell. Dev. Biol. Plant 27:175–182; 1991.
Finer, J. J.; Nagasawa, A. Development of an embryogenic suspension culture of soybean [Glycine max (L.) Merr.]. Plant Cell Rep. 7:238–241; 1988.
Gamborg, O. L.; Miller, R. A.; Ojima, K. Nutrient experiments of suspension culture of soybean root callus. Exp. Cell Res. 80:150–158; 1968.
Girija, S; Ganapathi, A; Ananthakrishnan, G. Somatic embryogenesis in Vigna radiata L. Wilezek. Ind. J. Exp. Biol. 38:1241–1244; 2000.
Gomez, K. A.; Gomez, K. A. Statistical procedures for agricultural research with emphasis on rice. Los Baños, Philippines: International Rice Research Institute; 1976.
Hall, R. D. The initiation and maintenance of plant suspension cultures. Plant Tiss. Cult. manual A3:1–21; 1991.
Hartweck, L. M.; Lazzeri, P. A.; Cui, D.; Collins, G. B.; Williams, E. G. Auxin-orientation effects on somatic embryogenesis from immature soybean cotyledons. In Vitro Cell. Dev. Biol. 24P:821–824; 1988.
Jeswani, L. M.; Baldev, B. Advances in pulse production technology publication and information division. New Delhi: Indian Council of Agricultural Research; 1990.
Khlifi, S.; Tremblay, F. M. Maturation of black spruce somatic embryos. Part I: effect of l-glutamine on the number and geminability of somatic embryos. Plant Cell tiss. Organ Cult. 41:23–32; 1995.
Kulothungan, S.; Ganapathi, A.; Shajahan, A.; Kathiravan, K. Somatic embryogenesis in cell suspension culture of cowpea [Vigna unguiculata (L.) Walp.]. Israel. J. Plant Sci. 43:385–390; 1995.
Kumar, A. S.; Gamborg, O. L.; Nabors, M. W. Regeneration from long-term cell suspension cultures of tepry bean (Phaseolus acutifolius). Plant Cell Rep. 7:322–325; 1988.
Lakshmanan, P.; Taji, A. Somatic embryogenesis in leguminous plants. Plant Biol. 2:136–148; 2000.
Linsmaier, F. M.; Skoog, F. Organic growth factor requirements of tobacco tissue cultures. Physiol. Plant. 8:100–127; 1965.
Loiseau, J.; Marche, C.; Le Deunff, Y. Effects of auxins, cytokinins, carbohydrates and amino acids on somatic embryogenesis induction from shoot apices of pea. Plant Cell Tiss. Organ Cult. 41:267–275; 1995.
Martins, I. S.; Sondahl, M. R. Early stages of somatic embryo differentiation from callus cells of bean (Phaseolus vulgaris L.) grwon in liquid medium. J. Plant Physiol. 117:97–108; 1984.
Merkle, S. A.; Parrott, W. A.; Flinn, B. S. Morphogenic aspects of somatic embryogenesis. In: Thorpe, T. A., ed. In vitro embryogenesis in plants. Dordrecht: Kluwer Academic Publishers; 1995:155–203.
Muniyappa, V.; Reddy, H. P.; Siva Shankar, G. Yellow mosaic diseases of Dolichos biflorus L. (Horsegram). Curr. Res. 4:176; 1975.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15:473–497; 1962.
Phillips, G. C.; Collins, G. B. Induction and development of somatic embryos from cell suspension cultures of soybean. Plant Cell Tiss. Organ Cult. 1:123–129; 1981.
Prem Anand, R.; Ganapathi, A.; Anbazhagan, V. R.; Vengadesen, G.; Selvaraj, N. High frequency plant regeneration via somatic embryogenesis in cell suspension cultures of cowpea [Vigna unguiculata (L.) Walp.. In Vitro Cell. Dev. Biol. Plant 36:475–480; 2000.
Ranch, J. P.; Oglesby, L.; Zielinski, A. C. Plant regeneration from embryoderived tissue cultures of soybean by somatic embryogenesis. In Vitro Cell. Dev. Biol. 21:653–657; 1985.
Rangasamy, N. S. Somatic embryogenesis in angiosperm cell, tissue and organ cultures. Proc. Ind. Acad. Sci. 95:247–271; 1986.
Samoylov, V. M.; Tucker, D. M.; Parrott, W. A. A liquid medium based protocol for rapid regeneration from embryogenic soybean cultures. Plant Cell Rep. 18:49–54; 1998a.
Samoylov, V. M.; Tucker, D. M.; Parrott, W. A. Soybean [Glycine max (L. Merr.)] embryogenic cultures: the role of sucrose and total nitrogen content on proliferation. In Vitro Cell. Dev. Biol. Plant 34:8–13; 1998b.
Saunders, J. W.; Hosfield, G. L.; Levi, A. Morphogenetic effects of 2,4-dichlorophenoxyacetic acid on pinto bean (Phaseolus vulgaris L.) leaf explants in vitro. Plant Cell Rep. 6:46–49; 1987.
Skokut, T. A.; Manchester, J.; Schaefer, J. Regeration of alfalfa tissue culture: stimulation of somatic embryo production by amino acids and N-15 NMR determination of nitrogen utilization. Plant Physiol. 79:579–583; 1985.
Sounder Raj, V.; Tejavathi, D. H.; Nijalingappa, B. H. M. Shoot-tip culture in Dolichos biflorus L. Curr. Sci. 58:1385–1388; 1989.
Sreekataradhya, R.; Jagadish, M.; Shivashankar, G. Early, day-neutral horsegram cultures with high yield potential. Curr. Res. 3:15–16; 1974.
Stuart, D. A.; Strickland, S. G. Somatic embryogenesis from cell cultures of Medicago sativa L. I: the role of amino acid additions to the regeneration medium. Plant Sci. Lett. 34:165–174; 1984.
Tetu, T.; Sangwan, R. S.; Sangwan-Noreel, B. S. Direct somatic embryogenesis and organogenesis in cultured immature zygotic embryos of Pisum sativum L. J. Plant Physiol. 137:102–109; 1990.
Varisai Mohamed, S.; Jawahar, M.; Jayabalan, N. Effect of AdS, BAP and IBA on plant regeneration from Macrotyloma uniflorum (Lam.) Verdc. Phytomorphology 48:61–65; 1998.
Varisai Mohamed, S.; Jawahar, M.; Thiruvengadam, M.; Jeyakumar, M.; Jayabalan, N. Effect of cytokinins on proliferation of multiple shoots in Horsegram [Macrotyloma uniflorum (Lam.) Verdc.]. J. Plant Biotechnol. 1(2): 79–83; 1999.
Williams, F. J.; Grewal, J. S.; Amin, K. S. Serious and new diseases of pulse crops in India in 1966. Plant Dis. Rep. 52:300–304; 1968.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Varisai Mohamed, S., Wang, C.S., Thiruvengadam, M. et al. In vitro plant regeneration via somatic embryogenesis through cell suspension cultures of horsegram [Macrotyloma uniflorum (Lam.) verdc.]. In Vitro Cell.Dev.Biol.-Plant 40, 284–289 (2004). https://doi.org/10.1079/IVP2003524
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1079/IVP2003524