Abstract
Coal balls are carbonate concretions that preserve peat in cellular detail. Despite their importance to paleobotany, the salinity of coal-ball peat remains controversial. Pennsylvanian coal balls from the Midland and Illinois basins contain echinoderms and early high-magnesium calcite cement. Echinoderm skeletons reflect the Mg/Ca ratio of the seawater in which they grew. Here we show that well-preserved echinoderms in coal balls and North American Pennsylvanian marine facies have similar average mole % MgCO3; 10.2–12.3 and 9.9–12.5 respectively. Coal-ball echinoderms reflect the magnesium content of the adjacent epicontinental seawater. Early high-magnesium calcite cement in coal balls has the same, or more magnesium than echinoderms from the same deposit, and high Sr/Ca and Na/Ca, consistent with formation in marine or brackish water. Subsequent coal-ball cement is low-magnesium calcite, suggesting freshwater diagenesis and cementation followed formation of marine high-magnesium calcite. Coal balls likely formed in the marine-freshwater mixing zone.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Coal balls are carbonate concretions in coal or wetland sediments that contain permineralized peat1 (Fig. 1a). Most come from Pennsylvanian and Permian swamps; however, the environment of coal-ball formation remains controversial2,3,4,5. Carbon and oxygen stable isotopes appear to support a freshwater origin, along with the habitat of the nearest living relatives of coal-ball plants4,6,7. However, in most coal balls with a known paragenetic sequence, the earliest coal-ball cements are high-magnesium calcite (HMC) or non-stoichiometric dolomite, carbonates that occur commonly in marine depositional environments and rarely in freshwater of normal ionic composition3,4,8,9,10. In dolomitic coal balls, subsequent cements are ferroan dolomites, suggesting diagenesis in anoxic marine environments10.
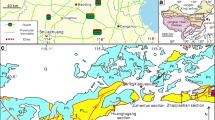
a Late Pennsylvanian (310 Ma) paleogeographic reconstruction of Scotese (ref. 34) showing Pennsylvanian basins with coal balls (white circles). Supplementary Data documents the presence of marine sediments overlying coals with coal balls, or the presence of marine invertebrates in coal balls in all coalball basins except the Stellarton Basin of Maritime Canada (white asterisk). Partial country boundaries and US state boundaries in black. b Late Pennsylvanian (310 Ma) paleogeographic map of North America (ref. 34) showing the location of marine facies as white triangles (ref. 15) and coal balls with echinoderm stereom as black triangles in white circles. Localities: 1— Pittsburg, KS; 2—Dalton coal, Palo Pinto Co., TX; 3—Calhoun Coal, Berryville, IL; 4—Holder Fm., Dry Canyon, NM; 5—Palo Pinto Co., TX.; 6—Pontotoc Co., OK. Partial US state boundaries in black. c Pennsylvanian stratigraphic column with coal-ball (black triangles in white circles) and marine facies (white triangles) localities.
In HMC coal balls, subsequent coal-ball cements are low-magnesium calcite (LMC), suggesting initial cementation in marine water followed by cementation and diagenesis in freshwater2,3,4. Within these coal balls, marine (HMC) and freshwater (LMC) cement intermingle, with LMC rimming individual crystals of HMC cement (Fig. 2). Because of this, all coal-ball isotopic studies have sampled a mix of marine and freshwater cement, although Scott and collaborators reported coal-ball carbon and oxygen stable isotopic ratios consistent with formation from a mixture of marine and fresh pore water2,5.
This plant-only coal ball is from the Williamson No. 3 Deposit, Williamson, Iowa, Pennsylvanian (late Atokan - early Desmoinesian; Midcontinent shelf). Columnar crystals of early HMC cement shown here in cross-section, appear speckled in the BSE image. Vuggy LMC (light grey calcite) surrounding the HMC polycrystals results from the diagenetic alteration of HMC. The small dark grey carbonates are non-stoichiometric dolomite crystals. HMC high-magnesium calcite, LMC low-magnesium calcite, V vug, Dolo non-stoichiometric dolomite. The scale is 30 µm. Image taken by Rachel Wells.
