Abstract
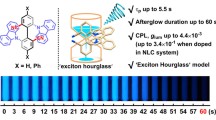
Circularly polarized luminescence (CPL) of chiral organic molecules has attracted attention as next-generation optoelectronic technology, including applications in biotechnology. Higher-order 3D π-extended carbohelicenes would be superb CPL emitters, combining high circular polarization, brightness and stability, but their highly distorted structure has hindered their synthesis and enantiocontrol. Here we report the design of 3D π-extended carbo[11] and [13]helicenes for the CPL emitters and their enantioselective synthesis (up to 87:13 enantiomeric ratio), overcoming high distortions. These helicenes are synthesized via the enantioselective triple [2 + 2 + 2] cycloaddition to construct low-distortion expanded carbo[13] and [15]helicene skeletons and the subsequent π-extension/helix diameter reduction by the Scholl reaction. X-ray crystallographic analyses demonstrate that the 3D π-extended carbo[11] and [13]helicenes with rigid molecular backbones are conglomerates, favouring preferential crystallization, and reveal van der Waals interactions throughout the hexa-peri-hexabenzocoronene layers. Furthermore, their CPL brightness reaches a maximum of 513 M−1 cm−1, the highest value among helicene derivatives.








Similar content being viewed by others
Data availability
The data that support the findings of this study are available in this paper and Supplementary Information (experimental procedures and characterization data). Crystallographic data for the structures reported in this paper have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2303728 [(±)-2a], CCDC 2303729 [(±)-2b], CCDC 2303730 [(M)-3a], CCDC 2303731 [(±)-3b], CCDC 2303732 [(±)-17b], CCDC 2303733 [(±)-23], CCDC 2303737 [(±)-24] and CCDC 2336973 [(±)-16a]. Copies of the data can be obtained free of charge via Cambridge Crystallographic Data Centre at https://www.ccdc.cam.ac.uk/structures/ (ref. 66).
References
Wang, X., Ma, S., Zhao, B. & Deng, J. Frontiers in circularly polarized phosphorescent materials. Adv. Funct. Mater. 33, 2214364 (2023).
Xu, H., Ma, C.-S., Yu, C.-Y., Tong, F. & Qu, D.-H. Reversible inversion of circularly polarized luminescence in a coassembly supramolecular structure with achiral sulforhodamine b dyes. ACS Appl. Mater. Interfaces 15, 25201–25211 (2023).
Schadt, M. Liquid crystal materials and liquid crystal displays. Annu. Rev. Mater. Sci. 27, 305–379 (1997).
Sherson, J. F. et al. Quantum teleportation between light and matter. Nature 443, 557–560 (2006).
Sánchez-Carnerero, E. M. et al. Circularly polarized luminescence from simple organic molecules. Chem. Eur. J. 21, 13488–13500 (2015).
Arrico, L., Di Bari, L. & Zinna, F. Quantifying the overall efficiency of circularly polarized emitters. Chem. Eur. J. 27, 2920–2934 (2021).
Nakai, Y., Mori, T. & Inoue, Y. Theoretical and experimental studies on circular dichroism of carbo[n]helicenes. J. Phys. Chem. A 116, 7372–7385 (2012).
Mori, K., Murase, T. & Fujita, M. One-step synthesis of [16]helicene. Angew. Chem. Int. Ed. 54, 6847–6851 (2015).
Liu, W., Qin, T., Xie, W. & Yang, X. Catalytic enantioselective synthesis of helicenes. Chem. Eur. J. 28, e202202369 (2022).
Wang, Y., Wu, Z.-G. & Shi, F. Advances in catalytic enantioselective synthesis of chiral helicenes and helicenoids. Chem. Catal. 2, 3077–3111 (2022).
Grandbois, A. & Collins, S. K. Enantioselective synthesis of [7]helicene: dramatic effects of olefin additives and aromatic solvents in asymmetric olefin metathesis. Chem. Eur. J. 14, 9323–9329 (2008).
Jančařík, A. et al. Rapid access to dibenzohelicenes and their functionalized derivatives. Angew. Chem. Int. Ed. 52, 9970–9975 (2013).
González-Fernández, E. et al. Enantioselective synthesis of [6]carbohelicenes. J. Am. Chem. Soc. 139, 1428–1431 (2017).
