Abstract
High-throughput characterization by soft X-ray absorption spectroscopy (XAS) and electrochemical characterization is used to establish a correlation between electronic structure and catalytic activity of oxygen evolution reaction (OER) catalysts. As a model system a quasi-ternary materials library of Ni1-y-zFeyCrzOx was synthesized by combinatorial reactive magnetron sputtering, characterized by XAS, and an automated scanning droplet cell. The presence of Cr was found to increase the OER activity in the investigated compositional range. The electronic structure of NiII and CrIII remains unchanged over the investigated composition spread. At the Fe L-edge a linear combination of two spectra was observed. These spectra were assigned to FeIII in Oh symmetry and FeIII in Td symmetry. The ratio of FeIII Oh to FeIII Td increases with the amount of Cr and a correlation between the presence of the FeIII Oh and a high OER activity is found.
Similar content being viewed by others
Introduction
To acquire an in-depth understanding of specific catalysts’ properties advanced analytical techniques are needed that can correlate electronic structure to functional properties. This may lead to the formulation of generic pathways and design criteria for highly complex reactions such as the oxygen evolution reaction (OER). One of such analysis techniques is X-ray absorption spectroscopy (XAS) that directly probes the unoccupied electronic structure. Making this technique accessible for combinatorial materials science allows for the correlation of electronic structure, crystallographic data and electrochemical performance and therefore expedites materials design. In the present work, a first attempt is presented to correlate electronic structure and electrochemical properties probed by high-throughput transition metal L-edges XAS and a scanning droplet cell (SDC) on a Ni-Fe-Cr-O continuous composition spread combinatorial materials library for the OER. The system Ni1-y-zFeyCrzOx was chosen, because Ni-Fe oxides1,2,3,4 are relevant OER catalysts and it was shown that the alloying of Cr into the spinel increased the electrocatalytic activity of Ni1-yFeyO45.
Complementary to the here in proposed experimental route of gaining a mechanistic understanding, high-throughput computational studies6,7,8,9 aiming at theory driven discovery were presented in literature. An important example for computationally guided design is found for perovskite based OER catalysts. For perovskite OER catalysts a generally accepted design principle is based on the occupancy of the eg (σ) orbital close to unity if the ligand environment of the transition metal shows octahedral (Oh) symmetry10,11,12. Other factors such as covalence, 3d occupation and geometry, however, are important as well13. The reasoning behind the single occupancy of the eg orbital is, that in this case the bonding strength between transition metal and absorbed oxygen species is neither too strong (low eg occupancy) nor too weak (high eg occupancy) and thus the Sabatier principle is fulfilled.
The correlation of such fundamental changes in electronic structure and their subtle influence on electrochemical properties, is however already challenging to elucidate for quasi-binary oxides. Whereas until very recently it was assumed for Ni-Fe-O catalysts that Ni was the catalytically active site14, recent DFT studies supported by XAS data suggest that Fe is the active center for the observed OER activity15. For Ni1-y-zFeyCrzOx, which is the focus of this work, Singh et al. revealed by XPS that the surface of the film contains Ni2+, Fe3+, Cr3+ and Cr6+ and found an improvement of OER with the incorporation of Cr. They tentatively assigned the hexavalent Cr-ion as OER active center, but the exact role of Cr is not well understood5.
For a thorough understanding of the functioning of OER catalysts direct probing of the involved 3d orbitals is performed by XAS at the transition metal L-edges. Combined with high-throughput electrochemical measurements an experimental study on the correlation of electronic structure and electrochemistry of Ni1-y-zFeyCrzOx based OER catalyst is presented. In this study 130 elemental compositions of Ni1-y-zFeyCrzOx were probed by XAS at all three metal L-edges and subsequently investigated for their electrochemical activity.
Results
The materials library of Ni1-y-zFeyCrzOx was centered around the composition 50 at. % Ni, 25 at. % Fe and 25 at. % Cr omitting the oxygen content as shown in the ternary composition spread in Fig. 1. Unless stated otherwise, the elemental compositions are reported without oxygen as it could not reliably be determined using energy dispersive X-ray spectroscopy (EDX). The obtained compositional spread was 10–30 at. % Cr, 20–55 at. % Fe, and 40–65 at. % Ni.
