Abstract
Testicular Leydig cells contain abundant cytoplasmic lipid droplets (LDs) as a cholesteryl-ester store for releasing cholesterols as the precursor substrate for testosterone biosynthesis. Here, we identified the protein composition of testicular LDs purified from adult mice by using mass spectrometry and immunodetection. Among 337 proteins identified, 144 were previously detected in LD proteomes; 44 were confirmed by microscopy. Testicular LDs contained multiple Rab GTPases, chaperones and proteins involved in glucuronidation, ubiquination and transport, many known to modulate LD formation and LD-related cellular functions. In particular, testicular LDs contained many members of both the perilipin family and classical lipase/esterase superfamily assembled predominately in adipocyte LDs. Thus, testicular LDs might be regulated similar to adipocyte LDs. Remarkably, testicular LDs contained a large number of classical enzymes for biosynthesis and metabolism of cholesterol and hormonal steroids, so steroidogenic reactions might occur on testicular LDs or the steroidogenic enzymes and products could be transferred through testicular LDs. These characteristics differ from the LDs in most other types of cells, so testicular LDs could be an active organelle functionally involved in steroidogenesis.
Similar content being viewed by others
Introduction
The testis consists of three major cell types: germ cells, Sertoli supporting cells within seminiferous tubules and Leydig cells in the interstitium between the tubules. Leydig cells are particularly enriched with endoplasmic reticulum (ER), mitochondria and cytoplasmic lipid droplets (LDs)1,2. This structure is associated with the androgen production function of Leydig cells.
Testosterone biosynthetic enzymes are generally located in the ER and mitochondrial membranes and the adjacent cytoplasm. The precursor substrate for steroidogenesis is cholesterol. An individual Leydig cell could secrete 20 ng of testosterone daily in humans3 and 0.5 ng in adult rodents2. To ensure such a high rate of steroidogenesis, the testis utilizes endogenous cholesterols de novo synthesized in situ rather than transported from the plasma4,5. The intracellular LDs of Leydig cells contain a large pool of cholesteryl ester that can be broken down into free cholesterol on demand for steroidogenesis5. In response to the varied androgen production during pubertal growth6 and breeding1, the size and number of LDs in Leydig cells may vary greatly, which reflects an altered demand for stored cholesterol-cholesteryl ester for testosterone biosynthesis1,6. Also, Sertoli cells contain a fair amount of small LDs that show cyclic variations throughout the spermatogenic cycle in rat7 and human8 and can transfer from Sertoli cells to spermatocytes8. Therefore, testicular LDs play functional roles in testes.
The LDs in all eukaryotes contain a core of neutral lipids, a monolayer surface of phospholipids and a number of proteins that are embedded in the surface9. In contrast to biochemically inert neutral lipids, the protein components on the LD surface are biologically active and control LD storage and hydrolysis and LD-related cellular functions. A considerable number of LD proteins have been identified in many types of cells by immunodetection or proteomic approaches. The investigation of these LD proteins has greatly extended our understanding of the properties and functions of LDs in given cells.
The LDs in testicular cells are particularly small, with mean diameter 1 μm2 and thus are not easily detected by common immunodetection approaches. Only a few LD-associated proteins have been identified in testicular cells. This insufficient information has long restricted the investigation of functional roles of testicular LDs.
This proteomic study aimed to identify protein components of testicular LDs of adult mice. We detected 337 proteins from testicular LD preparations; 144 were prevously detected in LD proteomes and 44 were previously verified in LDs by microscopy. Testicular LDs contained almost complete sets of LD-related protein members of both the perilipin (Plin) family and lipase/esterase superfamily that assemble predominantly in adipocyte LDs and contain many enzymes that govern biosynthesis of sterols and hormonal steroids. These distinct characteristics are different from the LDs in most other cells. Testicular LDs are a unique, biologically active cellular organelle that might be regulated like adipocyte LDs and play important roles in the biosynthesis and metabolism of hormonal steroids.
Methods and Materials
Animals and antibodies
Polyclonal antibodies against Plin1~4 and hormone-sensitive lipase (HSL) were from C. Londos (US National Institutes of Health). Other antibodies were from Abcam, Cell Signaling, or Santa Cruz Biotechnology. The animal study was performed in accordance with the NIH guidelines for the care and use of laboratory animals and was approved by the animal care and utilization committee of Peking University Health Science Center.
Purification of the LDs from mice testis
For each individual preparation, 20 testes obtained from 10-week-old C57BL/6 mice were used. LDs were purified by the protocol we developed recently10. Manipulations were performed at 4 °C or on ice, if required. After removal of blood vessels and connective tissues, 20 testes were grouped and homogenized by use of a Dounce glass homogenizer containing 10 ml buffer A (250 mM sucrose, 0.2 mM phenylmethylsulfonyl fluoride, 25 mM tricine, pH 7.6) by 20 strokes with a loose-fitting pestle and 40 strokes with a tight-fitting pestle. The homogenate was disrupted for 15 min at 750 psi in a nitrogen bomb chamber and cleaned by centrifugation at 3000 × g. The post-nuclear supernatant was transferred to a SW40 tube, then buffer B (20 mM HEPES, pH 7.4, 100 mM KCl and 2 mM MgCl2) was loaded on top of the supernatant. After centrifugation at 38,000 × g for 1 h, a white LD layer appeared on the top of the tube. The membrane was pelleted at the bottom and the infranatant was the cytosolic fraction. All 3 fractions were collected. The LD fraction was transferred to a new tube and centrifuged for 4 min at 14,000 × g. After removal of the underlying liquid, LDs were washed 3 times, each with 200 μl buffer B and centrifuged at 14,000 × g for 4 min. The LD fraction on the top was collected.