Yamano, R., Shibata, Y. & Tanaka, K. Synthesis of single and double dibenzohelicenes by rhodium-catalyzed intramolecular [2+2+2] and [2+1+2+1] cycloaddition. Chem. Eur. J. 24, 6364–6370 (2018).
Satoh, M., Shibata, Y. & Tanaka, K. Enantioselective synthesis of fully benzenoid single and double carbohelicenes via gold-catalyzed intramolecular hydroarylation. Chem. Eur. J. 24, 5434–5438 (2018).
Jia, S., Li, S., Liu, Y., Qin, W. & Yan, H. Enantioselective control of both helical and axial stereogenic elements though an organocatalytic approach. Angew. Chem. Int. Ed. 58, 18496–18501 (2019).
Yubuta, A. et al. Enantioselective synthesis of triple helicenes by cross-cyclotrimerization of a helicenyl aryne and alkynes via dynamic kinetic resolution. J. Am. Chem. Soc. 142, 10025–10033 (2020).
Guo, S.-M. et al. A C–H activation-based enantioselective synthesis of lower carbo[n]helicenes. Nat. Chem. 15, 872–880 (2023).
Rulíšek, L. et al. On the convergence of the physicochemical properties of [n]helicenes. J. Phys. Chem. C 111, 14948–14955 (2007).
Sehnal, P. et al. An organometallic route to long helicenes. Proc. Natl Acad. Sci. USA 106, 13169–13174 (2009).
Shoji, Y. et al. Boron-mediated sequential alkyne insertion and C–C coupling reactions affording extended π-conjugated molecules. Nat. Commun. 7, 12704 (2016).
Kiel, G. R. et al. Expanded helicenes: a general synthetic strategy and remarkable supramolecular and solid-state behavior. J. Am. Chem. Soc. 139, 18456–18459 (2017).
Nakakuki, Y., Hirose, T. & Matsuda, K. Synthesis of a helical analogue of kekulene: a flexible π-expanded helicene with large helical diameter acting as a soft molecular spring. J. Am. Chem. Soc. 140, 15461–15469 (2018).
Fujise, K., Tsurumaki, E., Wakamatsu, K. & Toyota, S. Construction of helical structures with multiple fused anthracenes: structures and properties of long expanded helicenes. Chem. Eur. J. 27, 4548–4552 (2021).
Suárez-Pantiga, S. et al. In-Fjord substitution in expanded helicenes: effects of the Insert on the inversion barrier and helical pitch. Chem. Eur. J. 27, 13358–13366 (2021).
Zheng, W., Ikai, T., Oki, K. & Yashima, E. Consecutively fused single-, double-, and triple-expanded helicenes. Nat. Sci. 2, e20210047 (2022).
Kiel, G. R. et al. Expanded [23]-helicene with exceptional chiroptical properties via an iterative ring-fusion strategy. J. Am. Chem. Soc. 144, 23421–23427 (2022).
Huo, G.-F. et al. Facile synthesis and chiral resolution of expanded helicenes with up to 35 cata-fused benzene rings. Angew. Chem. Int. Ed. 62, e202218090 (2023).
Toya, M. et al. Expanded [2,1][n]carbohelicenes with 15- and 17-benzene rings. J. Am. Chem. Soc. 145, 11553–11565 (2023).
Kubo, H. et al. Tuning transition electric and magnetic dipole moments: [7]helicenes showing intense circularly polarized luminescence. J. Phys. Chem. Lett. 12, 686–695 (2021).
Donckt, E. V., Nasielski, J., Greenleaf, J. R. & Birks, J. B. Fluorescence of the helicenes. Chem. Phys. Lett. 2, 409–410 (1968).
Sapir, M. & Donckt, E. V. Intersystem crossing in the helicenes. Chem. Phys. Lett. 36, 108–110 (1975).
Dhbaibi, K., Favereau, L. & Crassous, J. Enantioenriched helicenes and helicenoids containing main-group elements (B, Si, N, P). Chem. Rev. 119, 8846–8953 (2019).
Mori, T. Chiroptical properties of symmetric double, triple, and multiple helicenes. Chem. Rev. 121, 2373–2412 (2021).
Chen, X.-Y., Li, J.-K. & Wang, X.-Y. Recent advances in the syntheses of helicene-based molecular nanocarbons via the Scholl reaction. Chin. J. Org. Chem. 41, 4105–4137 (2021).
