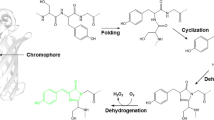
Abstract
Green Fluorescent Protein (GFP) and other related fluorescent proteins are generally used as genetically encoded, chemically inert labels in vivo. This review focuses on the emerging application of fluorescent proteins as light-inducible intracellular photochemical partners. The first example of a chemically active GFP-like protein was the phototoxic red fluorescent protein KillerRed, which can be used for precise light-induced killing of cells, protein inactivation, and studying reactive oxygen species signaling in different cellular compartments. Moreover, recent studies revealed that various GFPs can act as light-induced electron donors in photochemical reactions with biologically relevant electron acceptors. These findings have important implications for practical uses of fluorescent proteins as well as for our understanding of the evolution and biology of this protein family
Similar content being viewed by others
References
D. M. Chudakov, S. Lukyanov, K. A. Lukyanov, Fluorescent proteins as a toolkit for in vivo imaging, Trends Biotechnol., 2005, 23, 605–613.
F. H. Johnson, O. Shimomura, Y. Saiga, L. C. Gershman, G. T. Reynolds, J. R. Waters, Quantum efficiency of Cypridina luminescence, with a note on that of Aequorea, J. Cell. Comp. Physiol., 1962, 60, 85–103.
D. C. Prasher, V. K. Eckenrode, W. W. Ward, F. G. Prendergast, M. J. Cormier, Primary structure of the Aequorea victoria green-fluorescent protein, Gene, 1992, 111, 229–233.
M. Chalfie, Y. Tu, G. Euskirchen, W. W. Ward, D. C. Prasher, Green fluorescent protein as a marker for gene expression, Science, 1994, 263, 802–805.
S. Wang, T. Hazelrigg, Implications for bcd mRNA localization from spatial distribution of exu protein in Drosophila oogenesis, Nature, 1994, 369, 400–403.
M. V. Matz, A. F. Fradkov, Y. A. Labas, A. P. Savitsky, A. G. Zaraisky, M. L. Markelov, S. A. Lukyanov, Fluorescent proteins from nonbioluminescent Anthozoa species, Nat. Biotechnol., 1999, 17, 969–973.
S. H. Haddock, N. Mastroianni, L. M. Christianson, A photoactivatable green-fluorescent protein from the phylum Ctenophora, Proc. R. Soc. London, Ser. B, 2010, 277, 1155–1160.
D. A. Shagin, E. V. Barsova, Y. G. Yanushevich, A. F. Fradkov, K. A. Lukyanov, Y. A. Labas, T. N. Semenova, J. A. Ugalde, A. Meyers, J. M. Nunez, E. A. Widder, S. A. Lukyanov, M. V. Matz, GFP-like proteins as ubiquitous metazoan superfamily: evolution of functional features and structural complexity, Mol. Biol. Evol., 2004, 21, 841–850.
D. D. Deheyn, K. Kubokawa, J. K. McCarthy, A. Murakami, M. Porrachia, G. W. Rouse, N. D. Holland, Endogenous green fluorescent protein (GFP) in amphioxus, Biol. Bull., 2007, 213, 95–100.
B. Seefeldt, R. Kasper, T. Seidel, P. Tinnefeld, K. J. Dietz, M. Heilemann, M. Sauer, Fluorescent proteins for single-molecule fluorescence applications, J. Biophotonics, 2008, 1, 74–82.
D. Shcherbo, E. A. Souslova, J. Goedhart, T. V. Chepurnykh, A. Gaintzeva Shemyakina, II, T. W. Gadella, S. Lukyanov, D. M. Chudakov, Practical and reliable FRET/FLIM pair of fluorescent proteins, BMC Biotechnol., 2009, 9, 24.
J. Goedhart, L. van Weeren, M. A. Hink, N. O. Vischer, K. Jalink, T. W. Gadella, Jr., Bright cyan fluorescent protein variants identified by fluorescence lifetime screening, Nat. Methods, 2010, 7, 137–139.
V. V. Verkhusha, K. A. Lukyanov, The molecular properties and applications of Anthozoa fluorescent proteins and chromoproteins, Nat. Biotechnol., 2004, 22, 289–296.
T. A. Schüttrigkeit, T. Feilitzsch, C. K. Kompa, K. A. Lukyanov, A. P. Savitsky, A. A. Voityuk, M. E. Michel-Beyerl, Femtosecond study of light-induced fluorescence increase of the dark chromoprotein asFP595, Chem. Phys., 2006, 323, 149–160.
Y. Tanaka, N. Sasaki, A. Ohmiya, Biosynthesis of plant pigments: anthocyanins, betalains and carotenoids, Plant J., 2008, 54, 733–749.
S. J. Remington, Fluorescent proteins: maturation, photochemistry and photophysics, Curr. Opin. Struct. Biol., 2006, 16, 714–721.