Electrochemical evaluation
To evaluate the electrochemical activity for the OER in correlation with XAS the materials library was investigated by an automated SDC. The corresponding Tafel slopes (surface area independent) and current densities are reported.
In Fig. 1 the current density at an overpotential of η = 320 mV (1.55 V vs. reversible hydrogen electrode) is shown. At this potential the oxygen evolution rate is low and hence the current density is solely determined by the OER kinetics. A clear trend to higher current densities with higher Cr content is observed. The current densities obtained at these relatively low overpotentials, even with assumed flat surfaces, are relevant for the application in solar water splitting where current densities between 1–10 mA/cm2 are commonly achieved. For practical applications the current density can be increased by increasing the electrochemical active surface area, e.g. by nanostructuring. In order to exclude that the increase of current density that we observed is caused by changes in the surface area the sample was investigated with atomic force microscopy (AFM). AFM images were recorded in total at ten different positions of the materials library. These positions were systematically chosen to cover the full range of the investigated material compositions in order to test for material composition dependent changes in morphology. In Figure S1 in the Supplementary Information AFM measurements of the Ni1-y-zFeyCrzOx materials library at compositions with low Cr content (50 at. % Ni, 35 at. % Fe, 15 at. % Cr) and high Cr content (48 at. % Ni, 12 at. % Fe, 40 at. % Cr) are shown as examples. The investigated surfaces have the same electrochemically active surface area within the error range. (rms-roughness: 37 ± 1 nm, ratio of microscopic to geometric surface area: 1.228 ± 0.002).
The Tafel slope is shown in Fig. 2. The observed Tafel slopes are in the range from 70 to 115 mV/decade, which is rather high as compared to 60 mV/decade observed for Co-perovskites16 or amorphous Co-oxides17,18 and 40 mV/decade measured for Ni/Fe-oxides5,14,19,20. The lower thermodynamic limits in the Butler-Volmer formalism predict Tafel slopes of 24, 40 or 60 mV/decade21. It should be noted that in a scanning droplet cell slightly different conditions as compared with rotating disk electrode experiment exist, meaning that the reported Tafel slopes are rather an upper boundary. Since the Tafel slope is relatively constant above 30% Fe, it is probable that the rate-determining step (RDS) stays the same in this region. Below 30% Fe the Tafel slope increases from around 70 mV/decade to up to 115 mV/decade.
XAS at the transition metal L-edges
To gain insight into the changes of the electronic structure of the involved transition metals, the transition metals L-edges were evaluated across the materials library. No changes were found across the investigated elemental compositions in the L-edge spectra of Ni and Cr, whereas for Fe drastic changes occurred. A characteristic spectrum of the Ni L-edge is shown in Fig. 3. This spectrum is in very good agreement with the NiO reference spectrum22 shown in the same Fig., which indicates that Ni prevails as NiII in the materials library. A characteristic Cr spectrum of the measurement series is shown in Fig. 4. Based on the Cr2FeO4 reference spectrum23 it was identified as a CrIII species.
The Ni L3-edge was found to be constant across the entire materials library. The NiO reference spectrum is adopted from22 and shifted downwards by 0.5 for better readability.
No variation of the Cr L-edges is observed across the entire materials library. The reference spectrum of Cr2FeO4 is adopted from23 and shifted downwards by 0.5 for better readability.
Examples of Fe L-edge spectra are shown in Fig. 5. Fe is present in the FeIII state as deduced from the energy positions of the L-edge spectra24. Two linear components were determined from the dataset of Fe L-edges as shown in Fig. 6. The first component is very similar to the spectrum of Fe2O3 in accordance with FeIII in octahedral symmetry, thus we denote this spectrum as FeIII Oh. The second component is similar to spectra of the compound FePO4 in which FeIII is in tetrahedral coordination25 and this spectrum is denoted as FeIII Td. A small energy shift was found between the Fe L-edge of FePO4 obtained from literature and the observed FeIIIB species. The presence of FeIII in D4h symmetry was excluded by comparison to multiplet simulations shown in Fig. S2 in the Supplementary Information.
