Abstract
Growing evidence from recent studies has demonstrated an association between inflammatory bowel disease (IBD) susceptibility and two polymorphisms of DLG5 R30Q (rs1248696) and P1371Q (rs2289310), but the results remain controversial. We conducted a meta-analysis including a total of 22 studies with 10,878 IBD patients and 7917 healthy controls for R30Q and 5277 IBD cases and 4367 controls for P1371Q in order to systematically assess their association with the disease. The results indicated that R30Q was significantly associated with reduced susceptibility to IBD in Europeans by allelic and dominant comparisons, but not in overall population. No significant association was found between R30Q and Crohn’s disease (CD) or ulcerative colitis (UC). P1371Q was associated with increased risk of IBD in Europeans and Americans. On the contrary, a decreased risk of IBD was observed in Asian population for P1371Q. In disease subgroup analysis, we found that P1371Q was also significantly associated with CD, but this relationship was not present for UC. In conclusion, our results strongly suggest that the both polymorphisms of DLG5 are correlated with IBD susceptibility in an ethnic-specific manner. Additional well-designed studies with large and diverse cohorts are needed to further strengthen our findings.
Similar content being viewed by others
Introduction
Inflammatory bowel disease (IBD) is a relapsing and chronic inflammatory disorder mainly affecting the small intestine and colon1. Crohn’s disease (CD) and ulcerative colitis (UC) are two major types of IBD. A large number of studies have demonstrated that IBD is caused by multiple reasons including genetic, infectious, environmental and immunoregulatory factors2,3,4. Recently, genome-wide association studies (GWAS) and epidemiological studies have revealed that a considerable number of gene variations, such as in nucleotide oligomerization domain 2 (NOD2)5,6, autophagy related 16-like 1(ATG16L1)7, signal transducer and activator of transcription 3 (STAT3)8,9, immunity-related GTPase family M (IRGM)10 and interleukin 23 receptor (IL23R)11,12, are closely related to IBD susceptibility.
Discs large homolog 5 (DLG5) gene, a member of the membrane associated guanylate kinase (MAGUK) family, encodes a cell scaffolding protein, which is wildly expressed in human tissues including liver, heart, pancreas, small intestine and colon13,14. It has been reported that DLG5 participates in various physiological functions such as cell growth, polarity, intracellular signal transduction and the maintenance of epithelial cell integrity13,15,16. In 2004, Stoll and colleagues initially identified several polymorphisms in DLG5 relating to IBD in a German cohort16. In recent years, two of the variants R30Q (rs1248696, G113A), which changes amino acid 30 from arginine (G allele) to glutamine (A allele), and P1371Q (rs2289310, C4136A), which leads to the change of amino acid 1371 from proline (C allele) to glutamine (A allele), have been studied in different populations17,18,19. However, the results remain inconsistent among studies. Since Stoll et al. reported that the both variants of DLG5 were associated with increased risk for IBD, additional studies replicated the association in different populations16,20,21, but the trend was not further verified by other groups with samples from multiple countries22,23,24,25,26,27,28,29.
Although a meta-analysis performed by Browning et al.30 attempted to assess the relationship between R30Q and IBD in 2007, they did not observe any significant association. Thus, the conclusion is still doubtful. In addition, there is no meta-analysis investigating the association between P1371Q and IBD up to now. It is necessary to perform a comprehensive study to summarize the overall results of these inconsistent publications. Therefore, we conducted the present meta-analysis to evaluate the potential effects of the two DLG5 polymorphisms on IBD susceptibility.