Protein in-gel digestion and mass spectrometry analysis
Manipulations were performed as we reported recently11. Protein components in the LD preparation were precipitated with 100% acetone. Proteins were separated by 10% SDS-PAGE followed by Coomassie Blue or silver staining. For the total proteome, a full lane of Coomassie Blue-stained gel was cut into 23 slices from high to low molecular weight. Each slice was further cut into smaller pieces, destained, washed, dehydrated and vacuum-dried. Proteins in slices were reduced with 10 mM dithiothreitol for 1 h at 56 °C and alkylated with 55 mM iodoacetamide for 45 min. Gel slices were washed with 25 mM ammonium bicarbonate, acetonitrile and vacuum-dried. For in-gel digestion, slices were incubated with 10 ng/μl trypsin in 25 mM ammonium bicarbonate solution. The digestion reaction proceeded at 37 °C overnight and was stopped by adding 5% formic acid to adjust pH to <4.0. After two extractions with 60% acetonitrile, the tryptic peptide mixture was vacuum-dried and dissolved in 0.1% formic acid. Peptide extracts were purified on a C18 trap column and analyzed by use of a 2D-HPLC system coupled to a linear ion-trap mass spectrometer (Thermo Fisher Scientific, MA).
Immunoblotting
Proteins from the LD preparation were extracted with acetone, separated by 10% SDS-PAGE and underwent immunoblotting analysis with primary antibodies, then horseradish peroxidase-conjugated lgG. The blots were developed with enhanced chemiluminescence detection reagents (Applygen Technologies, Beijing).
Histology and immunofluorescence
Mice testes were fixed with 4% paraformaldehyde and embedded in paraffin and cut. For routine histology, sections were stained with hematoxylin-eosin. For immunofluorescence staining, sections were incubated for 10 min with 3% H2O2 to eliminate endogenous peroxidase activity and underwent antigen retrieval with 0.3% sodium citrate and phosphate buffered saline, pH 7.4, for 15 min at 72 °C. Sections were blocked with 1% defatted albumin and immunostained with primary antibody, then FITC-labeled lgG. Signals were observed under a Nikon Eclipse 50i fluorescence microscope.
LD staining
LDs in frozen testicular sections were stained with Nile Red. Nuclei were stained with Hoechst 33258. For in vitro staining, LDs purified from testicular tissue were spread on glass slides, dried and stained with Lipid-TOX Deep Red. Fluorescent signals were viewed under an Olympus FV1000 confocal microscope.
Thin-layer chromatography
LDs were purified from brown adipose tissue and testes of mice and from cultured Chinese hamster ovary (CHO) cells. Total lipids in different LD preparations were extracted in chloroform and acetone (1:2, v/v) and centrifuged at 14,000 × g for 10 min. The organic phase was collected and dried under nitrogen gas. Lipid extracts were dissolved in chloroform and loaded on silica gel plates for analysis. Neutral lipids were separated on plates in a hexane:diethyl ether:acetic acid (80:20:1, v/v/v) solvent system and visualized by the iodine vapor method.
Data mining and bioinformatics
To obtain reliable results, we performed at least two biological replicates of proteomic analysis and results were combined for further analysis. The online database used to sort the proteomic table was http://genome.ucsc.edu/cgi-bin/hgNear. Protein associations were revealed by the Website program String (http://string-db.org/).
Results
Testicular LD staining
Interstitial cells were located in the interstitium between the seminiferous tubules of mouse testicular tissue (Fig. 1A, panel a and b). Numerous small, concrete LDs stained with Nile Red were observed in interstitial Leydig cells rather than in the cells located within the seminiferous tubules (Fig. 1B). Lipid-TOX staining showed that the LDs prepared for proteomic analysis were morphologically intact, with a diameter of about 1 μm, despite the presence of a few large droplets (Fig. 1C).
Testicular lipid droplets (LDs) staining
A. Hematoxylin-eosin staining of mouse testicular tissue. The asterisk marks the interstitium between the seminiferous tubules in panel a. The amplified images of boxed area are in panel b. B. LDs stained with Nile Red in frozen testis sections. Nuclei were stained with Hoechst 33258. Panel a and b, Nild Red stained LDs. Panel c and d, the merged images. C. LDs purified from mice testes were spread on slides and stained with Lipid-TOX Deep Red.
Lipid and protein patterns of testicular LDs
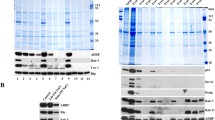
Thin-layer chromatography revealed that mice testicular LDs consisted of a fairly equivalent amount of cholesteryl esters and triacylglycerols and a small amount of ether lipid, similar to steroidogenic CHO cells; by contrast, adipose LDs contained a large amount of triacylglycerols but few cholesteryl esters and ether lipid (Fig. 2A). Equal amounts of protein extracted from different compartments were separated by SDS-PAGE. Silver staining revealed that the proteins in different LD preparations showed a highly consistent band pattern in gels (Fig. 2B), which indicated the reliability of the LD purification. In contrast, the protein band pattern of LD fractions differed from that of total membrane, cytosol and post-nuclear supernatant fractions (Fig. 2C).
Lipid and protein patterns in testicular LDs
A. Thin-layer chromatography analysis of total lipids extracted from LD preparations of mice testis, brown adipose tissue (BAT) and Chinese hamster ovary (CHO) cells. TG, triacylglycerols; CE, cholesteryl esters; EL, ether lipid. B. Silver-stained SDS-PAGE gels of protein extracts of different testicular lipid droplet preparations. C. Coomassie Blue-stained SDS-PAGE gels of proteins extracted from fractions of testicular LD, total membrane (TM), cytosol (Cyto) and post-nuclear supernatant (PNS). For the whole proteome, the lane running the testicular LD proteins was excised into 23 gel slices and underwent mass spectrometry.
Proteomic analysis of testicular LD proteins
For the whole proteome of testicular LDs, the lane running testicular LD protein was excised into 23 gel slices (Fig. 2C). After in-gel digestion, tryptic peptides underwent mass spectrometry analysis. Only proteins with at least two unique peptides were accepted for identification. A total of 337 proteins were identified; at least 144 (42.7% of total) were previously reported in LD proteomes of other mammalian cells or tissues and 44 were previously confirmed in LDs by microscopy. Each identified protein and its encoding gene were searched in the UniProt and NCBI databases and PubMed. The 337 proteins were classified into 16 groups by known or putative functional annotation for identified proteins (Fig. 3 and Table 1).