Zhang, Y., Pun, S. H. & Miao, Q. The Scholl reaction as a powerful tool for synthesis of curved polycyclic aromatics. Chem. Rev. 122, 14554–14593 (2022).
Luo, J., Xu, X., Mao, R. & Miao, Q. Curved polycyclic aromatic molecules that are π-isoelectronic to hexabenzocoronene. J. Am. Chem. Soc. 134, 13796–13803 (2012).
Fujikawa, T., Segawa, Y. & Itami, K. Synthesis, structures, and properties of π-extended double helicene: a combination of planar and nonplanar π-systems. J. Am. Chem. Soc. 137, 7763–7768 (2015).
Hu, Y. et al. Benzo-fused double [7]carbohelicene: synthesis, structures, and physicochemical properties. Angew. Chem. Int. Ed. 56, 3374–3378 (2017).
Zhu, Y. et al. Synthesis and characterization of hexapole [7]helicene, a circularly twisted chiral nanographene. J. Am. Chem. Soc. 140, 4222–4226 (2018).
Nakakuki, Y., Hirose, T., Sotome, H., Miyasaka, H. & Matsuda, K. Hexa-peri-hexabenzo[7]helicene: homogeneously π-extended helicene as a primary substructure of helically twisted chiral graphenes. J. Am. Chem. Soc. 140, 4317–4326 (2018).
Cruz, C. M., Castro-Fernández, S., Maçôas, E., Cuerva, J. M. & Campaña, A. G. Undecabenzo[7]superhelicene: a helical nanographene ribbon as a circularly polarized luminescence emitter. Angew. Chem. Int. Ed. 57, 14782–14786 (2018).
Zhu, Y., Guo, X., Li, Y. & Wang, J. Fusing of seven HBCs toward a green nanographene propeller. J. Am. Chem. Soc. 141, 5511–5517 (2019).
Cruz, C. M. et al. A triskelion-shaped saddle-helix hybrid nanographene. Angew. Chem. Int. Ed. 58, 8068–8072 (2019).
Hu, Y. et al. π-Extended pyrene-fused double [7]carbohelicene as a chiral polycyclic aromatic hydrocarbon. J. Am. Chem. Soc. 141, 12797–12803 (2019).
Guo, X. et al. A nitrogen-doped hexapole [7]helicene versus its all-carbon analogue. Angew. Chem. Int. Ed. 58, 16966–16972 (2019).
Chen, Y. et al. Double π-extended undecabenzo[7]helicene. Angew. Chem. Int. Ed. 60, 7796–7801 (2021).
Yao, X. et al. Synthesis of nonplanar graphene nanoribbon with fjord edges. J. Am. Chem. Soc. 143, 5654–5658 (2021).
Pun, S. H. et al. A near-infrared absorbing and emissive quadruple helicene enabled by the Scholl reaction of perylene. Angew. Chem. Int. Ed. 61, e202113203 (2022).
Xu, X. et al. Synthesis of bioctacene-incorporated nanographene with near-infrared chiroptical properties. Angew. Chem. Int. Ed. 62, e202218350 (2023).
Evans, P. J. et al. Synthesis of a helical bilayer nanographene. Angew. Chem. Int. Ed. 57, 6774–6779 (2018).
Wang, Y. et al. Hexapole [9]helicene. Angew. Chem. Int. Ed. 58, 587–591 (2019).
Liu, B., Bockmann, M., Jiang, W., Doltsinis, N. L. & Wang, Z. Perylene diimide-embedded double [8]helicenes. J. Am. Chem. Soc. 142, 7092–7099 (2020).
Qiu, Z. et al. Amplification of dissymmetry factors in π-extended [7]- and [9]helicenes. J. Am. Chem. Soc. 143, 4661–4667 (2021).
Shen, C. et al. Oxidative cyclo-rearrangement of helicenes into chiral nanographenes. Nat. Commun. 12, 2786 (2021).
Nakakuki, Y. et al. Doubly linked chiral phenanthrene oligomers for homogeneously π-extended helicenes with large effective conjugation length. Nat. Commun. 13, 1475 (2022).
Yang, L. et al. Helical bilayer nonbenzenoid nanographene bearing a [10]helicene with two embedded heptagons. Angew. Chem. Int. Ed. 62, e202216193 (2023).
Ju, Y.-Y. et al. Helical trilayer nanographenes with tunable interlayer overlaps. J. Am. Chem. Soc. 145, 2815–2821 (2023).
