Abstract
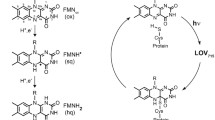
LOV domains are the light sensitive parts of phototropins and many other light-activated enzymes that regulate the response to blue light in plants and algae as well as some fungi and bacteria. Unlike all other biological photoreceptors known so far, the photocycle of LOV domains involves the excited triplet state of the chromophore. This chromophore is flavin mononucleotide (FMN) which forms a covalent adduct with a cysteine residue in the signaling state. Since the formation of this adduct from the triplet state involves breaking and forming of two bonds as well as a change from the triplet to the singlet spin state, various intermediates have been proposed, e.g. a protonated triplet state 3FMNH+, the radical anion 2FMN˙−, or the neutral semiquinone radical 2FMNH˙. We performed an extensive search for these intermediates by two-dimensional transient absorption (2D-TA) with a streak camera. However, no transient with a rate constant between the decay of fluorescence and the decay of the triplet state could be detected. Analysis of the decay associated difference spectra results in quantum yields for the formation of the adduct from the triplet of ΦA(LOV1) ≈ 0.75 and ΦA(LOV2) ≈ 0.80. This is lower than the values ΦA(LOV1) ≈ 0.95 and ΦA(LOV2) ≈ 0.99 calculated from the rate constants, giving indirect evidence of an intermediate that reacts either to form the adduct or to decay back to the ground state. Since there is no measurable delay between the decay of the triplet and the formation of the adduct, we conclude that this intermediate reacts much faster than it is formed. The LOV1-C57S mutant shows a weak and slowly decaying (τ > 100 µs) transient whose decay associated spectrum has bands at 375 and 500 nm, with a shoulder at 400 nm. This transient is insensitive to the pH change in the range 6.5–10.0 but increases on addition of ß-mercaptoethanol as the reducing agent. We assign this intermediate to the radical anion which is protected from protonation by the protein. We propose that the adduct is formed via the same intermediate by combination of the radical ion pair.
Article PDF
Similar content being viewed by others
References
J. M. Christie, M. Salomon, K. Nozue, M. Wada and W. R. Briggs, LOV (light, oxygen, or voltage) domains of the blue-light photoreceptor phototropin (nph1): Binding sites for the chromophore flavin mononucleotide Proc. Natl. Acad. Sci. U. S. A. 1999 96 8779–8783.
J. A. Jarillo, H. Gabrys, J. Capel, J. M. Alonso, J. R. Ecker and A. R. Cashmore, Phototropin-related NPL1 controls chloroplast relocation induced by blue light Nature 2001 410 952–954.
T. Kagawa, T. Sakai, N. Suetsugu, K. Oikawa, S. Ishiguro, T. Kato, S. Tabata, K. Okada and M. Wada, Arabidopsis NPL1: A phototropin homolog controlling the chloroplast high-light avoidance response Science 2001 291 2138–2141.
T. Kinoshita, M. Doi, N. Suetsugu, T. Kagawa, M. Wada, K.-i. Shimazaki, phot1 and phot2 mediate blue light regulation of stomatal opening Nature 2001 414 656–660.
J. M. Christie, P. Reymond, G. K. Powell, P. Bernasconi, A. A. Raibekas, E. Liscum and W. R. Briggs, Arabidopsis NPH1: a flavoprotein with the properties of a photoreceptor for phototropism Science 1998 282 1698–1701.
E. Huala, P. W. Oeller, E. Liscum, I.-S. Han, E. Larsen and W. R. Briggs, Arabidopsis NPH1: a protein kinase with a putative redox-sensing domain Science 1997 278 2120–2123.
K. Huang and C. F. Beck, Phototropin is the blue-light receptor that controls multiple steps in the sexual life cycle of the green alga Chlamydomonas reinhardtii Proc. Natl. Acad. Sci. U. S. A. 2003 100 6269–6274.