A. A. Pakhomov, V. I. Martynov, GFP family: structural insights into spectral tuning, Chem. Biol., 2008, 15, 755–764.
M. Ormo, A. B. Cubitt, K. Kallio, L. A. Gross, R. Y. Tsien, S. J. Remington, Crystal structure of the Aequorea victoria green fluorescent protein, Science, 1996, 273, 1392–1395.
C. W. Cody, D. C. Prasher, W. M. Westler, F. G. Prendergast, W. W. Ward, Chemical structure of the hexapeptide chromophore of the Aequorea green-fluorescent protein, Biochemistry, 1993, 32, 1212–1218.
K. A. Lukyanov, D. M. Chudakov, S. Lukyanov, V. V. Verkhusha, Innovation: Photoactivatable fluorescent proteins, Nat. Rev. Mol. Cell Biol., 2005, 6, 885–891.
R. Ando, H. Hama, M. Yamamoto-Hino, H. Mizuno, A. Miyawaki, An optical marker based on the UV-induced green-to-red photoconversion of a fluorescent protein, Proc. Natl. Acad. Sci. U. S. A., 2002, 99, 12651–12656.
H. Mizuno, T. K. Mal, K. I. Tong, R. Ando, T. Furuta, M. Ikura, A. Miyawaki, Photo-induced peptide cleavage in the green-to-red conversion of a fluorescent protein, Mol. Cell, 2003, 12, 1051–1058.
F. V. Subach, G. H. Patterson, S. Manley, J. M. Gillette, J. Lippincott-Schwartz, V. V. Verkhusha, Photoactivatable mCherry for high-resolution two-color fluorescence microscopy, Nat. Methods, 2009, 6, 153–159.
J. J. van Thor, T. Gensch, K. J. Hellingwerf, L. N. Johnson, Phototransformation of green fluorescent protein with UV and visible light leads to decarboxylation of glutamate 222, Nat. Struct. Biol., 2002, 9, 37–41.
J. N. Henderson, R. Gepshtein, J. R. Heenan, K. Kallio, D. Huppert, S. J. Remington, Structure and mechanism of the photoactivatable green fluorescent protein, J. Am. Chem. Soc., 2009, 131, 4176–4177.
L. Greenbaum, C. Rothmann, R. Lavie, Z. Malik, Green fluorescent protein photobleaching: a model for protein damage by endogenous and exogenous singlet oxygen, Biol. Chem., 2000, 381, 1251–1258.
A. Jimenez-Banzo, S. Nonell, J. Hofkens, C. Flors, Singlet oxygen photosensitization by EGFP and its chromophore HBDI, Biophys. J., 2008, 94, 168–172.
Z. Rajfur, P. Roy, C. Otey, L. Romer, K. Jacobson, Dissecting the link between stress fibres and focal adhesions by CALI with EGFP fusion proteins, Nat. Cell Biol., 2002, 4, 286–293.
O. Tour, R. M. Meijer, D. A. Zacharias, S. R. Adams, R. Y. Tsien, Genetically targeted chromophore-assisted light inactivation, Nat. Biotechnol., 2003, 21, 1505–1508.
M. E. Bulina, D. M. Chudakov, O. V. Britanova, Y. G. Yanushevich, D. B. Staroverov, T. V. Chepurnykh, E. M. Merzlyak, M. A. Shkrob, S. Lukyanov, K. A. Lukyanov, A genetically encoded photosensitizer, Nat. Biotechnol., 2006, 24, 95–99.
M. E. Bulina, K. A. Lukyanov, O. V. Britanova, D. Onichtchouk, S. Lukyanov, D. M. Chudakov, Chromophore-assisted light inactivation (CALI) using the phototoxic fluorescent protein KillerRed, Nat. Protoc., 2006, 1, 947–953.
W. Waldeck, G. Mueller, M. Wiessler, M. Brom, K. Toth, K. Braun, Autofluorescent proteins as photosensitizer in eukaryontes, Int. J. Med. Sci., 2009, 6, 365–373.
E. O. Serebrovskaya, E. F. Edelweiss, O. A. Stremovskiy, K. A. Lukyanov, D. M. Chudakov, S. M. Deyev, Targeting cancer cells by using an antireceptor antibody-photosensitizer fusion protein, Proc. Natl. Acad. Sci. U. S. A., 2009, 106, 9221–9225.
A. S. Sobolev, Modular transporters for subcellular cell-specific targeting of antitumor drugs, BioEssays, 2008, 30, 278–287.
S. Pletnev, N. Gurskaya, N. V. Pletneva, K. A. Lukyanov, D. M. Chudakov, V. I. Martynov, V. O. Popov, M. V. Kovalchuk, A. Wlodawer, Z. Dauter, V. Pletnev, Structural basis for phototoxicity of the genetically encoded photosensitizer KillerRed, J. Biol. Chem., 2009, 284, 32028–32039.