Shen, Y.-J. et al. A π-extended pentadecabenzo[9]helicene. Angew. Chem. Int. Ed. 62, e202300840 (2023).
Izquierdo-García, P. et al. Helical bilayer nanographenes: impact of the helicene length on the structural, electrochemical, photophysical, and chiroptical properties. J. Am. Chem. Soc. 145, 11599–11610 (2023).
Morita, F. et al. Regiodivergent synthesis and π-stacking-induced chiral self-recognition of hexabenzocoronene-based [6]helicenes. Eur. J. Org. Chem. 2022, e202200690 (2022).
Stará, I. G. & Starý, I. Helically chiral aromatics: the synthesis of helicenes by [2 + 2 + 2] cycloisomerization of π-electron systems. Acc. Chem. Res. 53, 144–158 (2020).
Tsurusaki, A. & Kamikawa, K. Multiple helicenes featuring synthetic approaches and molecular structures. Chem. Lett. 50, 1913–1932 (2021).
Ding, X.-H. et al. Halogen bonding in the co-crystallization of potentially ditopic diiodotetrafluorobenzene: a powerful tool for constructing multicomponent supramolecular assemblies. Natl Sci. Rev. 7, 1906–1932 (2020).
Duong, S. T. & Fujiki, M. The origin of bisignate circularly polarized luminescence (CPL) spectra from chiral polymer aggregates and molecular camphor: anti-Kasha’s rule revealed by CPL excitation (CPLE) spectra. Polym. Chem. 8, 4673–4679 (2017).
Access structures. Cambridge Crystallographic Data Centre https://www.ccdc.cam.ac.uk/structures/.
Acknowledgements
This work was supported by JSPS KAKENHI grant numbers JP19H00893, JP21K18949 and JP24H00005 to K.T., JP20H04661 and JP22K05032 to H.U., JP23H04020 to G.F. and JP22H00320 and JP22H05125 to M.U. and JST CREST grant number JPMJCR19R2 to M.U. This work was supported by JST, the establishment of university fellowships towards the creation of science and technology innovation, grant number JPMJFS2112 to F.M. We thank Takasago International Corporation for the gift of Segphos and H8-BINAP. A generous allotment of computational resources from TSUBAME (Tokyo Institute of Technology) is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
F.M. designed the project, conducted experimental works and computational studies, and wrote a draft paper. X-ray crystallographic analyses were performed by Y.K., H.S., M.A. and H.U. for (±)-2b, (M)-3a, (±)-3b, (±)-16a and (±)-17b, and Y.S. and J.N. for (±)-2a, (±)-23 and (±)-24. N.T., F.M., Y.N. and M.U. performed CPL measurements. T.K. and G.F. performed fluorescence lifetime measurements. K.T. designed, advised and directed the project, and wrote the paper. All authors edited the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Jeanne Crassous, Xiaoyu Yang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alison Stoddart, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Synthetic experiments, 1H and 13C NMR spectra, chiral high-performance liquid chromatography charts, photophysical and chiroptical properties, crystal data, theoretical calculations and Supplementary Figs. 1–77 and Tables 1–39.
Supplementary Data 1
Crystal data for compound 2a, CCDC 2303728.
Supplementary Data 2
Crystal data for compound 2b, CCDC 2303729.
Supplementary Data 3
Crystal data for compound 3a, CCDC 2303730.
Supplementary Data 4
Crystal data for compound 3b, CCDC 2303731.
Supplementary Data 5
Crystal data for compound 17b, CCDC 2303732.
Supplementary Data 6
Crystal data for compound 23, CCDC 2303733.
Supplementary Data 7
Crystal data for compound 24, CCDC 2303737.
Supplementary Data 8
Crystal data for compound 16a, CCDC 2336973.
Source data
Source Data Fig. 5
Statistical source data. Raw data (.txt) of ultraviolet–visible absorption, fluorescence, ECD and CPL spectra of compounds 2a, 16a and 3a.
Source Data Fig. 6
Statistical source data. Raw data (.txt) of ultraviolet–visible absorption, fluorescence, ECD and CPL spectra of compounds 2b, 17b and 3b.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Morita, F., Kishida, Y., Sato, Y. et al. Design and enantioselective synthesis of 3D π-extended carbohelicenes for circularly polarized luminescence. Nat. Synth (2024). https://doi.org/10.1038/s44160-024-00527-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s44160-024-00527-3
- Springer Nature Limited