Abstract
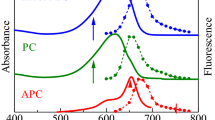
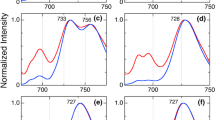
To improve the energy conversion efficiency of solar organic cells, the clue may lie in the development of devices inspired by an efficient light harvesting mechanism of some aquatic photosynthetic microorganisms that are adapted to low light intensity. Consequently, we investigated the pathways of excitation energy transfer (EET) from successive light harvesting pigments to the low energy level inside the phycobiliprotein antenna system of Acaryochloris marina, a cyanobacterium, using a time resolved absorption difference spectroscopy with a resolution time of 200 fs. The objective was to understand the actual biochemical process and pathways that determine the EET mechanism. Anisotropy of the EET pathway was calculated from the absorption change trace in order to determine the contribution of excitonic coupling. The results reveal a new electron energy relaxation pathway of 14 ps inside the phycocyanin component, which runs from phycocyanin to the terminal emitter. The bleaching of the 660 nm band suggests a broader absorption of the terminal emitter between 660 nm and 675 nm. Further, there are trimer depolarization kinetics of 450 fs and 500 fs in high and low ionic strength, respectively, which arise from the relaxation of the β84 and α84 in adjacent monomers of phycocyanin. Under conditions of low ionic strength buffer solution, the evolution of the kinetic amplitude during the depolarization of the trimer is suggestive of trimer conservation within the phycocyanin hexamer. The anisotropy values were 0.38 and 0.40 in high and in low ionic strength, respectively, indicating that there is no excitonic delocalization in the high energy level of phycocyanin hexamers.
Similar content being viewed by others
References
J. Wallentin, N. Anttu, D. Asoli, M. Huffman, I. Åberg, M. H. Magnusson, G. Siefer, P. Fuss-Kailuweit, F. Dimroth, B. Witzigmann, H. Q. Xu, L. Samuelson, K. Deppert, M. T. Borgström, Science 2013 339 1057–1060.
H. Miyashit, H. Ikemoto, N. Kurano, K. Adachi, M. Chihara, S. Miyachi, Nature 1996 383 402–402.
C. Chen, W. Cai, M. Long, B. Zhou, Y. Wu, D. Wu, Y. Feng, ACS Nano 2010 4 6425–6432.
A. W. D. Larkum, M. Kühl, Trends Plant Sci. 2005 10 355–357.
M. Akiyama, H. Miyashita, H. Kise, T. Watanabe, S. Miyachi, M. Kobayashi, Anal. Sci. 2001 17 205–208.
Q. Hu, J. Marquardt, I. Iwasaki, H. Miyashita, N. Kurano, E. Morschel, S. Miyachi, Biochim. Biophys. Acta, Bioenerg. 1999 1412 250–261.
E. Romero, R. Augulis, V. I. Novoderezhkin, M. Ferretti, J. Thieme, D. Zigmantas, R. van Grondelle, Nat. Phys. 2014 10 676–682.
P. Apotoli, Fresenius’ J. Anal. Chem. 1999 362 499–504.
N. Adir, Photosynth. Res. 2005 85 15–32.
C. Theiss, F.-J. Schmitt, J. Pieper, C. Nganou, M. Grehn, M. Vitali, R. Olliges, H. J. Eichler, H.-J. Eckert, J. Plant Physiol. 2011 168 1473–1487.
H. Liu, H. Zhang, D. M. Niedzwiedzki, M. Prado, G. He, M. L. Gross, R. E. Blankenship, Science 2013 342 1104–1107.
C. Nganou, J. Chem. Phys. 2013 139 10.1063/1.4813803.
A. R. Holzwarth, E. Bittersmann, W. Reuter, W. Wehrmeyer, Biophys. J. 1990 57 133–145.
K.-H. Zhao, P. Su, S. Böhm, B. Song, M. Zhou, C. Bubenzer, H. Scheer, Biochim. Biophys. Acta, Bioenerg. 2005 1706 81–87.
I. H. M. van Stokkum, D. S. Larsen, R. van Grondelle, Biochim. Biophys. Acta, Bioenerg. 2004 1657 82–104.
J.-m. Zhang, J.-q. Zhao, L.-j. Jiang, X.-g. Zheng, F.-l. Zhao, H.-z. Wang, Biochim. Biophys. Acta, Bioenerg. 1997 1320 285–296.
Å. Sandström, T. Gillbro, V. Sundström, R. Fischer, H. Scheer, Biochim. Biophys. Acta, Bioenerg. 1988 933 42–53.
M. Mimuro, C. Lipschultz, E. Gantt, Biochim. Biophys. Acta, Bioenerg. 1986 852 126–132.
D. J. Lundell, A. N. Glazer, J. Biol. Chem. 1983 258 8708–8713.
Z. Petrasek, F.-J. Schmitt, C. Theiss, J. Huyer, M. Chen, A. Larkum, H. J. Eichler, K. Kemnitz, H.-J. Eckert, Photochem. Photobiol. Sci. 2005 4 1016–1022.
T. Gillbro, Å. Sandström, V. Sundström, J. Wendler, A. R. Holzwarth, Biochim. Biophys. Acta, Bioenerg. 1985 808 52–65.
A. R. Holzwarth, J. Wendler, G. W. Suter, Biophys. J. 1987 51 1–12.
M. D. Edington, R. E. Riter, W. F. Beck, J. Phys. Chem. 1996 100 14206–14217.
J. R. Lakowicz, Plasmonics 2006 1 5–33.
M. D. Edington, R. E. Riter, W. F. Beck, J. Phys. Chem. 1995 99 5699–15704.
R. MacColl, Biochim. Biophys. Acta, Bioenerg. 2004 1657 73–81.
M. P. Debreczeny, K. Sauer, J. Zhou, D. A. Bryant, J. Phys. Chem. 1993 97 9852–9862.
K. Sauer, H. Scheer, Biochim. Biophys. Acta, Bioenerg. 1988 157–170.
T. Gillbro, A. V. Sharkov, I. V. Kryukov, E. V. Khoroshilov, P. G. Kryukov, R. Fischer, H. Scheer, Biochim. Biophys. Acta, Bioenerg. 1993 1140 321–326.
M. P. Debreczeny, K. Sauer, J. Zhou, D. A. Bryant, J. Phys. Chem. 1995 99 8420–8431.
M. Chen, M. Floetenmeyer, T. S. Bibby, FEBS Lett. 2009 583 2535–2539.
A. M. Weiner, Rev. Sci. Instrum. 2000 71 1929–1960.
C. Nganou, L. David, R. Meinke, N. Adir, J. Maultzsch, M. Mkandawire, D. Pouhè, C. Thomsen, J. Chem. Phys. 2014 140 10.1063/1.4866293.
A. Bandyopadhyay, M. J. Deen, L. E. Tarof, W. Clark, IEEE J. Quantum Electron. 1998 34 691–699.
M. J. Deen, and P. K. Basu, Silicon Photonics: Fundamentals and Devices, John Wiley & Sons, 2012, 10.1002/9781119945161.ch1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nganou, A.C., David, L., Adir, N. et al. Evidence of additional excitation energy transfer pathways in the phycobiliprotein antenna system of Acaryochloris marina. Photochem Photobiol Sci 14, 429–438 (2015). https://doi.org/10.1039/c4pp00352g
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c4pp00352g