Properties of murine testicular LD proteins
A. Protein categories of mouse testis LDs. All proteins identified by 2D-LC MS/MS were sorted by subcellular distributions and known functions based on literature or NCBI online sources. B. Network of function-related LD proteins. Lines in different colors represent functional association in various types of evidence. Red, fusion evidence; green, neighborhood evidence; blue, co-occurrence evidence; purple, experiment evidence; yellow, text-mining evidence; black, co-expression evidence.
Group 1 proteins represented vimentin12 and stomatin13 and particularly Plin1, Plin2/ADRP, Plin3/Tip47 and Plin4/S3-12, 4 classical LD proteins belonging to the perilipin family of 5 LD proteins (Plin1~5) conserved in their first ~100 amino-terminal residues9. Plin1 binds and links vimentin to LDs, then vimentin filaments wrap the LDs tightly in a cage-like spherical structure surrounded by multiple ER tubules, thus facilitating LD formation12. Plin2~4 widely express and localize at LDs and non-LD compartments, but Plin1 expresses exclusively in adipose and steroidogenic cells and localizes only at the LD surface9. Plin1~4 provide a barrier and protect LDs against access by HSL and adipose triglyceride lipase (ATGL), but native Plin1 is more protective than Plin2~414,15. Interestingly, testicular LDs contained 4 variants of Plin1, termed Plin1a, 1b, 1c and 1d, which share conserved N-terminal 198 residues and 11-mer regions. This was the first identification of Plin1d protein in the tissue (Table 1).
Group 2 included 7 lipases/esterases/thioesterases, which cover almost all currently known cellular lipases/esterases. HSL14, ATGL and its co-lipase CGI-58 represent more than 95% of the lipolytic activity in adipocytes16, with the remaining hydrolase activity contributed by triacylglycerol hydrolase/carboxylesterase 317,18 and monoglyceride lipase19,20. LD-associated hydrolase (C2orf43 protein) is a cholesteryl ester hydrolase that normally localizes to the ER but is translocated to LDs on lipid loading21,22. ATGL expresses specifically in adipose tissue23, but HSL expresses primarily in both adipose and steroidogenic tissues.
Group 3 proteins represented 22 enzymes involved in the metabolism of fatty acid and glycerolipids and as well as phospholipids and sterols. Five were previously observed in LDs by microscopy. Fatty acid transport protein 1 binds diacylglycerol acyltransferase 2 and colocalizes to the ER-LD interface to facilitate glycerolipid biosynthesis and LD expansion24. Long-chain acyl-CoA synthetase Acsl125 and Acsl325,26, along with glycerol-3-phosphate O-acyltransferase (Gpat4), are normally localized in the ER microdomain but effectively translocated to nascent LDs to facilitate LD biosynthesis on lipid loading25,26. Acsl4 and fatty aldehyde dehydrogenase were morphologically localized in yeast LDs27 and proteomically detected in LDs of CHO cells28, adipocytes29 and mouse muscle30. Carnitine O-palmitoyltransferase 2, very-long-chain acyl-CoA dehydrogenase and mitochondrial trifunctional enzyme subunit α were detected from mouse muscle LDs30. Fatty acid synthase was detected from LDs of granulosa steroidogenic cells from rat ovary20. Many proteins in this group are known to specifically or highly express in testes (Table 1).
Group 4 proteins represented 11 phospholipid metabolic enzymes; 3 were previously physiologically confirmed in LDs. Phospholipase B is highly expressed in testis and activated by sterol removal in murine sperm membrane, which localizes at the LD surface and hydrolyzes glycerophospholipids to facilitate the LD structure31. Cytosolic phospholipase A2 (cPLA2) is activated by extracellular stimuli-hydrolyzed arachidonic acids from the sn-2 position of glycerophospholipids; in turn, released arachidonic acids induce the translocation of cPLA2 to the ER and LD interface to regulate lipid synthesis and nascent LD formation32,33. Phosphocholine cytidylyltransferase binds to growing LDs and then catalyzes phospholipid synthesis and promotes LD expansion34,35. Phosphatidylglycerophosphate synthase 1 and phosphoinositide lipid phosphatase are highly expressed in testes and phospholipase DDHD1 is required for spermatogenesis. The proteins in this group also participate in glycerolipid and sterol metabolism.
Group 5 contained 19 proteins that participate in biosynthesis and metabolism of cholesterol, retinol and hormonal steroids; 6 were previously observed in LDs by microscopy and another 7 were previously detected in LD proteomes. Short-chain dehydrogenase/reductase 3 and retinol dehydrogenase 10 are reciprocally activated and on acyl ester biosynthesis, are translocated from the ER to LDs36,37,38. The key steroidogenic enzymes lanosterol synthase27, 3β-hydroxysteroid dehydrogenase (HSD) 1 and 730,39,40, 17β-HSD-4, −7, −11 and −1711,30,40,41,42 and NAD(P)H steroid dehydrogenase-like43,44 were previously microscopically or proteomically detected in intracellular LDs. Many of these enzymes, such as 17-α-hydroxyprogesterone aldolase and scavenger receptor class B-I20, are highly expressed in testes and regulate cholesterol homeostasis.
Group 6 proteins represented 17 enzymes involving in glucuronidation and glycosylation. UDP-glucuronosyltransferase 1–640, DolP-glucosyltransferase11,28, α-glucosidase20 and methyltransferase-like protein 7A40 were previously found in LD proteomes and methyltransferase-like protein 7B was observed in LDs by microscopy19,45,46. CGI-49 proteins are frequently found in LD proteomes11,29,30,41. Large oligosaccharyltransferase complexes contain ribophorin I, Stt3a, Stt3b, p97/Vcp, Sel1l and Ubxd847 and may also interact with ancient ubiquitous protein 1 (Aup1), Acsl3 and stomatin48. Ubxd849,50, p97/Vcp49,50, Aup148, Acsl325,26 and stomatin13 have been verified in LDs by microscopy, which suggests that the present identification is reliable. Several enzymes in this group catalyze glucuronidation reactions of estrogens, testosterones, retinoic acids and various metabolites of xenobiotics and endobiotics47.
Group 7 and 8 proteins included 29 enzymes involved in the metabolism of carbohydrate and tricarboxylic acid cycle. NADH-cytochrome b5 reductase was verified in LDs by microscopy45. Glutamate dehydrogenase, malate dehydrogenase, succinate dehydrogenase, lactate dehydrogenase, pyruvate kinase 2/3 and citrate synthase were previously reported in LD proteomes11,20,51. The identification of 17 other metabolic enzymes in testicular LDs is novel, which might reflect the close relationship between LDs and mitochondria in testicular cells52.
