Abstract
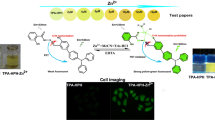
A terpyridine based compound L1 was designed and synthesized as an “off-on” chemosensor for the detection of Zn2+. Chemosensor L1 showed excellent selectivity and sensitivity toward Zn2+ by exhibiting a large fluorescence enhancement (~51-fold) at 370 nm whereas other competitive metal ions did not show any noticeable change in the emission spectra of chemosensor L1. The chemosensor (L1)was shown to detect Zn2+ ions down to 9.76 μM at pH 7.4. However, chemosensor L1 binds Zn2+ in a 1: 2 ratio (receptor: metal) with an association constant of 1.85 x 104 (R2 = 0.993) and this 1: 2 stoichiometric fashion is established on the basis of a Job plot and mass spectroscopy. DFT/TD-DFT calculations were carried out to understand the binding nature, coordination features and electronic properties of L1 and the L1-2Zn2+ complex. In addition, this “turn-on” fluorescence probe was effectively used to image intracellular Zn2+ ions in cultured MDA-MB-468 cells.
Similar content being viewed by others
References
R. P. Haugland, The handbook: a guide to fluorescent probes and labeling technologies, Molecular Probes, 2005
J. E. Kwon, S. Lee, Y. You, K. H. Baek, K. Ohkubo, J. Cho, S. Fukuzumi, I. Shin, S. Y. Park and W. Nam, Inorg. Chem., 2012, 51, 8760–8774
B. Valeur and I. Leray, Coord. Chem. Rev., 2000, 205, 3–40
P. G. Georgopoulos, A. Roy, M. J. Yonone-Lioy, R. E. Opiekun and P. J. Lioy, J. Toxicol. Environ. Health, Part B, 2001, 4, 341–394
R. Y. Tsien, Fluorescent and photochemical probes of dynamic biochemical signals inside living Cell, American Chemical Society, Washington, DC, USA, 1993, pp. 130–146.
S. K. Kim, D. H. Lee, J. Hong and J. Yoon, Acc. Chem. Res., 2009, 42, 23–31
J. Zhao, Y. Wang, W. Dong, Y. Wu, D. Li and Q. Zhang, Inorg. Chem., 2016, 55, 3265–3271.
B. L. Vallee and K. H. Falchuk, Physiol. Rev., 1993, 73, 79–118
S. C. Burdette and S. J. Lippard, Proc. Natl. Acad. Sci. U. S. A., 2003, 100, 3605–3610
Z. K. Wu, Y. F. Zhang, J. S. Ma and G. Yang, Inorg. Chem., 2006, 45, 3140–3142
A. S. Prasad, Nutrition, 1995, 11, 93–99.
J. M. Berg and Y. Shi, Science, 1996, 271, 1081–1085
W. N. Lipscomb and N. Sträter, Chem. Rev., 1996, 96, 2375–2433
T. Kambe, Y. Yamaguchi-Iwai, R. Sasaki and M. Nagao, Cell. Mol. Life Sci., 2004, 61, 49–68
P. Jiang and Z. Guo, Coord. Chem. Rev., 2004, 248, 205–229
S. Yamasaki, K. Sakata-Sogawa, A. Hasegawa, T. Suzuki, K. Kabu, E. Sato, T. Kurosaki, S. Yamashita, M. Tokunaga, K. Nishida and T. Hirano, J. Cell Biol., 2007, 177, 637–645.
J. J. R. F. da Silva and R. J. P. Williams, The biological chemistry of the elements: the inorganic chemistry of life, Oxford University Press, Oxford, 2001, pp. 315–335
B. L. Vallee and D. S. Auld, Acc. Chem. Res., 1993, 26, 543–551
A. I. Bush, W. H. Pettingell, G. Multhaup, M. D. Paradis, J. P. Vonsattel, J. F. Gusella, K. Beyreuther, C. L. Masters and R. E. Tanzi, Science, 1994, 265, 1464–1468.