The two spectra are linear components of the Fe L3-edge spectra of Ni1-y-zFeyCrzOx given in Fig. S2 in the Supplementary Information. The Fe2O3 reference spectrum is taken from22 and shifted downwards by 0.5 for better readability.
Linear combination fitting was performed on the whole dataset of Fe L-edges with the two spectra FeIII Oh and FeIII Td. No systematic trend of the error of the fit was found (see Fig. S3 in the Supplementary Information), which confirms that indeed the chosen references are adequate for the fit and no third component is present. The thus obtained amounts of the species FeIII Oh and FeIII Td are shown in Fig. 7 and as ternary plot as Fig. S4 in the Supplementary Information. It is found that the amount of the FeIII Oh species increases strongly with the Cr content. Also, it decreases weakly with the ratio Fe/(Ni + Fe) for a constant amount of Cr.
Ratio of the amounts of the two Fe species FeIII Oh and FeIII Td plotted over (a) the Cr content, (b) Fe/(Fe + Ni) content as measured by EDX. Spectra for these species are shown in Fig. 6. The color code denotes the compositional ratio of Fe/(Fe + Ni). There is a stronger correlation of the ratio FeIII Oh/(FeIII Oh + FeIII Td) to the Cr content than to the ratio Fe/(Fe + Ni).
Discussion
Ni was found to be in the NiII state and no clear increase of the OER activity with the Ni content was observed. It was previously shown9 that NiO (containing exclusively NiII species) exhibited a relatively high overpotential as compared to LaFeO3 and SrFeO3 suggesting that NiII is not forming the active center for OER catalysis.
The increase of OER activity with Cr content, while only CrIII is present, is either caused by Cr being the active species or a positive effect on Fe or Ni and one of those being the active species. We consider it unlikely that CrIII is the active site, since six 3d electrons as found in CrIII, result in a too strong Cr-O bond and hence should lead to a low OER activity. The situation may be different in CrVI, but different from Singh et al.5 we did not observe the presence of CrVI.
As shown in Figs 2 and 7, the ratio FeIII Oh/(FeIII Td + FeIII Oh) increases with the amount of Cr, similarly to the current density at an overpotential of 320 mV. This either indicates that the presence of FeIII Td has a negative effect on the OER activity or that FeIII Oh has a positive effect, or both.
We exclude FeIII Td being the active site for two reasons, namely the correlation between the amount of FeIII Td and the OER activity and the electronic structure of FeIII in a Td symmetric ligand environment (LS), which is  . The high eg electron count results in a too weak Fe-O bond, making FeIII Td supposedly relatively inactive for the OER.
. The high eg electron count results in a too weak Fe-O bond, making FeIII Td supposedly relatively inactive for the OER.
Since the observed OER current increases with FeIII Oh this species is a candidate for being involved in the active site. Also the electronic configuration of FeIII Oh (LS),  , is very different from the FeIII Td case. One example is the semiconductor Fe2O3, which is often used in solar water splitting despite its very low charge carrier lifetime. It is then usually combined with an OER catalysts, due to its relatively low OER activity26,27.
, is very different from the FeIII Td case. One example is the semiconductor Fe2O3, which is often used in solar water splitting despite its very low charge carrier lifetime. It is then usually combined with an OER catalysts, due to its relatively low OER activity26,27.
Conclusion
In the material system Ni1-y-zFeyCrzOx an increase in OER activity with the introduction of Cr was observed using a SDC. Tafel slopes were found to be in the range of 70 to 115 mV/decade with a relatively constant slope of 80 mV/decade for Fe concentrations above 30%. This increase in activity was investigated by XAS of the transition metal L-edges. Ni and Cr were found to be in the NiII and CrIII states throughout the whole composition range with electronic structures similar to NiO and Cr2FeO4. Fe on the contrary showed variations with the elemental compositions, which were assigned to two species FeIII Oh and FeIII Td. The amount of FeIII Oh relative to the total amount of Fe was found to increase with Cr content. Since the OER activity also increases with the amount of Cr, either FeIII Td has a negative effect on the OER activity or FeIII Oh has a positive effect.