Group 9 proteins represented 28 small GTPases; 27 were previously reported in LD proteomes. In cells loaded with fatty acids, Rab5a53, Rab11a53, Arl2 GTPase Elmod254 and Rab1853,55 can localize to both the ER and LDs, where Rab18 recruits unknown effectors and microtubules to facilitate membrane trafficking and lipid exchange53,55. Testicular LDs might serve as a dock for various small GTPases for mediating Rab signaling.
Group 10 listed 30 protein chaperones; 18 were previously reported in LD proteomes. We previously showed that heat shock protein 70 (Hsp70) can translocate to adipocyte LDs on heat stimulation56. Spermatid-specific Hsp70, Hsp70.2 (Hspa2), T-complex protein 11 and protein disulfide-isomerase A3 (PDI3a) are testis-specific and play roles in spermatogenesis. PDI is a component of microsomal triacylglycerol transfer protein complex. T-complex protein 1 contains 8 distinct subunits to form a unique chaperone for escorting actin, tubulin and numerous other proteins. In Leydig cells, the intermediate filaments of the cytoskeletons may bind to LDs52.
Group 11 listed 18 proteins involved in proteasome and membrane trafficking. Among them, p97, Atad3a and Afg3l2 are AAA ATPase family proteins that regulate ubiquination, membrane trafficking and organelle biogenesis. p97, Ubxd2 and Ubxd8/Faf2) bind with each other and colocalize to LDs46,49,50. Aup1 localizes to the ER and LDs48,57. Aup1 may exist in several subcomplexes and associate with numerous other proteins48 such as Ubxd8, Ubxd2, Atad3a, RuvB-like 1, stomatin, ribophorin I and II, T-complex proteins, epoxide hydrolase 1, atlastin-3, Acsl3, pyruvate kinase 2/3, PDI and ATP synthase48. Dozens of Aup1-associated proteins were also identified in testicular LDs, which might reflect the close association of these protein complexes with cellular LDs.
Group 12 contained 43 transport proteins; 16 were proteomically reported11,30,40,58 and 5 were microscopically confirmed in LDs59,60. Coatomer protein complex I (COPI) and clathrin adaptor complex mediate intra-Golgi transport and retrograde transport from the Golgi to ER. Arf1/COPI complexes localize between the ER and LDs for targeting the triacylglycerol synthesis enzyme Gpat4 to the LD surface and bud 60-nm nanodroplets from the LDs. In cells loaded with fatty acids, both COPI and COPII (Sec23) structures tend to localize to discrete foci surrounding LDs to create a membrane bridge for transporting ATGL and Plin2 to nascent LDs60.
Group 13 contained 26 proteins involved in nucleotide-catabolic processes, such as ion transport, transcription, translation and cell signaling. Nine proteins were detected by previous LD proteomes. Some proteins might not easily fit into this single category because of the divergence of protein functions. MAPK/ERK kinase 2 is colocalized with cPLA2 in LDs, then rapidly activates cPLA2 for releasing arachidonate from LDs33; it is required for testosterone synthesis in Leydig cells. ATP synthase subunit α and sodium pump subunit α1 and α4 are expressed abundantly in testis and regulate spermatogenesis.
Group 14~16 included cytoskeletal proteins, testis-specific and miscellaneous proteins. Only 11 of these 81 proteins were previously reported in LD proteomes. The identification of albumin in the present and previous LD proteomes should represent a contamination because of its abundance in serum. The identification of testis-specific proteins could be due to the contamination or the difficult separation of these protein components from testicular LDs. For example, GAPDH2 and A-kinase anchor protein 3 and 4 participate in spermatogenesis, which can bind the cytoskeletal fibrous sheath and thus might be co-purified with LD-associated cytoskeletons. Also, these testis-specific or spermatogenesis-related proteins might exist in cellular subcomplex structures that associate with testicular LDs52.
Confirmation of testicular LD protein identification by immunoblotting and immunofluorescence
Some of the identified testicular LD proteins were confirmed by immunoblotting by using marker proteins corresponding to different cellular compartments (Fig. 4A). Four members of the perilipin family, Plin1~4, including the 4 variants of Plin1, Plin1a, 1b, 1c and 1d, were detected only in the LD fraction. This was the first immunodetection of Plin1d in tissue (Fig. 4A). Plin5 signal was not detectable in testicular LD extracts (data not shown), which is consistent with its low level of expression in non-oxidative tissues. ATGL and CGI-58 appeared only in the LD fraction; HSL and 3β-HSD1 were highly enriched in the LD fraction but also detectable in the membrane and cytosol compartments (Fig. 4A). Caveolin-1 and -3, caveolae marker proteins, were not identified in the testicular LD proteome (Table 1) but were immunodetected in the LD fraction or other cellular compartments (Fig. 4A). Aromatase, a cyp19 enzyme that converts androgen to estrogen in seminiferous epithelium, was marginally detected in the testicular LD fraction but appeared mainly in the membrane fraction (Fig. 4A). Lysosome protein Lamp-1, ER protein p62 and cytoplasmic enzyme GAPDH were not detected in the LD fraction. The ER chaperone GRP78 and mitochondrial protein Tim 23 were detected predominately in the membrane and post-nuclear supernatant fractions, but a small amount appeared in the LD fraction (Fig. 4A). Clearly, the isolated LD fraction of mice testes was largely free of other organelle contamination, although a small amount of the ER and mitochondria components might be introduced, likely because of their abundance or general interactions with LDs61. Furthermore, immunofluorescent signals of Plin1 appeared strongly in the interstitium of mice testis (Fig. 4B, panel a and c) and the fluorescent signal pattern was consistent with that of interstitial LDs stained with Nile Red (Fig. 1B, panel a and b). Immunofluorescent signals were weaker for Plin2 and 3β-HSD1 than Plin1 but still detectable in interstitial locations (Fig. 4B, panel e and g). The immunofluorescent signal for 17β-HSD11 was not detected (data not shown).