M. Salomon, J. M. Christie, E. Knieb, U. Lempert and W. R. Briggs, Photochemical and Mutational Analysis of the FMN-Binding Domains of the Plant Blue Light Receptor, Phototropin Biochemistry 2000 39 9401–9410.
T. E. Swartz, S. B. Corchnoy, J. M. Christie, J. W. Lewis, I. Szundi, W. R. Briggs and R. A. Bogomolni, The photocycle of a flavin-binding domain of the blue light photoreceptor phototropin J. Biol. Chem. 2001 276 36493–36500.
J. T. M. Kennis, S. Crosson, M. Gauden, I. H. M. van Stokkum, K. Moffat, R. van Grondelle, Primary Reactions of the LOV2 Domain of Phototropin, a Plant Blue-Light Photoreceptor Biochemistry 2003 42 3385–3392.
A. Losi, Flavin-based blue-light photosensors: a photobiophysics update Photochem. Photobiol. 2007 83 1283–1300.
S.-H. Song, D. Madsen, J. B. van der Steen, R. Pullman, L. H. Freer, K. J. Hellingwerf and D. S. Larsen, Primary Photochemistry of the Dark- and Light-Adapted States of the YtvA Protein from Bacillus subtilis Biochemistry 2013 52 7951–7963.
M. Salomon, W. Eisenreich, H. Dürr, E. Schleicher, E. Knieb, V. Massey, W. Rüdiger, F. Müller, A. Bacher and G. Richter, An optomechanical transducer in the blue light receptor phototropin from Avena sativa Proc. Natl. Acad. Sci. U. S. A. 2001 98 12357–12361.
S. Crosson and K. Moffat, Photoexcited Structure of a Plant Photoreceptor Domain Reveals a Light-Driven Molecular Switch Plant Cell 2002 14 1067–1075.
R. Fedorov, I. Schlichting, E. Hartmann, T. Domratcheva, M. Fuhrmann and P. Hegemann, Crystal structures and molecular mechanism of a light-induced signaling switch: The Phot-LOV1 domain from Chlamydomonas reinhardtii Biophys. J. 2003 84 2474–2482.
A. Losi, B. Quest and W. Gaertner, Listening to the blue: the time-resolved thermodynamics of the bacterial blue-light receptor YtvA and its isolated LOV domain Photochem. Photobiol. Sci. 2003 2 759–766.
S. Kikuchi, M. Unno, K. Zikihara, S. Tokutomi and S. Yamauchi, Vibrational Assignment of the Flavin-Cysteinyl Adduct in a Signaling State of the LOV Domain in FKF1 J. Phys. Chem. B 2009 113 2913–2921.
Y. Nakasone, K. Zikihara, S. Tokutomi and M. Terazima, Kinetics of Conformational Changes of the FKF1-LOV Domain upon Photoexcitation. Biophys. J. 2010 99 3831–3839.
W. Holzer, A. Penzkofer, M. Fuhrmann and P. Hegemann, Spectroscopic characterization of flavin mononucleotide bound to the LOV1 domain of Phot1 from Chlamydomonas reinhardtii Photochem. Photobiol. 2002 75 479–487.
T. Kottke, J. Heberle, D. Hehn, B. Dick and P. Hegemann, Phot-LOV1: Photocycle of a blue-light receptor domain from the green alga Chlamydomonas reinhardtii Biophys. J. 2003 84 1192–1201.
H. Guo, T. Kottke, P. Hegemann and B. Dick, The phot LOV2 domain and its interaction with LOV1 Biophys. J. 2005 89 402–412.
T. Bednarz, A. Losi, W. Gaertner, P. Hegemann and J. Heberle, Functional variations among LOV domains as revealed by FT-IR difference spectroscopy Photochem. Photobiol. Sci. 2004 3 575–579.
M. A. van der Horst and K. J. Hellingwerf, Photoreceptor proteins, “star actors of modern times”: a review of the functional dynamics in the structure of representative members of six different photoreceptor families Acc. Chem. Res. 2004 37 13–20.