Methods
Sample Preparation
Thin film materials libraries were deposited in a combinatorial reactive magnetron sputtering system (AJA International ATC-2200 V) as described in ref. 28. The substrate used was a 7.5 × 7.5 cm FTO coated glass substrate. The pressure prior to sputtering was below 0.66·10−8 Pa. The reactive atmosphere of Ar/O2 was kept at a constant pressure of 0.66 Pa over the entire deposition with an Ar flow of 6.66·10−7 m3s−1 and an O2 (6 N purity) flow of 8.33·10−8 m3s−1. In the cylindrical deposition chamber the azimuthal angle was 90°, with the Ni and Fe targets at opposite sides and the Cr target in between them. The deposition of Fe was performed by direct current (DC) magnetron sputtering with a power of 30 W. Cr was sputtered with a radio frequency (RF) source at 280 W. Ni was sputtered using a pulsed direct current (pDC) power source run at 65 W. No intentional substrate heating was performed during deposition. After deposition the materials library was annealed in air at 873 K for 90 min with a heating rate of 16 K/min and a cooling rate of 4 K/min. Two materials libraries were deposited subsequently, one for electrochemical measurements and one for XAS. Elemental composition was determined by EDX using 20 kV acceleration voltage and an INCA x-act detector from Oxford mounted to a Jeol 5800 SEM.
Electrochemical Measurements
The electrochemical high-throughput characterization of one materials library was performed using an automated SDC as described in refs 29, 30, 31. The SDC contained a double junction reference electrode (Ag/AgCl/3M KCl/0.1M KOH) with a potential of +239 mV vs. the normal hydrogen electrode (NHE) and a Pt-wire counter electrode housed in a special PTFE tip that was mounted on a three-axes micropositioning system32. Prior to the electrochemical measurements an electrochemical impedance spectrum (EIS) was acquired. Using this EIS the cell resistance was determined to perform a correction of the ohmic drop. To account for possible instability due to the electrochemical measurements two conditioning curves were performed prior to the actual electrochemical measurement under the exact same conditions. From this there is no obvious corrosion of the sample visible before and after the electrochemical measurement. The potentials were swept at a scan rate of 10 mV/s. All electrochemical measurements were performed in an aqueous solution of oxygen saturated 0.1M KOH. Both half-waves of cyclic voltammogramms were averaged, which allowed minimization of the contribution of capacitive currents. From these mean currents the tafel slopes were determined by fitting a line to the logarithm of the mean current density in the voltage range from 1.50 V to 1.54 V. In this potential range good linearity was observed. All potentials are reported in V vs. the reversible hydrogen electrode (RHE). It should be noted, that there are alternative ways to determine the tafel slope as for example by rotating disk electrodes than the one employed here. It is however believed that the comparability between the tafel slopes presented herein is good since all experiments are performed robotically under the exact same conditions.
X-ray Absorption Spectroscopy
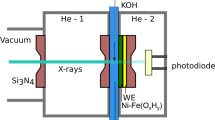
XAS measurements were performed with the LiXEdrom 2.0 endstation at the soft X-ray beamline UE56/2-PGM2 at BESSY II. An automated measurement system was set up to measure over 300 XAS spectra of the Ni, Fe and Cr L-edges in total fluorescence yield mode. Three GaAs photodiodes (Hamamatsu G1127) were employed as detectors. No radiation damage was observed in repeated measurements on the same position on the sample. Spectra were normalized to maximum intensity and a linear background subtraction was performed. The L-edges of Ni, Fe and Cr were energy calibrated with references of NiO22, Fe2O322 and Cr2FeO423.