Confirmation of LD proteins by immunodetection
A. The fractions of LD, total membrane (TM), cytosol (Cyto) and post-nuclear supernatant (PNS) were prepared from mice testes. An equal amount of proteins extracted from different fractions was separated by SDS-PAGE and underwent immunoblotting with the primary antibodies indicated. A representative silver-stained gel showed equivalent protein loading. Plin variants Plin1a~1d were detected on a full-length blot. The blots of proteins were derived from the sample or different samples that were processed in parallel and the corresponding full-length blots are shown in Supplementary Figure S1. Arom, aromatase; Cav, caveolin; Plin, perilipin; HSL, hormone-sensitive lipase; ATGL, adipose triglyceride lipase. B. Immunofluorescent staining of Plin1 (a,c) Plin2 (e) and 3β-hydroxysteroid dehydrogenase (HSD3B1) (g) in sections of mouse testis. The merged images are shown in panel b,d,f and h.
Discussion
We report the first proteomic analysis of LDs purified from adult mice testes. Testicular LDs contained 337 proteins; 144 were previously detected in LD proteomes and 44 were verified by microscopy. From the functions of identified proteins, testicular LDs showed several characteristics different from the LDs of most other cell types. Testicular LDs may be unique, biologically active cellular organelles that might have functional roles in the biosynthesis of hormonal steroids.
First, testicular LDs featured most Plin family and lipase/esterase superfamily proteins and various enzymes for biosynthesis and metabolism of glycerolipids and phospholipids. The classical LD proteins, Plin1~4 and 4 variants of Plin1, are crucial for regulating LD formation9. During LD expansion in differentiating adipocytes, nascent small LDs are coated with Plin3 and Plin4, medial-size LDs require both Plin2 and Plin1 and finally, Plin1 replaces Plin2 as a major coat of large LDs in mature adipocytes62. We previously revealed that Plin2 is degraded by the proteasome with the induction of Plin163 and if Plin1 is null for replacing Plin2, LD growth and adipocyte differentiation are retarded64. Different Plins target different types of LDs and have unique functions to govern triacylglyceride–cholesterol ester balance15. Plin1a and Plin1b favor triacylglyceride-rich LDs15, Plin1c and Plin4 prefer cholesteryl ester-rich LDs, but Plin2 and Plin3 show less specific localization to LDs15. Plin1 expresses exclusively in adipose and steroidogenic cells9. Thin-layer chromatography revealed that the LD of adipocytes was triacylglyceride-rich, so it associates mainly with Plin1a and Plin1b. In contrast, the testicular LD had a relatively equivalent proportion of triacylglycerides and cholesteryl esters. The accumulation of triacylglycerides promotes and stabilizes storage of cholesteryl esters within Leydig cells5. Likely, the coats of Plin1~4, including Plin1a~1d, could cooperatively manipulate the appropriate balance of cholesteryl ester-triacylglycerides in steroidogenic cells of testes.
Also, testicular LDs contained most of the known lipases/esterases/phospholipases and enzymes of glycerolipid and phospholipid metabolism. HSL and ATGL represent ~95% of the lipolytic activity in adipocytes16 and the remaining activity is contributed by triacylglycerol hydrolase17,18 and monoglyceride lipase19. We and others previously revealed that Plin1 phosphorylation induces the translocation of HSL from the cytosol to LDs14,65 and also indirectly activates ATGL by unsequestering the ATGL coactivator CGI-58, hence conferring a full lipolytic reaction in adipocytes. HSL is stimulated by catecholamine, thyroxine and glucocorticoid66 and in testes, HSL is activated by chorionic gonadotropin. Inactivation of ATGL causes diacylglyceride accumulation in testes23, but HSL ablation disables spermatogenesis and causes male infertility67. Despite these crucial roles of lipases, the control of lipolysis and even the catalog of lipases (except HSL) are largely unknown in testes. Although lipases can act on broad lipid substrates (e.g., glycerolipids in adipocytes), in Leydig cells, they predominately hydrolyze cholesterol esters to cholesterols for steroidogenesis68. Unlike testicular LDs, the LDs in other types of cells including adipocytes were not found to contain so many lipases/esterases and enzymes for glycerolipid and phospholipid metabolism. Likely, testicular LDs need to be accurately modulated by these different enzymes, to facilitate the biosynthesis and hydrolysis of cholesteryl esters and thereby ensure cholesterol supply for steroidogenesis in testes.
The second unique feature is that testicular LDs contained a large number of steroidogenic enzymes such as lanosterol synthase and demethylase, various hydroxysteroid and retinol dehydrogenases and various glucuronidation enzymes. Currently, steroidogenic enzymes are known to locate in the ER and mitochondrial membranes and in the adjacent cytoplasm, where they catalyze different reactions, their substrates and products being shuttled between these compartments47,69. The enzymes identified in testicular LDs, such as short-chain dehydrogenase 337,38, retinol dehydrogenase 1036, 17β-HSD1141,42, 3β-HSD139,40 and NAD(P)H steroid dehydrogenase-like protein43,44, another 3β-HSD, can translocate from the ER membrane to the LD surface on acyl ester biosynthesis. The substrates, products and metabolites of steroidogenic reactions are mostly insoluble and cannot distribute and move freely in the cytoplasm but instead could be chaperoned and escorted by hydrophobic LDs. Thus, considering that testicular LDs are spatially close to the ER and mitochondria and contain so many steroidogenic enzymes at the oil–water interface, the present data suggests that testicular LDs could be a new compartment for carrying out steroidogenic reactions, more than just a simple pool of cholesterol substrates. At least, testicular LDs could be a chaperone vehicle to facilitate the biosynthesis of hormonal steroids, by transferring insoluble intermediate substrates and products between the mitochondria and the adjacent cytoplasm.