Results
Study characteristics
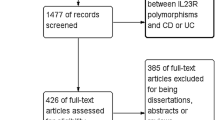
Based on our search strategy, 80 articles were retrieved. According to the inclusion and exclusion criteria, 22 articles16,17,18,19,20,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38 with 31 case-control studies were included in this meta-analysis, and 58 studies were excluded for the reason that they were reviews, meeting reports or had no relation to DLG5 polymorphisms. Specially, for R30Q, two cohorts with detailed allelic and genotype distribution data in the studies of Medici et al.25 and Pearce et al.29 were derived from previous publications by Stoll et al.16 and Daly et al.21, respectively. Hence, we included the latest and more specific studies of Medici et al.25 and Pearce et al.29 in this meta-analysis. The flow chart of the study selection is shown in Fig. 1. Among the 22 eligible studies, 12 investigated the correlation between R30Q and IBD susceptibility, four researched P1371Q and IBD risk, and six studied both R30Q and P1371Q with IBD risk. Finally, a total of 10,878 IBD patients (including 6223 CD and 4551 UC cases) and 7917 controls for R30Q and 5277 IBD patients (including 2745 CD and 1699 UC cases) and 4367 controls for P1371Q were included in the present meta-analysis. The characteristics of the eligible studies are summarized in supplementary Table S1. The allele and genotype distributions of R30Q and P1371Q are listed in supplementary Tables S2 and S3, respectively.
R30Q polymorphism and IBD susceptibility
The meta-analysis of R30Q was based on a total of 18 articles including 10,878 IBD patients and 7917 controls. Statistical heterogeneity was present in allelic model (I2 = 36%, P = 0.050), heterozygote model (I2 = 49%, P = 0.009) and dominant model (I2 = 46%, P = 0.020). Therefore, random effects model was applied in these analyses. No significant association was identified between R30Q and IBD in overall population (A vs. G: OR = 0.94, 95%CI: 0.86–1.03, P = 0.17; GA vs. GG: OR = 0.93, 95%CI: 0.83–1.05, P = 0.25; AA vs. GG: OR = 0.88, 95%CI: 0.65–1.18, P = 0.38; AA + GA vs. GG: OR = 0.93, 95%CI: 0.83–1.04, P = 0.22; AA vs. GA + GG: OR = 0.89, 95%CI: 0.66–1.20, P = 0.44). Nevertheless, a trend of reduced risk could be seen (Table 1).
Intriguingly, subgroup analysis based on ethnicity disclosed a significant correlation between R30Q and IBD susceptibility in Europeans in allelic model (A vs. G: OR = 0.91, 95%CI: 0.85–0.98, P = 0.02) and dominant model (AA + GA vs. GG: OR = 0.91, 95%CI: 0.84–0.99, P = 0.03) by fixed-effect model because no significant statistical heterogeneity was identified (Table 1 and Fig. 2). In order to detect the robustness of the effects, random effects model was used to further analyze the correlation. The results showed that there was still a significant association between R30Q and IBD in Europeans by random effects model (A vs. G: OR = 0.91, 95%CI: 0.83–0.99, P = 0.03; AA + GA vs. GG: OR = 0.90, 95%CI: 0.81–0.99, P = 0.04). However, no association was observed for the other three genetic models. As for the American and Australasian populations, no significant correlation was found in either cohort (data not shown).
Subsequently, the relationships between R30Q and two major types of IBD (UC and CD) were analyzed. However, no modifying effect of R30Q on the risk of UC or CD was discovered in any genetic model (Table 1).
P1371Q polymorphism and IBD susceptibility
Ten studies with eleven cohorts including 4985 IBD participants and 4097 controls were utilized to analyze the correlation between P1371Q polymorphism and the risk of IBD. Random effects model was used in the allelic model, homozygote model, dominant model and recessive model due to the presence of heterogeneity, while fixed-effect model was used in heterozygote model. The results showed that P1371Q was not significantly related to IBD in overall population (Table 2). Subgroup analysis indicated an increased risk of IBD in Americans by allele comparison (A vs. C: OR = 1.48, 95%CI: 1.12–1.95, P = 0.01), and in Europeans in homozygote comparison (AA vs. CC: OR = 4.06, 95%CI: 1.15–14.29, P = 0.03) and recessive comparison (AA vs. CA + CC: OR = 4.05, 95%CI: 1.15–14.26, P = 0.03) using fixed-effect model. However, we observed an opposite relationship between P1371Q and IBD in Asian cohort (CA vs. CC: OR = 0.72, 95%CI: 0.54–0.96, P = 0.02) (Table 2). Considering the limited number of studies and heterogeneity, these results should be treated with caution.