Additional Information
How to cite this article: Schwanke, C. et al. Correlating Oxygen Evolution Catalysts Activity and Electronic Structure by a High-Throughput Investigation of Ni1-y-zFeyCrzOx. Sci. Rep. 7, 44192; doi: 10.1038/srep44192 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Chen, J. Y. C., Miller, J. T., Gerken, J. B. & Stahl, S. S. Inverse spinel NiFeAlO4 as a highly active oxygen evolution electrocatalyst: promotion of activity by a redox-inert metal ion. Energy Environ. Sci. 7, 1382 (2014).
Gong, M. & Dai, H. A mini review of NiFe-based materials as highly active oxygen evolution reaction electrocatalysts. Nano Res. 8, 23–39 (2014).
Landon, J. et al. Spectroscopic characterization of mixed Fe–Ni oxide electrocatalysts for the oxygen evolution reaction in alkaline electrolytes. ACS Catal. 2, 1793–1801 (2012).
Li, Y.-F. & Selloni, A. Mechanism and activity of water oxidation on selected surfaces of pure and Fe-doped NiOx. ACS Catal. 4, 1148–1153 (2014).
Singh, R. N., Singh, J. P., Lal, B., Thomas, M. J. K. & Bera, S. New NiFe2−xCrxO4 spinel films for O2 evolution in alkaline solutions. Electrochim. Acta 51, 5515–5523 (2006).
Nørskov, J. K., Bligaard, T., Rossmeisl, J. & Christensen, C. H. Towards the computational design of solid catalysts. Nat. Chem. 1, 37–46 (2009).
Greeley, J. & Nørskov, J. K. Combinatorial density functional theory-based screening of surface alloys for the oxygen reduction reaction. J. Phys. Chem. C 113, 4932–4939 (2009).
Nørskov, J. K., Abild-Pedersen, F., Studt, F. & Bligaard, T. Density functional theory in surface chemistry and catalysis. Proc. Natl. Acad. Sci. USA 108, 937–43 (2011).
Man, I. C. et al. Universality in oxygen evolution electrocatalysis on oxide surfaces. ChemCatChem 3, 1159–1165 (2011).
Suntivich, J. et al. Design principles for oxygen-reduction activity on perovskite oxide catalysts for fuel cells and metal-air batteries. Nat. Chem. 3, 546–50 (2011).
Suntivich, J., May, K. J., Gasteiger, H. A., Goodenough, J. B. & Shao-Horn, Y. A perovskite oxide optimized for oxygen evolution catalysis from molecular orbital principles. Science 334, 1383–5 (2011).
Calle-Vallejo, F. et al. Number of outer electrons as descriptor for adsorption processes on transition metals and their oxides. Chem. Sci. 4, 1245 (2013).
Hong, W. T., Welsch, R. E. & Shao-Horn, Y. Descriptors of oxygen-evolution activity for oxides: a statistical evaluation. J. Phys. Chem. C 120, 78–86 (2016).
Louie, M. W. & Bell, A. T. An investigation of thin-film Ni-Fe oxide catalysts for the electrochemical evolution of oxygen. J. Am. Chem. Soc. 135, 12329–37 (2013).
Friebel, D. et al. Identification of highly active Fe sites in (Ni,Fe)OOH for electrocatalytic water splitting. J. Am. Chem. Soc. 137, 1305–1313 (2015).
Bockris, J. O. The Electrocatalysis of Oxygen Evolution on Perovskites. J. Electrochem. Soc. 131, 290 (1984).
Surendranath, Y., Kanan, M. W. & Nocera, D. G. Mechanistic studies of the oxygen evolution reaction by a cobalt-phosphate catalyst at neutral pH. J. Am. Chem. Soc. 132, 16501–9 (2010).
Gerken, J. B. et al. Electrochemical water oxidation with cobalt-based electrocatalysts from pH 0-14: the thermodynamic basis for catalyst structure, stability, and activity. J. Am. Chem. Soc. 133, 14431–42 (2011).
Bockris, J. O. & Otagawa, T. Mechanism of oxygen evolution on perovskites. J. Phys. Chem. 87, 2960–2971 (1983).
Bediako, D. K., Surendranath, Y. & Nocera, D. G. Mechanistic studies of the oxygen evolution reaction mediated by a nickel-borate thin film electrocatalyst. J. Am. Chem. Soc. 135, 3662–74 (2013).