C. J. Frederickson, S. W. Suh, D. Silva, C. J. Frederickson and R. B. Thompson, J. Nutr., 2000, 130, 1471S–1483S
D. W. Choi and J. Y. Koh, Annu. Rev. Neurosci., 1998, 21, 347–375
W. Maret, C. Jacob, B. L. Vallee and E. H. Fischer, Proc. Natl. Acad. Sci. U. S. A., 1999, 96, 1936–1940.
P. J. Fraker and L. E. King, Annu. Rev. Nutr., 2004, 24, 277–298
M. P. Cuajungco and G. J. Lees, Neurobiol. Dis., 1997, 4, 137–169
S. Frassinetti, G. L. Bronzetti, L. Caltavuturo, M. Cini and C. Della Croce, J. Environ. Pathol., Toxicol. Oncol., 2006, 25, 597–610.
X. Chen, T. Pradhan, F. Wang, J. S. Kim and J. Yoon, Chem. Rev., 2012, 112, 1910–1956
J. H. Weiss, S. L. Sensi and J. Y. Koh, Trends Pharmacol. Sci., 2000, 21, 395–401.
A. I. Bush, Alzheimer Dis. Assoc. Disord., 2003, 17, 147–150
C. J. Frederickson, M. D. Hernandez and J. F. McGinty, Brain Res., 1989, 480, 317–321
K. S. Aboody, A. Brown, N. G. Rainov, K. A. Bower, S. Liu, W. Yang, J. E. Small, U. Herrlinger, V. Ourednik, P. M. Black and X. O. Breakefield, Proc. Natl. Acad. Sci. U. S. A., 2000, 97, 12846–12851.
J. F. Zhang, S. Bhuniya, Y. H. Lee, C. Bae, J. H. Lee and J. S. Kim, Tetrahedron Lett., 2010, 51, 3719–3723
A. Voegelin, S. Pfister, A. C. Scheinost, M. A. Marcus and R. Kretzschmar, Environ. Sci. Technol., 2005, 39, 6616–6623
E. Callender and K. C. Rice, Environ. Sci. Technol., 2000, 34, 232–238
L. Li, Y. Q. Dang, H. W. Li, B. Wang and Y. Wu, Tetrahedron Lett., 2010, 51, 618–621.
Z. Xu, J. Yoon and D. R. Spring, Chem. Soc. Rev., 2010, 39, 1996–2006
P. Carol, S. Sreejith and A. Ajayaghosh, Chem. - Asian J., 2007, 2, 338–348
E. Kimura and T. Koike, Chem. Soc. Rev., 1998, 27, 179–184
H. G. Lee, J. H. Lee, S. P. Jang, H. M. Park, S. Kim, Y. Kim, C. Kim and R. G. Harrison, Tetrahedron, 2011, 67, 8073–8078.
A. K. Mahapatra, S. K. Manna, C. D. Mukhopadhyay and D. Mandal, Sens. Actuators, B, 2014, 200, 123–131
S. A. Ingale and F. Seela, J. Org. Chem., 2012, 77, 9352–9356
G. J. Park, M. M. Lee, G. R. You, Y. W. Choi and C. Kim, Tetrahedron Lett., 2014, 55, 2517–2522
Y. Chen, Y. Bai, Z. Han, W. He and Z. Guo, Chem. Soc. Rev., 2015, 44, 4517–4546.
J. Wang, Y. Li, E. Duah, S. Paruchuri, D. Zhou and Y. Pang, J. Mater. Chem. B, 2014, 2, 2008–2012
S. Lohar, S. Pal, M. Mukherjee, A. Maji, N. Demitri and P. Chattopadhyay, RSC Adv., 2017, 7, 25528–25534
Z. Dai and J. W. Canary, New J. Chem., 2007, 31, 1708–1718
M. Macias-Contreras, K. L. Daykin, J. T. Simmons, J. R. Allen, Z. S. Hooper, M. W. Davidson and L. Zhu, Org. Biomol. Chem., 2017, 15, 9139–9148.