A. Moeglich, X. Yang, R. A. Ayers and K. Moffat, Structure and function of plant photoreceptors Annu. Rev. Plant Biol. 2010 61 21–47.
O. P. Ernst, D. T. Lodowski, M. Elstner, P. Hegemann, L. S. Brown and H. Kandori, Microbial and animal rhodopsins: Structures, functions, and molecular mechanisms Chem. Rev. 2014 114 126–163.
M. T. A. Alexandre, T. Domratcheva, C. Bonetti, L. J. G. W. van Wilderen, R. van Grondelle, M.-L. Groot, K. J. Hellingwerf and J. T. M. Kennis, Primary reactions of the LOV2 domain of phototropin studied with ultrafast mid-infrared spectroscopy and quantum chemistry Biophys. J. 2009 97 227–237.
C. Thoeing, A. Pfeifer, S. Kakorin and T. Kottke, Protonated triplet-excited flavin resolved by step-scan FTIR spectroscopy: implications for photosensory LOV domains Phys. Chem. Chem. Phys. 2013 15 5916–5926.
C. Neiss and P. Saalfrank, Ab initio quantum chemical investigation of the first steps of the photocycle of phototropin: a model study Photochem. Photobiol. 2003 77 101–109.
E. Schleicher, R. M. Kowalczyk, C. W. M. Kay, P. Hegemann, A. Bacher, M. Fischer, R. Bittl, G. Richter and S. Weber, On the Reaction Mechanism of Adduct Formation in LOV Domains of the Plant Blue-Light Receptor Phototropin J. Am. Chem. Soc. 2004 126 11067–11076.
M. Dittrich, P. L. Freddolino and K. Schulten, When light falls in LOV: A quantum mechanical/molecular mechanical study of photoexcitation in Phot-LOV1 of Chlamydomonas reinhardtii J. Phys. Chem. B 2005 109 13006–13013.
T. Domratcheva, R. Fedorov and I. Schlichting, Analysis of the Primary Photocycle Reactions Occurring in the Light, Oxygen, and Voltage Blue-Light Receptor by Multiconfigurational Quantum-Chemical Methods J. Chem. Theory Comput. 2006 2 1565–1574.
S. B. Corchnoy, T. E. Swartz, J. W. Lewis, I. Szundi, W. R. Briggs and R. A. Bogomolni, Intramolecular Proton Transfers and Structural Changes during the Photocycle of the LOV2 Domain of Phototropin 1 J. Biol. Chem. 2003 278 724–731.
G. Noell, G. Hauska, P. Hegemann, K. Lanzl, T. Noell, M. von Sanden-Flohe and B. Dick, Redox properties of LOV domains: chemical versus photochemical reduction, and influence on the photocycle ChemBioChem 2007 8 2256–2264.
K. Lanzl, G. Noell and B. Dick, LOV1 protein from Chlamydomonas reinhardtii is a template for the photoadduct formation of FMN and methylmercaptane ChemBioChem 2008 9 861–864.
K. Lanzl, M. v. Sanden-Flohe, R.-J. Kutta and B. Dick, Photoreaction of mutated LOV photoreceptor domains from Chlamydomonas reinhardtii with aliphatic mercaptans: Implications for the mechanism of wild-type LOV Phys. Chem. Chem. Phys. 2010 12 6594–6604.
C. Bauer, C.-R. Rabi, J. Heberle and T. Kottke, Indication for a radical intermediate preceding the signaling state in the LOV domain photocycle Photochem. Photobiol. 2011 87 548–553.
R.-J. Kutta, T. Langenbacher, U. Kensy and B. Dick, Setup and performance of a streak camera apparatus for transient absorption measurements in the ns to ms range Appl. Phys. B: Lasers Opt. 2013 111 203–216.