Next, the analyses were carried out according to disease type. There were nine studies with 2745 patients and 4097 controls involved in the CD meta-analysis. Interestingly, we detected a significant association between P1371Q and CD in overall population (heterozygote model CA vs. CC: OR = 0.81, 95%CI: 0.68–0.97, P = 0.02; dominant model AA + CA vs. CC: OR = 0.80, 95%CI: 0.68–0.95, P = 0.01) by fixed-effect model (Table 2 and Fig. 3). Likewise, the reliability of these results was demonstrated by random effects model (CA vs. CC: OR = 0.81, 95%CI: 0.68–0.97, P = 0.02; AA + CA vs. CC: OR = 0.80, 95%CI: 0.64–0.99, P = 0.04). In addition, a significant statistical association between P1371Q and CD in Asian was identified by heterozygote comparison (CA vs. CC: OR = 0.72, 95%CI: 0.54–0.96, P = 0.02). However, no significant association was found in Europeans or Americans (Table 2). For the other major type of IBD, the results did not indicate any statistical association between P1371Q and UC in the overall population, or in Europeans, Americans or Asians for any of the genotype comparisons (Table 2).
Sensitivity analysis and publication bias
In order to examine the influence set by the two studies23,37 that did not comply with Hardy-Weinberg equilibrium (HWE) on the pooled effects, sensitivity analysis was performed by omitting them in all genetic models. The results showed that deleting the study of Torok et al.23 did not materially alter the pooled ORs of R30Q (data not shown). However, the significant effects of P1371Q on CD and IBD disappeared in overall and Asian populations by excluding Chua et al.37 (data not shown). Egger’s regression test was used to evaluate the publication bias. No significant publication bias was identified in overall comparisons (Tables 1 and 2).
Discussion
The present meta-analysis examined the association between two crucial polymorphisms of DLG5 and IBD risk. We uncovered a positive relationship between R30Q and IBD susceptibility in the European subset of cohorts in allelic model and dominant model, which indicated that carriers of the A allele have a lower risk of developing IBD. Moreover, the robustness of the results was also verified by random effects model and a sensitivity analysis by omitting the study derived from HWE. However, no significant correlation was identified in the overall population. It is possible that the presence of allelic heterogeneity among different populations decreased the pooled effects. In addition, subgroup analysis based on disease type did not find a significant relationship between R30Q and CD or UC for the possible reason that stratification reduced the sample size, so the statistics power was insufficient to detect a weak association. As for P1371Q, we demonstrated that this polymorphism increased the risk of IBD in Americans and Europeans, but not in the overall population. On the contrary, P1371Q was associated with decreased risk of IBD in Asians. However, this finding should be interpreted with caution due to the limited number of studies and small sample size. Moreover, in the CD subgroup analysis, we uncovered that P1371Q was significantly related to a lower risk of CD in overall population and Asian ethnicity. But P1371Q did not modify UC risk in all cohorts.
A previous meta-analysis including 13 studies was performed to analyze the association between R30Q polymorphism and IBD risk in Caucasians30. However, that study failed to find any evidence for an association. Meanwhile, a case-control study carried out by the same authors found that neither R30Q nor P1371Q was associated with CD, UC or IBD in New Zealand Caucasian population. The main reason for this negative result may be due to the relatively small sample size and lack of deep and complete analysis. Another study focused on the association of R30Q with CD via stratifying the Caucasian population by sex from 12 case-control studies39. The authors demonstrated that R30Q was marginally associated with decreased risk for CD in women (P = 0.049). Nevertheless, R30Q was not significantly related to CD susceptibility in men (P = 0.058). In summary, they provided an evidence of a trend toward reduced CD risk in both males and females. This finding is consistent with our subgroup analysis of CD in European cohort, mainly from Caucasian. Compared to the above two publications, our meta-analysis summarized 22 eligible case-control studies with a larger sample size and offered convincing evidence by systematically analyzing the association of IBD susceptibility with two DLG5 polymorphisms, including not only the most studied variant, R30Q, but also another significant variant, P1371Q.