P. Carpentier, S. Violot, L. Blanchoin, D. Bourgeois, Structural basis for the phototoxicity of the fluorescent protein KillerRed, FEBS Lett., 2009, 583, 2839–2842.
O. M. Subach, V. N. Malashkevich, W. D. Zencheck, K. S. Morozova, K. D. Piatkevich, S. C. Almo, V. V. Verkhusha, Structural characterization of acylimine-containing blue and red chromophores in mTagBFP and TagRFP fluorescent proteins, Chem. Biol., 2010, 17, 333–341.
A. M. Bogdanov, A. S. Mishin, I. V. Yampolsky, V. V. Belousov, D. M. Chudakov, F. V. Subach, V. V. Verkhusha, S. Lukyanov, K. A. Lukyanov, Green fluorescent proteins are light-induced electron donors, Nat. Chem. Biol., 2009, 5, 459–461.
A. M. Bogdanov, E. A. Bogdanova, D. M. Chudakov, T. V. Gorodnicheva, S. Lukyanov, K. A. Lukyanov, Cell culture medium affects GFP photostability: a solution, Nat. Methods, 2009, 6, 859–860.
C. C. Page, C. C. Moser, X. Chen, P. L. Dutton, Natural engineering principles of electron tunnelling in biological oxidation-reduction, Nature, 1999, 402, 47–52.
E. Epifanovsky, I. Polyakov, B. Grigorenko, A. Nemukhin, A. I. Krylov, The effect of oxidation on the electronic structure of the green fluorescent protein chromophore, J. Chem. Phys., 2010, 132, 115104.
M. Fernandez-Suarez, A. Y. Ting, Fluorescent probes for super-resolution imaging in living cells, Nat. Rev. Mol. Cell Biol., 2008, 9, 929–943.
M. Chalfie, Green fluorescent protein, Photochem. Photobiol., 1995, 62, 651–656.
A. Salih, A. Larkum, G. Cox, M. Kuhl, Fluorescent pigments in corals are photoprotective, Nature, 2000, 408, 850–853.
S. F. Field, M. Y. Bulina, I. V. Kelmanson, J. P. Bielawski, M. V. Matz, Adaptive evolution of multicolored fluorescent proteins in reef-building corals, J. Mol. Evol., 2006, 62, 332–339.
M. V. Matz, N. J. Marshall, M. Vorobyev, Are corals colorful?, Photochem. Photobiol., 2006, 82, 345–350.
F. Bou-Abdallah, N. D. Chasteen, M. P. Lesser, Quenching of superoxide radicals by green fluorescent protein, Biochim. Biophys. Acta, Gen. Subj., 2006, 1760, 1690–1695.
N. Agmon, Proton pathways in green fluorescence protein, Biophys. J., 2005, 88, 2452–2461.
J. A. Ugalde, B. S. W. Chang, M. V. Matz, Evolution of coral pigments recreated, Science, 2004, 305, 1433.
N. O. Alieva, K. A. Konzen, S. F. Field, E. A. Meleshkevitch, M. E. Hunt, V. Beltran-Ramirez, D. J. Miller, J. Wiedenmann, A. Salih, M. V. Matz, Diversity and evolution of coral fluorescent proteins, PLoS One, 2008, 3, e2680.
J. Lin, I. A. Balabin, D. N. Beratan, The nature of aqueous tunneling pathways between electron-transfer proteins, Science, 2005, 310, 1311–1313.
O. Miyashita, M. Y. Okamura, J. N. Onuchic, Interprotein electron transfer from cytochrome c2 to photosynthetic reaction center: tunneling across an aqueous interface, Proc. Natl. Acad. Sci. U. S. A., 2005, 102, 3558–3563.
G. Sainz, C. J. Carrell, M. V. Ponamarev, G. M. Soriano, W. A. Cramer, J. L. Smith, Interruption of the internal water chain of cytochrome f impairs photosynthetic function, Biochemistry, 2000, 39, 9164–9173.
M. V. Ponamarev, W. A. Cramer, Perturbation of the internal water chain in cytochrome f of oxygenic photosynthesis: loss of the concerted reduction of cytochromes f and b6, Biochemistry, 1998, 37, 17199–17208.
Author information
Authors and Affiliations
Additional information
This article is published as part of a themed issue on photofunctional proteins: from understanding to engineering.
Rights and permissions
About this article
Cite this article
Lukyanov, K.A., Serebrovskaya, E.O., Lukyanov, S. et al. Fluorescent proteins as light-inducible photochemical partners. Photochem Photobiol Sci 9, 1301–1306 (2010). https://doi.org/10.1039/c0pp00114g
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c0pp00114g