T. Kottke, B. Dick, R. Fedorov, I. Schlichting, R. Deutzmann and P. Hegemann, Irreversible Photoreduction of Flavin in a Mutated Phot-LOV1 Domain Biochemistry 2003 42 9854–9862.
B. J. Fritz, K. Matsui, S. Kasai and A. Yoshimura, Triplet lifetimes of some flavins Photochem. Photobiol. 1987 45 539–541.
K. Zenichowski, M. Gothe and P. Saalfrank, Exciting flavins: Absorption spectra and spin–orbit coupling in light-oxygen-voltage (LOV) domains J. Photochem. Photobiol., A 2007 190 290–300.
S. Salzmann, V. Martinez-Junza, B. Zorn, S. E. Braslavsky, M. Mansurova, C. M. Marian and W. Gaertner, Photophysical Properties of Structurally and Electronically Modified Flavin Derivatives Determined by Spectroscopy and Theoretical Calculations J. Phys. Chem. A 2009 113 9365–9375.
S. Salzmann and C. M. Marian, The photophysics of alloxazine: a quantum chemical investigation in vacuum and solution. Photochem. Photobiol. Sci. 2009 8 1655–1666.
B. Liu, H. Liu, D. Zhong and C. Lin, Searching for a photocycle of the cryptochrome photoreceptors Curr. Opin. Plant Biol. 2010 13 578–586.
U. Megerle, M. Wenninger, R.-J. Kutta, R. Lechner, B. König, B. Dick and E. Riedle, Unraveling the flavin-catalyzed photooxidation of benzylic alcohol with transient absorption spectroscopy from sub-pico- to microseconds. Phys. Chem. Chem. Phys. 2011 13 8869–8880.
G. Richter, S. Weber, W. Roemisch, A. Bacher, M. Fischer and W. Eisenreich, Photochemically Induced Dynamic Nuclear Polarization in a C450A Mutant of the LOV2 Domain of the Avena sativa Blue-Light Receptor Phototropin J. Am. Chem. Soc. 2005 127 17245–17252.
C. W. M. Kay, E. Schleicher, A. Kuppig, H. Hofner, W. Ruediger, M. Schleicher, M. Fischer, A. Bacher, S. Weber and G. Richter, Blue light perception in plants. J. Biol. Chem. 2003 278 10973–10982.
V. Massey and G. Palmer, The existence of spectrally distinct classes of flavoprotein semiquinones. A new method for the quantitative production of flavoprotein semiquinones Biochemistry 1966 5 3181–3189.
K. B. Henbest, K. Maeda, P. J. Hore, M. Joshi, A. Bacher, R. Bittl, S. Weber, C. R. Timmel and E. Schleicher, Magnetic-field effect on the photoactivation reaction of Escherichia coli DNA photolyase Proc. Natl. Acad. Sci. U. S. A. 2008 105 14395–14399.
R. J. Kutta, U. Kensy, and B. Dick, Measuring photochemical intermediates with a streak camera–a new technique for transient absorption, in Chemical Photocatalysis, ed. B. König De Gruyter, 2013.
R. Miura, Versatility and specificity in flavoenzymes: control mechanisms of flavin reactivity Chem. Rec. 2001 1 183–194.
T. Langenbacher, D. Immeln, B. Dick and T. Kottke, Microsecond Light-induced Proton Transfer to Flavin in the Blue Light Sensor Plant Cryptochrome J. Am. Chem. Soc. 2009 131 14274–14280.
M. Sakai and H. Takahashi, One-electron photoreduction of flavin mononucleotide: time-resolved resonance Raman and absorption study J. Mol. Struct. 1996 379 9–18.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available. See DOI: 10.1039/c4pp00155a
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kutta, R.J., Magerl, K., Kensy, U. et al. A search for radical intermediates in the photocycle of LOV domains. Photochem Photobiol Sci 14, 288–299 (2015). https://doi.org/10.1039/c4pp00155a
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c4pp00155a