Third, testicular LDs contained large numbers of proteins involved in cellular signaling, chaperon, ubiquination, transport, cytoskeleton and spermatogenesis. Proteins in the GTPase superfamily and Rab GTPase subfamily were particularly abundant. Rab1853,55 can recruit microtubules and localize between the ER and LDs to facilitate membrane trafficking and lipid exchange53,55. Ubxd8 and p97/VCP colocalize at the ER-LD interface and promote LD expansion by binding ATGL and inhibiting ATGL-mediated LD lipolysis49. Similarly, the vesicle transporters COPI and COPII are membrane bridges between the ER and LDs to deliver and modulate ATGL, Plin2 and Plin3 levels at nascent LDs60. Because many of these proteins may exist in large multicomponent complexes, their simultaneous identification from testicular LDs was not surprising. For example, Aup1 localizes to the ER and LDs and contributes to the formation of LDs that may temporarily store misfolded ER proteins under certain conditions48,57. Actually, Aup1 is a component of the Hrd1–Sel1l ER quality-control complex and physiologically associates with a hundred other proteins48. In comparison, testicular LDs contained at least dozens of Aup1-associated proteins48, such as Ubxd8, Ubxd2, p97/VCP, Atad3a, Sel1l, Ruvb-like 1, stomatin, ribophorin I, T-complex proteins, epoxide hydrolase, atlastin-3, Acsl3, pyruvate kinase 2/3 and PDI3a. In addition, testicular LDs contained many cytoskeletal proteins, which might not be simply considered contamination. In steroidogenic cells, the LDs and mitochondria are known to tightly attach to the cytoskeleton and intermediate filaments that are thought to mediate transport of cholesterol70. An example is vimentin filaments, which bind Plin1 and wrap LDs12. Vimentin ablation results in defective steroidogenesis in adrenocortical and granulosa cells69. Overall, these findings suggest that testicular LDs could participate initially in cellular signaling, chaperon, ubiquination, transport, cytoskeleton and spermatogenesis.
In summary, testicular LDs could be considered active cellular organelles participating in the regulation of multiple testicular functions. Plins and lipases/esterases/phospholipases could govern accurate control of the biosynthesis and hydrolysis of cholesteryl esters, thus ensuring appropriate cholesteryl ester-triacylglyceride balance and cholesterol supply for steroidogenesis. Notably, the association with various kinds of steroidogenic enzymes suggests that steroidogenic reactions might occur in testicular LDs or the steroidogenic enzymes and products could be transferred through testicular LDs. Because little was known about testicular LD proteins, the investigation of the roles of testicular LDs has been largely restricted to morphological observations. The present finding uncovers the full set of testicular LD proteins, for further examination of the functional roles of testicular LDs and their proteins in steroidogenesis and spermatogenesis in testes.
Additional Information
How to cite this article: Wang, W. et al. Proteomic analysis of murine testes lipid droplets. Sci. Rep. 5, 12070; doi: 10.1038/srep12070 (2015).
References
Neaves, W. B. Changes in testicular leydig cells and in plasma testosterone levels among seasonally breeding rock hyrax. Biol Reprod 8, 451–466 (1973).
Mori, H. & Christensen, A. K. Morphometric analysis of Leydig cells in the normal rat testis. J Cell Biol 84, 340–354 (1980).
Mori, H., Hiromoto, N., Nakahara, M. & Shiraishi, T. Stereological analysis of Leydig cell ultrastructure in aged humans. The Journal of clinical endocrinology and metabolism 55, 634–641 (1982).
Morris, M. D. & Chaikoff, I. L. The origin of cholesterol in liver, small intestine, adrenal gland and testis of the rat: dietary versus endogenous contributions. J Biol Chem 234, 1095–1097 (1959).
Freeman, D. A. & Ascoli, M. Studies on the source of cholesterol used for steroid biosynthesis in cultured Leydig tumor cells. J Biol Chem 257, 14231–14238 (1982).
Ariyaratne, H. B. & Chamindrani Mendis-Handagama, S. Changes in the testis interstitium of Sprague Dawley rats from birth to sexual maturity. Biol Reprod 62, 680–690 (2000).
Paniagua, R., Rodriguez, M. C., Nistal, M., Fraile, B. & Amat, P. Changes in the lipid inclusion/Sertoli cell cytoplasm area ratio during the cycle of the human seminiferous epithelium. J Reprod Fertil 80, 335–341 (1987).
Kerr, J. B. & De Kretser, D. M. Cyclic variations in Sertoli cell lipid content throughout the spermatogenic cycle in the rat. J Reprod Fertil 43, 1–8 (1975).
Brasaemle, D. L. The perilipin family of structural lipid droplet proteins: stabilization of lipid droplets and control of lipolysis. J Lipid Res 48, 2547–2559 (2007).
Ding, Y. et al. Isolating lipid droplets from multiple species. Nat Protoc 8, 43–51 (2013).
Bartz, R. et al. Dynamic activity of lipid droplets: protein phosphorylation and GTP-mediated protein translocation. J Proteome Res 6, 3256–3265 (2007).
Heid, H. et al. On the formation of lipid droplets in human adipocytes: the organization of the perilipin-vimentin cortex. PLoS One 9, e90386 (2013).
Umlauf, E. et al. Association of stomatin with lipid bodies. J Biol Chem 279, 23699–23709 (2004).
Sztalryd, C. et al. Perilipin A is essential for the translocation of hormone-sensitive lipase during lipolytic activation. J Cell Biol 161, 1093–1103 (2003).
Hsieh, K. et al. Perilipin family members preferentially sequester to either triacylglycerol-specific or cholesteryl-ester-specific intracellular lipid storage droplets. J Cell Sci 125, 4067–4076 (2012).
Schweiger, M. et al. Adipose triglyceride lipase and hormone-sensitive lipase are the major enzymes in adipose tissue triacylglycerol catabolism. J Biol Chem 281, 40236–40241 (2006).
Soni, K. G. et al. Carboxylesterase 3 (EC 3.1.1.1) is a major adipocyte lipase. J Biol Chem 279, 40683–40689 (2004).
Wang, H. et al. Altered lipid droplet dynamics in hepatocytes lacking triacylglycerol hydrolase expression. Mol Biol Cell 21, 1991–2000 (2010).
Turro, S. et al. Identification and characterization of associated with lipid droplet protein 1: A novel membrane-associated protein that resides on hepatic lipid droplets. Traffic 7, 1254–1269 (2006).
Khor, V. K. et al. The proteome of cholesteryl-ester-enriched versus triacylglycerol-enriched lipid droplets. PLoS One 9, e105047 (2014).