Marine facies overlie nearly all Pennsylvanian coals that contain coal balls1,7,11 (Supplementary Data). In North America, a third of Pennsylvanian transgressive-regressive cycles from the mid-Moscovian through Ghzelian have coals with coal balls1,12,13. Of the coals with coal balls, more than 10% have mixed coal balls, which contain plant debris and marine invertebrate fossils including echinoderms1,12,13.
Echinoderms are marine organisms that secrete HMC skeletons with distinctive 3D microstructure, called stereom (Fig. 3a–c), which reflects the Mg/Ca ratio of the seawater in which they grew14,15. Mg/CaC ratios of echinoderm stereom have been used to reconstruct the Mg/CaSW ratio of paleo-oceans15,16,17. However, the Mg content of modern seawater varies with temperature, salinity and location, particularly in coastal seas, and this method of predicting global Mg/CaSW ratios has been questioned18,19,20. In this contribution, we use the Mg content of echinoderms from the Late Pennsylvanian Midcontinent Sea (LPMS) of North America as a local indicator of the origin of coal-ball cements in two LPMS coals21. Echinoderm fossils in coal balls appear to be stem columnals, probably crinoids. These organisms almost certainly grew in shallow marine environments adjacent to the swamp, and were transported into the swamp where they became incorporated into coal balls. Because of the link between echinoderms and seawater chemistry, echinoderms in coal balls provide new data on pore-water chemistry during coal-ball formation.
100 µm scale. HMC is medium grey, LMC is light grey and Pyrite (P) is white. a Calhoun echinoderm ossicle with stereom microstructure, boxed area enlarged in b. b Calhoun stereom microstructure enlarged from a. Interstereom pores near the lower edge of the image partially filled with black, organic carbon (OC). c Dalton echinoderm ossicle with stereom microstructure. Interstereom pores filled with organic carbon (black) and LMC (light grey). d Pyritized (P, black arrow) benthic foram growing on root parenchyma cells permineralized with LMC cement, Dalton mixed coal ball. e HMC cement in tree-fern root cells, Dalton plant-only coal ball. LMC next to cell walls results from diagenetic alteration of HMC filling cell lumen. f HMC and LMC cement in wood cells, Calhoun plant-only coal ball. LMC surrounding cell walls results from the diagenetic alteration of HMC.
We propose and test three hypotheses. One, echinoderm stereom in coal balls records the Mg content of seawater in the epicontinental sea adjacent to the peat swamp. Echinoderms in coal balls likely resulted from storm transport of marine invertebrate shell hash into the peat swamp during storms or due to marine transgression12,13,22. If coal-ball echinoderms reflect the Mg/Ca ratio of the LPMS, the Mg content of echinoderm stereom in coal balls should match the Mg content of echinoderm stereom in LPMS marine facies reported by Dickson15.
Two, in coal balls with echinoderms and plant-only coal balls from the same locality, early HMC cement formed from marine or brackish pore water should have more Mg than echinoderm stereom. We test this hypothesis by comparing the Mg content of early HMC cement in coal balls to the Mg content of echinoderm stereom in coal balls from the same deposit. Experimental studies show that abiotic cement precipitated from seawater has more Mg than echinoderm stereom grown in seawater of the same composition23. Accordingly, coal-ball cement precipitated from marine or brackish water should have the same or higher Mg content as echinoderm stereom precipitated from seawater of the same composition. Coal-ball cement precipitated from brackish water would have similar amounts, or slightly less Mg than echinoderm stereom. The Sr and Na content of HMC coal-ball cement provides an additional test of this hypothesis. Cements precipitated from either seawater or brackish water have higher Sr/Ca and Na/Ca values than cements precipitated from freshwater24, and should match the ratios of modern marine cement25.
Three, fresh pore water replaced marine pore water relatively early in coal ball formation. We test this hypothesis by determining the Mg, Sr and Na content of second-generation calcite cement permineralizing echinoderm stereom and plant debris in coal balls. HMC cements in coal balls also show evidence of early diagenesis in freshwater3. Coal-ball cement precipitated from freshwater will have a freshwater Mg signature26 (≤5 mole% MgCO3), and low Sr/CaC and Na/CaC27.