As a chronic and relapsing inflammatory disease, a growing number of studies have shown that IBD is associated with both genetic and environmental factors40,41. Recently, many single nucleotide polymorphisms contributing to the susceptibility of IBD have been identified by genome-wide association study (GWAS) including variants in DLG511,42,43. DLG5 encodes a cell scaffolding proteins that has been found to play an important role in the determination and maintenance of intestinal epithelial cells polarity44. Moreover, DLG5 participates in epithelial cell proliferation, migration and adhesion45. The DLG5 protein contains four domain types, including one DUF622 domain, one SH3 domain, one GK domain and four PDZ domains46. The protein–protein interaction domains support DLG5 as a multifunctional adapter and scaffolding protein. Based on observations from in silico analysis, both the R30Q and P1371Q variants probably influence or change the structure and function of DLG516. Considering the role of DLG5 in maintaining intestinal barrier integrity47, DLG5 mutations might interfere with the epithelial barrier function of the intestine, and therefore be implicated in IBD pathogenesis.
In 2004, Stoll et al.16 initially reported that four DLG5 polymorphisms were associated with IBD in a European cohort, and among them, R30Q was strongly related to an increased risk for IBD. Subsequently, several studies observed similar results21,27,48. However, further studies with samples from different countries attempting to replicate the potential association between R30Q and IBD susceptibility have not been generally successful22,23,24,26,28,33,36. One earlier meta-analysis performed by Browning et al. summarized R30Q data from published studies and attempted to resolve this discrepancy but failed30. In 2008, Browning and colleagues carried out another study by gender-stratified analysis of R30Q in a relatively large cohort39. Even so, only a weak relationship was found in women. Simultaneously, the association between P1371Q and IBD has been replicated in only two studies20,27, and the results of many more studies were inconsistent18,26,28,30,37,38. Since there were so many discrepant results, it was necessary to perform a meta-analysis to better understand the correlation between the two polymorphisms of DLG5 and IBD susceptibility.
There were several strengths of our meta-analysis. First of all, we performed the present study based on large sample sizes including a total number of 10878 IBD patients with 7917 controls on R30Q polymorphism and 5277 IBD patients with 4367 controls on P1371Q polymorphism, which helped to add convincing evidence. Compared with previous meta-analyses, more studies were included. Additionally, subgroup analysis, sensitivity analysis and genotype comparison were also carried out. Furthermore, we systematically investigated the relationship between P1371Q and IBD risk. To the best of our knowledge, this is the first meta-analysis regarding P1371Q and IBD. Moreover, we set and implemented strict inclusion and exclusion criteria to guarantee the quality of the included studies and the reliability of the results. Finally, selection bias was well-controlled via literature search, and no publication bias was identified by Egger’s test.
Nevertheless, the present meta-analysis has several limitations that should be considered. Firstly, potential heterogeneity observed among the studies of both R30Q and P1371Q, which could not be effectively reduced via subgroup and sensitivity analysis, might partially influence the results. Hence, some other details are needed to analyze the sources of the heterogeneity. For instance, sample size, age of patients and genotyping methods might contribute to the heterogeneity. Secondly, there were two studies with genotypes deprived from HWE, of which one was for R30Q and the other for P1371Q. Although the pooled ORs were not significantly influenced by this study for R30Q, the significant association between P1371Q and CD was diminished by omitting the study for P1371Q. Thirdly, although we found that individuals with P1371Q variant have lower risk of suffering IBD in Asian population, the number of eligible studies and patients was limited. Therefore, many more high quality case-control studies are needed to further explore the association between P1371Q and IBD susceptibility in Asian cohorts.
In summary, despite the limitations above, our results suggest that the DLG5 R30Q polymorphism is associated with a reduced risk of IBD in Europeans, while P1371Q is associated with an increased risk in Europeans and Americans, but could serve as a protective factor in Asian population. Particularly, P1371Q may decrease the risk of CD in overall population. However, caution should be taken for P1371Q because of insufficient data and population heterogeneity, which might have impact on the convincingness of the results. Well-designed case-control studies with larger sample sizes and multiple ethnicities are required to strengthen the results of the current study. More importantly, studies focusing on the biological functions and mechanisms involving the IBD pathogenesis of DLG5 are also urgently needed.