Thiel, K. et al. The evolutionarily conserved protein CG9186 is associated with lipid droplets, required for their positioning and for fat storage. J Cell Sci 126, 2198–2212 (2013).
Goo, Y. H., Son, S. H., Kreienberg, P. B. & Paul, A. Novel lipid droplet-associated serine hydrolase regulates macrophage cholesterol mobilization. Arterioscler Thromb Vasc Biol 34, 386–396 (2014).
Zimmermann, R. et al. Fat mobilization in adipose tissue is promoted by adipose triglyceride lipase. Science 306, 1383–1386 (2004).
Xu, N. et al. The FATP1-DGAT2 complex facilitates lipid droplet expansion at the ER-lipid droplet interface. J Cell Biol 198, 895–911 (2012).
Wilfling, F. et al. Triacylglycerol synthesis enzymes mediate lipid droplet growth by relocalizing from the ER to lipid droplets. Dev Cell 24, 384–399 (2013).
Poppelreuther, M. et al. The N-terminal region of acyl-CoA synthetase 3 is essential for both the localization on lipid droplets and the function in fatty acid uptake. J Lipid Res 53, 888–900 (2012).
Natter, K. et al. The spatial organization of lipid synthesis in the yeast Saccharomyces cerevisiae derived from large scale green fluorescent protein tagging and high resolution microscopy. Mol Cell Proteomics 4, 662–672 (2005).
Liu, P. et al. Chinese hamster ovary K2 cell lipid droplets appear to be metabolic organelles involved in membrane traffic. J Biol Chem 279, 3787–3792 (2004).
Brasaemle, D. L., Dolios, G., Shapiro, L. & Wang, R. Proteomic analysis of proteins associated with lipid droplets of basal and lipolytically stimulated 3T3-L1 adipocytes. J Biol Chem 279, 46835–46842 (2004).
Zhang, H. et al. Proteome of skeletal muscle lipid droplet reveals association with mitochondria and apolipoprotein a-I. J Proteome Res 10, 4757–4768 (2011).
Selvaraju, K., Rajakumar, S. & Nachiappan, V. Identification of a phospholipase B encoded by the LPL1 gene in Saccharomyces cerevisiae. Biochim Biophys Acta 1842, 1383–1392 (2014).
Wooten, R. E. et al. Novel translocation responses of cytosolic phospholipase A2alpha fluorescent proteins. Biochim Biophys Acta 1783, 1544–1550 (2008).
Yu, W. et al. Co-compartmentalization of MAP kinases and cytosolic phospholipase A2 at cytoplasmic arachidonate-rich lipid bodies. Am J Pathol 152, 759–769 (1998).
Krahmer, N. et al. Phosphatidylcholine synthesis for lipid droplet expansion is mediated by localized activation of CTP:phosphocholine cytidylyltransferase. Cell Metab 14, 504–515 (2011).
Guo, Y. et al. Functional genomic screen reveals genes involved in lipid-droplet formation and utilization. Nature 453, 657–661 (2008).
Jiang, W. & Napoli, J. L. The retinol dehydrogenase Rdh10 localizes to lipid droplets during acyl ester biosynthesis. J Biol Chem 288, 589–597 (2013).
Deisenroth, C., Itahana, Y., Tollini, L., Jin, A. & Zhang, Y. p53-Inducible DHRS3 is an endoplasmic reticulum protein associated with lipid droplet accumulation. J Biol Chem 286, 28343–28356 (2011).
Adams, M. K., Belyaeva, O. V., Wu, L. & Kedishvili, N. Y. The retinaldehyde reductase activity of DHRS3 is reciprocally activated by retinol dehydrogenase 10 to control retinoid homeostasis. J Biol Chem 289, 14868–14880 (2014).
Beilstein, F., Bouchoux, J., Rousset, M. & Demignot, S. Proteomic analysis of lipid droplets from Caco-2/TC7 enterocytes identifies novel modulators of lipid secretion. PLoS One 8, e53017 (2013).
Bouchoux, J. et al. The proteome of cytosolic lipid droplets isolated from differentiated Caco-2/TC7 enterocytes reveals cell-specific characteristics. Biol Cell 103, 499–517 (2011).
Fujimoto, Y. et al. Identification of major proteins in the lipid droplet-enriched fraction isolated from the human hepatocyte cell line HuH7. Biochim Biophys Acta 1644, 47–59 (2004).
Horiguchi, Y., Araki, M. & Motojima, K. Identification and characterization of the ER/lipid droplet-targeting sequence in 17beta-hydroxysteroid dehydrogenase type 11. Arch Biochem Biophys 479, 121–130 (2008).
Ohashi, M., Mizushima, N., Kabeya, Y. & Yoshimori, T. Localization of mammalian NAD(P)H steroid dehydrogenase-like protein on lipid droplets. J Biol Chem 278, 36819–36829 (2003).
Caldas, H. & Herman, G. E. NSDHL, an enzyme involved in cholesterol biosynthesis, traffics through the Golgi and accumulates on ER membranes and on the surface of lipid droplets. Hum Mol Genet 12, 2981–2991 (2003).
Zehmer, J. K., Bartz, R., Liu, P. & Anderson, R. G. Identification of a novel N-terminal hydrophobic sequence that targets proteins to lipid droplets. J Cell Sci 121, 1852–1860 (2008).
Zehmer, J. K. et al. Targeting sequences of UBXD8 and AAM-B reveal that the ER has a direct role in the emergence and regression of lipid droplets. J Cell Sci 122, 3694–3702 (2009).
Ritter, J. K. Roles of glucuronidation and UDP-glucuronosyltransferases in xenobiotic bioactivation reactions. Chem Biol Interact 129, 171–193 (2000).
Klemm, E. J., Spooner, E. & Ploegh, H. L. Dual role of ancient ubiquitous protein 1 (AUP1) in lipid droplet accumulation and endoplasmic reticulum (ER) protein quality control. J Biol Chem 286, 37602–37614 (2011).
Olzmann, J. A., Richter, C. M. & Kopito, R. R. Spatial regulation of UBXD8 and p97/VCP controls ATGL-mediated lipid droplet turnover. Proc Natl Acad Sci U.S.A. 110, 1345–1350 (2013).
