Abstract
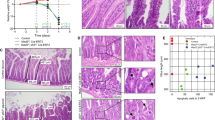
The p53-binding protein 53BP1 has been implicated in the DNA damage response and genomic instability. Previous reports have highlighted these roles in vivo in haematopoietic lineages. To investigate the importance of 53BP1 to the DNA damage response in epithelial cells in vivo, we have investigated the role of 53BP1 in mediating apoptosis and proliferation within the murine small intestine following γ-irradiation. 53BP1 deficiency does not affect the immediate response to γ-irradiation with normal levels of apoptosis, proliferation and p53 and p21 accumulation. However, 48 h post-γ-irradiation there was a significant accumulation of cells with much larger nuclei marked by p53 and p21 accumulation. These data reflect increases in polyploidy observed 53BP1−/− deficient fibroblasts following γ-irradiation. At 72 h post-irradiation both the 4N and 8N populations were significantly increased in 53BP1−/− MEFS. Taken together, these results show that following in vivo exposure to γ-irradiation, 53BP1 is dispensable for signalling apoptosis and cell-cycle arrest in the intestinal epithelium. However, it is important for prevention of genomic instability within this epithelial cell population.



Similar content being viewed by others
References
Anderson L, Henderson C, Adachi Y . (2001). Phosphorylation and rapid relocalization of 53BP1 to nuclear foci upon DNA damage. Mol Cell Biol 21: 1719–1729.
Bartkova J, Horejsi Z, Koed K, Kramer A, Tort F, Zieger K et al. (2005). DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 434: 864–870.
Clarke AR, Gledhill S, Hooper ML, Bird CC, Wyllie AH . (1994). p53 dependence of early apoptotic and proliferative responses within the mouse intestinal epithelium following γ-irradiation. Oncogene 9: 1767–1773.
Clarke AR, Howard LA, Harrison DJ, Winton DJ . (1997). p53, mutation frequency and apoptosis in murine small intestine. Oncogene 14: 2015–2018.
Derbyshire DJ, Basu BP, Serpell LC, Joo WS, Date T, Iwabuchi K et al. (2002). Crystal structure of human 53BP1 BRCT domains bound to p53 tumour suppressor. EMBO J 21: 3863–3872.
DiTullio RA, Mochan TA, Venere M, Bartkova J, Sehested M, Bartek J et al. (2002). 53BP1 functions in an ATM-dependent checkpoint that is constitutively activated in human cancer. Nat Cell Biol 4: 998–1002.
Fernandez-Capetillo O, Chen H-T, Celeste A, Ward I, Romanienko PJ, Morales JC et al. (2003). DNA damage-induced G2-M checkpoint activation by histone H2AX and 53BP1. Nat Cell Biol 4: 993–997.
Finke D, Kraehenbuhl J-P . (2001). Formation of Peyer's patches. Curr Opin Genet Dev 11: 561–567.
Fujiwara T, Bandi M, Nitta M, Ivanova EV, Bronson RT, Pellman D . (2005). Cytokinesis failure generating tetraploids promotes tumorigenesis in p53-null cells. Nature 437: 1043–1047.
Gorgoulis VG, Vassiliou LV, Karakaidos P, Zacharatos P, Kotsinas A, Liloglou T et al. (2005). Activation of the DNA damage checkpoint and genomic instability in human precancerous lesions. Nature 434: 907–913.
Iwabuchi K, Bartel PL, Li B, Marraccino R, Fields S . (1994). Two cellular proteins that bind to wild-type but not mutant p53. PNAS 91: 6098–6102.
Iwabuchi K, Li B, Massa HF, Trask BJ, Date T, Fields S . (1998). Stimulation of p53-mediated transcriptional activation by the p53-binding proteins, 53BP1 and 52BP2. J Biol Chem 273: 26061–26068.
Joo WS, Jeffrey PD, Cantor SB, Finnin MS, Livingston DM, Pavletich NP . (2002). Structure of the 53BP1 BRCT region bound to p53 and its comparison to the Brca1 BRCT structure. Genes Dev 16: 583–593.
Lanni JS, Jacks T . (1998). Characterization of the p53-dependent postmitotic checkpoint following spindle disruption. Mol Cell Biol 18: 1055–1064.
Marshman E, Ottewell PD, Potten CS, Watson AJM . (2001). Caspase activation during spontaneous and radiation-induced apoptosis in the murine intestine. J Pathol 195: 285–292.