Materials and Methods
Literature search
Two independent investigators conducted a literature search for relevant studies evaluating the relationship between DLG5 polymorphisms (R30Q and P1371Q) and IBD risk in the Pubmed and Web of Science up to March 14, 2016. The following search terms were used: (inflammatory bowel disease OR IBD OR ulcerative colitis OR UC OR Crohn’s disease OR CD) and (genetic polymorphism OR polymorphism OR variant) and (DLG5 OR discs large homolog 5), without any limitation. Manual search was also performed to obtain additional publications through the reference lists of retrieved studies and reviews.
Inclusion and exclusion criteria
Studies meeting the following criteria were included: (1) cases-control study; (2) evaluation the association between DLG5 R30Q or P1371Q polymorphism and IBD risk; (3) provided detail genotype or allelic distribution; (4) contained sufficient data to calculate odds ratios (ORs) and 95% confidence intervals (CIs). Exclusion criteria: (1) reviews, meeting articles, laboratory studies or duplication of previous publications; (2) studies without detail genotype data; (3) researches did not focus on DLG5 R30Q or P1371Q polymorphism; (4) family-based studies. When there were several articles about a study or the same cohort included in two or more publications were identified, only the latest or complete study was selected.
Data extraction
The following information was independently extracted by two authors from eligible studies: name of the first author, year of publication, country and ethnicity of the study population, genotyping method, number of cases and controls, allele and genotype frequency in cases and controls. Any disagreement was resolved by discussing with a third author to reach a consensus.
Statistical analysis
The present meta-analysis was conducted according to the PRISMA checklists and following the guideline49. ORs and 95%CI were calculated to measure the strength of association between DLG5 R30Q and P1371Q variants and IBD risk. Meta-analyses were performed by allelic model (R30Q: A vs. G, P1371Q: A vs. C), heterozygote model (R30Q: GA vs. GG, P1371Q: CA vs. CC), homozygote model (R30Q: AA vs. GG, P1371Q: AA vs. CC), dominant model (R30Q: GA + AA vs. GG, P1371Q: CA + AA vs. CC), recessive model (R30Q: AA vs. GC + GG, P1371Q: AA vs. CA + CC). Cochran’s Q statistic and I2 test were performed to evaluate the heterogeneity among the eligible studies. Statistical heterogeneity was defined as P < 0.1 or I2 > 50%. Random effects models or fixed-effect model was used to pool the effect size depending on the existence of statistical heterogeneity or not, respectively. Chi-square test was applied to evaluate the Hardy-Weinberg equilibrium (HWE) in control group. Publication bias was detected by Egger’s test. All the P value were two-sided and P < 0.05 was considered statistically significant. All statistical analyses were performed by Review Manager software version 5.2 and Stata software version 12.0 (StataCorp, College Station, TX, USA).
Additional Information
How to cite this article: Li, Y. et al. Meta-analysis of associations between DLG5 R30Q and P1371Q polymorphisms and susceptibility to inflammatory bowel disease. Sci. Rep. 6, 33550; doi: 10.1038/srep33550 (2016).
References
Baumgart, D. C. & Sandborn, W. J. Inflammatory bowel disease: clinical aspects and established and evolving therapies. Lancet 369, 1641–1657 (2007).
Budarf, M. L., Labbe, C., David, G. & Rioux, J. D. GWA studies: rewriting the story of IBD. Trends Genet 25, 137–146 (2009).
Danese, S. et al. Platelets: new players in the mucosal scenario of inflammatory bowel disease. Eur Rev Med Pharmacol Sci 8, 193–198 (2004).
Cho, J. H. Inflammatory bowel disease: Genetic and epidemiologic considerations. World Journal of Gastroenterology 14, 338–347 (2008).
Ogura, Y. et al. A frameshift mutation in NOD2 associated with susceptibility to Crohn’s disease. Nature 411, 603–606 (2001).
Ogura, Y. et al. Genetic variation and activity of mouse Nod2, a susceptibility gene for Crohn’s disease. Genomics 81, 369–377 (2003).
Hampe, J. et al. A genome-wide association scan of nonsynonymous SNPs identifies a susceptibility variant for Crohn disease in ATG16L1. Nat Genet 39, 207–211 (2007).
Polgar, N. et al. Investigation of JAK2, STAT3 and CCR6 polymorphisms and their gene-gene interactions in inflammatory bowel disease. Int J Immunogenet 39, 247–252 (2012).