Suzuki, M. et al. Derlin-1 and UBXD8 are engaged in dislocation and degradation of lipidated ApoB-100 at lipid droplets. Mol Biol Cell 23, 800–810 (2012).
Orban, T., Palczewska, G. & Palczewski, K. Retinyl ester storage particles (retinosomes) from the retinal pigmented epithelium resemble lipid droplets in other tissues. J Biol Chem 286, 17248–17258 (2011).
Almahbobi, G., Williams, L. J., Han, X. G. & Hall, P. F. Binding of lipid droplets and mitochondria to intermediate filaments in rat Leydig cells. J Reprod Fertil 98, 209–217 (1993).
Liu, P. et al. Rab-regulated interaction of early endosomes with lipid droplets. Biochim Biophys Acta 1773, 784–793 (2007).
East, M. P., Bowzard, J. B., Dacks, J. B. & Kahn, R. A. ELMO domains, evolutionary and functional characterization of a novel GTPase-activating protein (GAP) domain for Arf protein family GTPases. J Biol Chem 287, 39538–39553 (2012).
Martin, S., Driessen, K., Nixon, S. J., Zerial, M. & Parton, R. G. Regulated localization of Rab18 to lipid droplets: effects of lipolytic stimulation and inhibition of lipid droplet catabolism. J Biol Chem 280, 42325–42335 (2005).
Jiang, H., He, J., Pu, S., Tang, C. & Xu, G. Heat shock protein 70 is translocated to lipid droplets in rat adipocytes upon heat stimulation. Biochim Biophys Acta 1771, 66–74 (2007).
Jo, Y., Hartman, I. Z. & DeBose-Boyd, R. A. Ancient ubiquitous protein-1 mediates sterol-induced ubiquitination of 3-hydroxy-3-methylglutaryl CoA reductase in lipid droplet-associated endoplasmic reticulum membranes. Mol Biol Cell 24, 169–183 (2013).
Dahlhoff, M. et al. Characterization of the sebocyte lipid droplet proteome reveals novel potential regulators of sebaceous lipogenesis. Exp Cell Res 332, 146–155 (2015).
Wilfling, F. et al. Arf1/COPI machinery acts directly on lipid droplets and enables their connection to the ER for protein targeting. Elife 3, e01607 (2014).
Soni, K. G. et al. Coatomer-dependent protein delivery to lipid droplets. J Cell Sci 122, 1834–1841 (2009).
Zehmer, J. K. et al. A role for lipid droplets in inter-membrane lipid traffic. Proteomics 9, 914–921 (2009).
Nagayama, M., Uchida, T. & Gohara, K. Temporal and spatial variations of lipid droplets during adipocyte division and differentiation. J Lipid Res 48, 9–18 (2007).
Xu, G. et al. Post-translational regulation of adipose differentiation-related protein by the ubiquitin/proteasome pathway. J Biol Chem 280, 42841–42847 (2005).
Lyu, Y. et al. Defective differentiation of adipose precursor cells from lipodystrophic mice lacking perilipin 1. PLoS One 10, e0117536 (2015).
He, J. et al. Calyculin and okadaic acid promote perilipin phosphorylation and increase lipolysis in primary rat adipocytes. Biochim Biophys Acta 1761, 247–255 (2006).
Xu, C. et al. Direct effect of glucocorticoids on lipolysis in adipocytes. Mol Endocrinol 23, 1161–1170 (2009).
Osuga, J. et al. Targeted disruption of hormone-sensitive lipase results in male sterility and adipocyte hypertrophy, but not in obesity. Proc. Natl. Acad. Sci. U.S.A 97, 787–792 (2000).
Freeman, D. A. Regulation of the cholesterol ester cycle of cultured Leydig tumor cells. Eur J Biochem 164, 351–356 (1987).
Kraemer, F. B., Khor, V. K., Shen, W. J. & Azhar, S. Cholesterol ester droplets and steroidogenesis. Mol Cell Endocrinol 371, 15–19 (2012).
Hall, P. F. & Almahbobi, G. Roles of microfilaments and intermediate filaments in adrenal steroidogenesis. Microsc Res Tech 36, 463–479 (1997).
Cho, S. Y. et al. Identification of mouse Prp19p as a lipid droplet-associated protein and its possible involvement in the biogenesis of lipid droplets. J Biol Chem 282, 2456–2465 (2007).
Currie, E. et al. High confidence proteomic analysis of yeast LDs identifies additional droplet proteins and reveals connections to dolichol synthesis and sterol acetylation. J Lipid Res 55, 1465–1477 (2014).
Naslavsky, N., Rahajeng, J., Rapaport, D., Horowitz, M. & Caplan, S. EHD1 regulates cholesterol homeostasis and lipid droplet storage. Biochem Biophys Res Commun 357, 792–799 (2007).
Acknowledgements
This work was supported by the National Natural Science Foundation of China [91439119, 31300964] and by the Beijing Natural Science Foundation [7152080] and the National Basic Research Program of China [2012CB517505].
Author information
Authors and Affiliations
Contributions
G.X. and W.W. conceived and designed the experiments. W.W., S.W., L.L., X.S., C.D., F.L., B.G. and P.L. performed experiments and prepared Figs 1–4. W.W., G.X. and P.L. analyzed the data and wrote the paper. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Wang, W., Wei, S., Li, L. et al. Proteomic analysis of murine testes lipid droplets. Sci Rep 5, 12070 (2015). https://doi.org/10.1038/srep12070
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep12070
- Springer Nature Limited
This article is cited by
-
Protective Effect of Low-Volume High-Intensity Interval Training on Aspirin-Induced Reproductive Impairments in Adult Male Wistar Rats
Reproductive Sciences (2024)
-
PSMD1 and PSMD2 regulate HepG2 cell proliferation and apoptosis via modulating cellular lipid droplet metabolism
BMC Molecular Biology (2019)
-
Lipid Droplet-Associated Hydrolase Promotes Lipid Droplet Fusion and Enhances ATGL Degradation and Triglyceride Accumulation
Scientific Reports (2017)
-
Integrated omics study of lipid droplets from Plasmodiophora brassicae
Scientific Reports (2016)