Merritt AJ, Allen TD, Potten CS, Hickman JA . (1997). Apoptosis in small intestinal epithelial from p53-null mice. Oncogene 14: 2759–2766.
Morales JC, Franco S, Murphy MM, Bassing CH, Mills KD, Adams MM et al. (2006). 53BP1 and p53 synergize to suppress genomic instability and lymphomagenesis. PNAS 103: 3310–3315.
Morales JC, Xia Z, Lu T, Aldrich MB, Wang B, Rosales C et al. (2003). Role for the BRCA1 C-terminal repeats (BRCT) protein 53BP1 in maintaining genomic stability. J Biol Chem 278: 14971–14977.
Motoyama N, Naka K . (2004). DNA damage tumour suppressor genes and genomic instability. Curr Opin Genet Dev 14: 1–6.
Pryde F, Khalili S, Robertson K, Selfridge J, Ritchie AM, Melton DW et al. (2005). 53BP1 exchanges slowly at the sites of DNA damage and appears to require RNA for its association with chromatin. J Cell Sci 118 (Part 9): 2043–2055.
Rappold I, Iwabuchi K, Date T, Chen J . (2001). Tumour suppressor p53 binding protein 1 (53BP1) is involved in DNA damage-signalling pathways. J Cell Biol 153: 613–620.
Sansom OJ, Clarke AR . (2000). p53 null mice: damaging the hypothesis? Mutat Res 452: 149–162.
Sansom OJ, Reed KR, Hayes AJ, Ireland H, Brinkmann H, Newton IP et al. (2004). Loss of Apc in vivo immediately perturbs Wnt signaling, differentiation, and migration. Genes Dev 18: 1385–1390.
Schultz LB, Chehab NH, Malikzay A, Halazonetis TD . (2000). p53 binding protein 1 (53BP1) is an early participant in the cellular response to DNA double-strand breaks. J Cell Biol 151: 1381–1390.
Sengupta S, Robles AI, Linke SP, Sinogeeva NI, Zhang R, Pedeux R et al. (2004). Functional interaction between BLM helicase and 53BP1 in a Chk1-mediated pathway during S-phase arrest. J Cell Biol 166: 801–813.
Shi Q, King RW . (2005). Chromosome nondisjunction yields tetraploid rather than aneuploid cells in human cell lines. Nature 437: 1038–1042.
Shigeta T, Takagi M, Delia D, Chessa L, Iwata S, Kanke Y et al. (1999). Defective control of apoptosis and mitotic spindle checkpoint in heterozygous carriers of ATM mutations. Cancer Res 59: 2602–2607.
Strathdee G, Sansom OJ, Sim A, Clarke AR, Brown R . (2001). A role of mismatch repair in control of DNA ploidy following DNA damage. Oncogene 20: 1923–1927.
Wang B, Matsuoka S, Carpenter PB, Elledge SJ . (2002). 53BP1, a mediator of the DNA damage checkpoint. Science 298: 1435–1438.
Ward IM, Difilippantonio S, Minn K, Mueller MD, Molina JR, Yu X et al. (2005). 53BP1 cooperates with p53 and functions as a haploinsufficient tumor suppressor in mice. Mol Cell Biol 25: 10079–10086.
Ward IM, Minn K, van Deursen J, Chen J . (2003). p53 binding protein 1 is required for DNA damage responses and tumour suppression in mice. Mol Cell Biol 23: 2556–2563.
Ward IM, Reina-San-Martin B, Olaru A, Minn K, Tamada K, Lau JS et al. (2004). 53BP1 is required for class switch recombination. J Cell Biol 165: 459–464.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Clarke, A., Jones, N., Pryde, F. et al. 53BP1 deficiency in intestinal enterocytes does not alter the immediate response to ionizing radiation, but leads to increased nuclear area consistent with polyploidy. Oncogene 26, 6349–6355 (2007). https://doi.org/10.1038/sj.onc.1210457
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210457
- Springer Nature Limited
Keywords
This article is cited by
-
53BP1 loss induces chemoresistance of colorectal cancer cells to 5-fluorouracil by inhibiting the ATM–CHK2–P53 pathway
Journal of Cancer Research and Clinical Oncology (2017)
-
Growth factors protect intestinal stem cells from radiation-induced apoptosis by suppressing PUMA through the PI3K/AKT/p53 axis
Oncogene (2010)
-
Gender and genetic control of resistance to intestinal amebiasis in inbred mice
Genes & Immunity (2008)