Cenit, M. C. et al. STAT3 locus in inflammatory bowel disease and multiple sclerosis susceptibility. Genes Immun 11, 264–268 (2010).
Parkes, M. et al. Sequence variants in the autophagy gene IRGM and multiple other replicating loci contribute to Crohn’s disease susceptibility. Nat Genet 39, 830–832 (2007).
Duerr, R. H. et al. A genome-wide association study identifies IL23R as an inflammatory bowel disease gene. Science 314, 1461–1463 (2006).
Grigoras, C. A., Ziakas, P. D., Jayamani, E. & Mylonakis, E. ATG16L1 and IL23R variants and genetic susceptibility to crohn’s disease: mode of inheritance based on meta-analysis of genetic association studies. Inflamm Bowel Dis 21, 768–776 (2015).
Humbert, P., Russell, S. & Richardson, H. Dlg, Scribble and Lgl in cell polarity, cell proliferation and cancer. Bioessays 25, 542–553 (2003).
Shah, G. et al. The cloning, genomic organization and tissue expression profile of the human DLG5 gene: Correction. BMC Genomics 3, 14 (2002).
Wakabayashi, M. et al. Interaction of lp-dlg/KIAA0583, a membrane-associated guanylate kinase family protein, with vinexin and beta-catenin at sites of cell-cell contact. J Biol Chem 278, 21709–21714 (2003).
Stoll, M. et al. Genetic variation in DLG5 is associated with inflammatory bowel disease. Nature Genetics 36, 476–480 (2004).
Dema, B. et al. The R30Q DLG5 variant is not associated with celiac disease or inflammatory bowel disease in the Spanish population. Tissue Antigens 77, 62–64 (2011).
Wagner, J. et al. Interaction of Crohn’s Disease Susceptibility Genes in an Australian Paediatric Cohort. Plos One 5 (2010).
Lin, Z. et al. Genetic association of DLG5 R30Q with familial and sporadic inflammatory bowel disease in men. Dis Markers 27, 193–201 (2009).
Lin, Z. et al. DLG5 P1371Q is associated with inflammatory bowel disease and complementary to R30Q in disease susceptibility. Swiss Med Wkly 141, w13290 (2011).
Daly, M. J. et al. Association of DLG5 R30Q variant with inflammatory bowel disease. Eur J Hum Genet 13, 835–839 (2005).
Noble, C. L. et al. DLG5 variants do not influence susceptibility to inflammatory bowel disease in the Scottish population. Gut 54, 1416–1420 (2005).
Torok, H. P. et al. Polymorphisms in the DLG5 and OCTN cation transporter genes in Crohn’s disease. Gut 54, 1421–1427 (2005).
Lakatos, P. L. et al. DLG5 R30Q is not associated with IBD in Hungarian IBD patients but predicts clinical response to steroids in Crohn’s disease. Inflammatory Bowel Diseases 12, 362–368 (2006).
Medici, V. et al. Extreme heterogeneity in CARD15 and DLG5 Crohn disease-associated polymorphisms between German and Norwegian populations. Eur J Hum Genet 14, 459–468 (2006).
Buning, C. et al. DLG5 variants in inflammatory bowel disease. American Journal of Gastroenterology 101, 786–792 (2006).
Newman, W. G. et al. DLG5 variants contribute to Crohn disease risk in a Canadian population. Human Mutation 27, 353–358 (2006).
Tremelling, M., Waller, S., Bredin, F., Greenfield, S. & Parkes, M. Genetic variants in TNF-alpha but not DLG5 are associated with inflammatory bowel disease in a large United Kingdom cohort. Inflamm Bowel Dis 12, 178–184 (2006).
Pearce, A. V. et al. Investigation of association of the DLG5 gene with phenotypes of inflammatory bowel disease in the British population. Int J Colorectal Dis 22, 419–424 (2007).
Browning, B. L. et al. Association of DLG5 variants with inflammatory bowel disease in the New Zealand Caucasian population and meta-analysis of the DLG5 R30Q variant. Inflamm Bowel Dis 13, 1069–1076 (2007).