P. S. Yao, Z. Liu, J. Z. Ge, Y. Chen and Q. Y. Cao, Dalton Trans., 2015, 44, 7470–7476
A. Gogoi, S. Samanta and G. Das, Sens. Actuators, B, 2014, 202, 788–794.
S. Goswami, A. K. Das, K. Aich, A. Manna, S. Maity, K. Khanra and N. Bhattacharyya, Analyst, 2013, 138, 4593–4598
Y. Ma, H. Chen, F. Wang, S. Kambam, Y. Wang, C. Mao and X. Chen, Dyes Pigm., 2014, 102, 301–307.
P. Manna, S. K. Seth, M. Mitra, S. R. Choudhury, A. Bauzâ, A. Frontera and S. Mukhopadhyay, Cryst. Growth Des., 2014, 14, 5812–5821
A. Das, A. D. Jana, S. K. Seth, B. Dey, S. R. Choudhury, T. Kar, S. Mukhopadhyay, N. J. Singh, I. C. Hwang and K. S. Kim, J. Phys. Chem. B, 2010, 114, 4166–4170
P. Manna, S. K. Seth, A. Bauzâ, M. Mitra, S. R. Choudhury, A. Frontera and S. Mukhopadhyay, Cryst. Growth Des., 2014, 14, 747–755
H. Hofmeier and U. S. Schubert, Chem. Soc. Rev., 2004, 33, 373–399
A. Wild, A. Winter, F. Schlütter and U. S. Schubert, Chem. Soc. Rev., 2011, 40, 1459–1511
T. Yasuda, I. Yamaguchi and T. Yamamoto, Adv. Mater., 2003, 15, 293–296
H. Li, S. J. Zhang, C. L. Gong, Y. F. Li, Y. Liang, Z. G. Qi and S. Chen, Analyst, 2013, 138, 7090–7093
B. G. Lohmeijer and U. S. Schubert, Macromol. Chem. Phys., 2003, 204, 1072–1078
R. Dobrawa, M. Lysetska, P. Ballester, M. Grüne and F. Würthner, Macromolecules, 2005, 38, 1315–1325.
P. C. Yang, H. Wu, C. L. Lee, W. C. Chen, H. J. He and M. T. Chen, Polymer, 2013, 54, 1080–1090
X. Lou, Y. Zhang, S. Li, D. Ou, Z. Wan, J. Qin and Z. Li, Polym. Chem., 2012, 3, 1446–1452.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, J. J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, T. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision D.01, Gaussian, Wallingford, CT, 2013.
L. Long, D. Zhang, X. Li, J. Zhang, C. Zhang and L. Zhou, Anal. Chim. Acta, 2013, 775, 100–105
S. Mondal, S. K. Manna, K. Maiti, R. Maji, S. S. Ali, S. Manna, S. Mandal, M. R. Uddin and A. K. Mahapatra, Supramol. Chem., 2017, 29, 616–626
S. Goswami, A. Manna, S. Paul, K. Aich, A. K. Das and S. Chakraborty, Dalton Trans., 2013, 42, 8078–8085.
H. A. Benesi and J. H. Hildebrand, J. Am. Chem. Soc., 1949, 71, 2703–2707
Y. Shiraishi, S. Sumiya, Y. Kohno and T. Hirai, J. Org. Chem., 2008, 73, 8571–8574
C. Yang, L. Liu, T. W. Mu and Q. X. Guo, Anal. Sci., 2000, 16, 537–539.
A. Duerrbeck, S. Gorelik, J. Hobley, A. M. Yong, G. S. Subramanian, A. Hor and N. Long, J. Mater. Chem. C, 2015, 3, 8992–9002.
A. D'Aléo, E. Cecchetto, L. De Cola and R. M. Williams, Sensors, 2009, 9, 3604–3626.
Y. P. Kumar, P. King and V. S. R. K. Prasad, Chem. Eng. J., 2006, 124, 63–70
J. H. Kim, J. Y. Noh, I. H. Hwang, J. Kang, J. Kim and C. Kim, Tetrahedron Lett., 2013, 56, 2415–2418.
Acknowledgements
TM and AH thankfully acknowledge the UGC, New Delhi for senior research fellowships. SP and SKM thank Haldia Government College for laboratory facility. AD is grateful to UGC, India for the DSK Postdoctoral fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mandal, T., Hossain, A., Dhara, A. et al. Terpyridine derivatives as “turn-on” fluorescence chemosensors for the selective and sensitive detection of Zn2+ ions in solution and in live cells. Photochem Photobiol Sci 17, 1068–1074 (2018). https://doi.org/10.1039/c8pp00186c
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c8pp00186c