Bard, A. J. & Faulkner, L. R. Electrochemical methods: fundamentals and applications. 6 (2001).
van Elp, J., Searle, B. G., Sawatzky, G. A. & Sacchi, M. Ligand hole induced symmetry mixing of d8 states in LixNi1−xO, as observed in Ni 2p x-ray absorption spectroscopy. Solid State Commun. 80, 67–71 (1991).
Kendelewicz, T. et al. X-ray absorption and photoemission study of the adsorption of aqueous Cr(VI) on single crystal hematite and magnetite surfaces. Surf. Sci. 424, 219–231 (1999).
Miedema, P. S. & de Groot, F. M. F. The iron L edges: Fe 2p X-ray absorption and electron energy loss spectroscopy. J. Electron Spectros. Relat. Phenomena 187, 32–48 (2013).
de Groot, F. M. F. et al. 1s2p resonant inelastic X-ray scattering of iron oxides. J. Phys. Chem. B 109, 20751–62 (2005).
Kay, A., Cesar, I. & Grätzel, M. New benchmark for water photooxidation by nanostructured alpha-Fe2O3 films. J. Am. Chem. Soc. 128, 15714–21 (2006).
Jang, J.-W. et al. Enabling unassisted solar water splitting by iron oxide and silicon. Nat. Commun. 6, 7447 (2015).
Ludwig, A., Zarnetta, R., Hamann, S., Savan, A. & Thienhaus, S. Development of multifunctional thin films using high-throughput experimentation methods. Int. J. Mater. Res. 99, 1144–1149 (2008).
Sliozberg, K., Schäfer, D., Meyer, R., Ludwig, A. & Schuhmann, W. A combinatorial study of photoelectrochemical properties of Fe-W-O thin films. Chempluschem 80, 136–140 (2015).
Meyer, R., Sliozberg, K., Khare, C., Schuhmann, W. & Ludwig, A. High-throughput screening of thin-film semiconductor material libraries II: characterization of Fe-W-O libraries. ChemSusChem 8, 1279–85 (2015).
Sliozberg, K. et al. Fe-cr-al containing oxide semiconductors as potential solar water-splitting materials. ACS Appl. Mater. Interfaces 7, 4883–4889 (2015).
Sliozberg, K. et al. High-throughput screening of thin-film semiconductor material libraries I: system development and case study for Ti-W-O. ChemSusChem 8, 1270–1278 (2015).
Acknowledgements
The authors are grateful to the Deutsche Forschungsgemeinschaft (DFG) in the framework of the SPP 1613 (SCHU929/12-1, 12-2 and LU1175/10-1, 10-2) and to the Helmholtz Association (VH-NG-1140). H.S.S. and K.S. acknowledge a PhD fellowship from the International Max Planck Research School for Surface and Interface Engineering (IMPRS-SurMat). C.S. thanks Fanxing Xi for AFM measurements.
Author information
Authors and Affiliations
Contributions
H.S. prepared the samples, C.S. and L.X. performed XAS measurements, and K.S. performed the electrochemical measurements. C.S. and H.S. analyzed the data and wrote the manuscript, K.M.L supervised the project. All authors revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Schwanke, C., Stein, H., Xi, L. et al. Correlating Oxygen Evolution Catalysts Activity and Electronic Structure by a High-Throughput Investigation of Ni1-y-zFeyCrzOx. Sci Rep 7, 44192 (2017). https://doi.org/10.1038/srep44192
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep44192
- Springer Nature Limited
This article is cited by
-
Tuning coordination environment of iron ions to ensure ultra-high pseudocapacitive capability in iron oxide
Nano Research (2023)
-
Modulation to favorable surface adsorption energy for oxygen evolution reaction intermediates over carbon-tunable alloys towards sustainable hydrogen production
Materials for Renewable and Sustainable Energy (2022)
-
Atomic-level insight into super-efficient electrocatalytic oxygen evolution on iron and vanadium co-doped nickel (oxy)hydroxide
Nature Communications (2018)