Cucchiara, S. et al. Role of CARD15, DLG5 and OCTN genes polymorphisms in children with inflammatory bowel diseases. World J Gastroenterol 13, 1221–1229 (2007).
de Ridder, L. et al. Genetic susceptibility has a more important role in pediatric-onset Crohn’s disease than in adult-onset Crohn’s disease. Inflammatory Bowel Diseases 13, 1083–1092 (2007).
Ferraris, A. et al. Relationship between CARD15, SLC22A4/5, and DLG5 polymorphisms and early-onset inflammatory bowel diseases: an Italian multicentric study. Inflamm Bowel Dis 12, 355–361 (2006).
Gaj, P., Habior, A., Mikula, M. & Ostrowski, J. Lack of evidence for association of primary sclerosing cholangitis and primary biliary cirrhosis with risk alleles for Crohn’s disease in Polish patients. BMC Med Genet 9, 81 (2008).
Karlsen, T. H. et al. Genetic polymorphisms associated with inflammatory bowel disease do not confer risk for primary sclerosing cholangitis. Am J Gastroenterol 102, 115–121 (2007).
Vermeire, S. et al. Association of organic cation transporter perianal penetrating Crohn’s disease but susceptibility to IBD. Gastroenterology 129, 1845–1853 (2005).
Chua, K. H. et al. Identification of DLG5 and SLC22A5 gene polymorphisms in Malaysian patients with Crohn’s disease. J Dig Dis 12, 459–466 (2011).
Yamazaki, K. et al. Association analysis of SLC22A4, SLC22A5 and DLG5 in Japanese patients with Crohn disease. J Hum Genet 49, 664–668 (2004).
Browning, B. L. et al. Gender-stratified analysis of DLG5 R30Q in 4707 patients with Crohn disease and 4973 controls from 12 Caucasian cohorts. Journal of Medical Genetics 45, 36–42 (2008).
Loftus, E. V., Jr. Clinical epidemiology of inflammatory bowel disease: Incidence, prevalence, and environmental influences. Gastroenterology 126, 1504–1517 (2004).
Latella, G. & Papi, C. Crucial steps in the natural history of inflammatory bowel disease. World J Gastroenterol 18, 3790–3799 (2012).
Franke, A. et al. Genome-wide meta-analysis increases to 71 the number of confirmed Crohn’s disease susceptibility loci. Nat Genet 42, 1118–1125 (2010).
Anderson, C. A. et al. Meta-analysis identifies 29 additional ulcerative colitis risk loci, increasing the number of confirmed associations to 47. Nat Genet 43, 246–252 (2011).
Peeters, M. et al. Clustering of increased small intestinal permeability in families with Crohn’s disease. Gastroenterology 113, 802–807 (1997).
Liu, J., Li, J., Ren, Y. & Liu, P. DLG5 in cell polarity maintenance and cancer development. Int J Biol Sci 10, 543–549 (2014).
Shah, G. et al. The cloning, genomic organization and tissue expression profile of the human DLG5 gene. BMC Genomics 3, 6 (2002).
Festen, E. A., Zhernakova, A., Wijmenga, C. & Weersma, R. K. Association of DLG5 variants with gluten-sensitive enteropathy. Gut 57, 1027–1028 (2008).
Friedrichs, F. et al. Evidence of transmission ratio distortion of DLG5 R30Q variant in general and implication of an association with Crohn disease in men. Hum Genet 119, 305–311 (2006).
Moher, D., Liberati, A., Tetzlaff, J. & Altman, D. G. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg 8, 336–41 (2010).
Acknowledgements
This study was supported by National Natural Science Foundation of China (NO. 81472475 and NO. 81102007), and Scientific Research Foundation of Chongqing Medical University (NO. 201408).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: G.R., H.L. and Y.L. Performed the experiments: Y.L., P.C. and J.S. Analyzed the data: Y.L., H.T., P.C. and J.S. Contributed reagents/materials/analysis tools: Y.L., J.H., H.L. and L.L. Wrote the paper: Y.L., P.C. and H.L.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Li, Y., Chen, P., Sun, J. et al. Meta-analysis of associations between DLG5 R30Q and P1371Q polymorphisms and susceptibility to inflammatory bowel disease. Sci Rep 6, 33550 (2016). https://doi.org/10.1038/srep33550
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep33550
- Springer Nature Limited