In testing our second and third hypotheses, we consider mixed and plant-only coal balls separately. Mixed coal balls, i.e. coal balls with marine invertebrates12, often occur at the top of the coal bed and contain fragmented plant debris rather than rooted peat. These coal balls record the Mg/Ca ratio of peat pore water during marine transgression of coastal swamps, and may not reflect the Mg/Ca ratio of pore water during peat accumulation when plant-only coal balls formed. In both types of coal balls, calcite cement precipitated from seawater should have more Mg than echinoderm stereom, and Sr/CaC and Na/CaC indicative of marine HMC cement17,28. Coal-ball cement precipitated from brackish water may have similar amounts, or slightly less Mg than echinoderm stereom and higher Sr/CaC and Na/CaC than freshwater cement.
Sample locations and stratigraphic context
Coal-balls for this study come from the informally named Dalton coal in Texas (Midland Basin) and the Calhoun Coal of Illinois (Illinois Basin), both Late Pennsylvanian (Missourian) in age (Fig. 1b, c). The Dalton coal of Texas lies within the Wolf Mountain Shale, and belongs to the Wyandotte Intermediate cycle1,29,30. It is underlain by marine shale or thin fossiliferous limestone beds and overlain by calcareous sand or shale31,32. Throughout most of its thickness, the Dalton deposit contains limonite (diagenetically altered siderite) concretions; however, coal balls occur at the top 0.3 m of the bed31. Plants in Dalton coal balls include tree ferns, medullosan seed ferns, cordaiteans, and Calamites. These coal balls also contain a diverse assemblage of marine invertebrates including crinoids, brachiopods, gastropods, bivalves, and encrusting forams (Fig. 3d).
The Late Pennsylvanian (latest Missourian) Calhoun Coal Member from Berryville, Illinois belongs to the Mattoon Formation of the McLeansboro Group32. Overlain by the Omega Limestone Member of the Mattoon Formation, the Calhoun Coal belongs to the Cass cycle30. Coal balls occur throughout the deposit but increase in abundance toward the top12. Their floral assemblage consists of tree ferns with subdominant seed ferns and lycopsids; their invertebrate assemblage consists of crinoids, brachiopods, bivalves, gastropods and conodonts12,32,33.
The Mg content of Missourian crinoids has not been reported, thus we compare the Mg content of coal-ball echinoderms and calcite to the Mg content of echinoderms from the preceding Desmoinesian, and succeeding Virgilian stages15 (Table 1 and Fig. 1c).
The Late Pennsylvanian (310 Ma) paleogeographic reconstruction places Pennsylvanian basins with coal balls between 0° and 5° paleolatitude34 (Fig. 1a).
Results
Echinoderm Mg, Sr and Na Content
Late Pennsylvanian echinoderms from marine facies contain 11.0 mole% MgCO3 averaged by locality15, (range 9.9–12.5 mole% MgCO3; Table 1). Echinoderm fossils from both the Dalton and Calhoun coals preserve stereom microstructure and yield average Mg values within this range (Fig. 3a–c and Table 1). Neither population differs significantly from echinoderms in Pennsylvanian marine facies; however, Calhoun coal-ball echinoderms (av. 12.3 mole% MgCO3) have significantly more Mg than Dalton echinoderms (av. 10.2 mole% MgCO3) (Fig. 4; Supplementary Data). Average Sr/CaC in Dalton and Calhoun coal-ball echinoderms are 1.3 and 1.5 Sr/CaC mmol/mol respectively (Fig. 5a; Table 1). Dalton and Calhoun echinoderm stereom has significantly higher Na/CaC than their interstereom cements (stereom respectively 3.7 and 3.3 Na/CaC mmol/mol; interstereom cement respectively 1.4 and 0.6 Na/CaC mmol/mol; Fig. 6a; Supplementary Data).
a Echinoderms from Pennsylvanian marine facies (ref. 15), Dalton coal, and Calhoun Coal. b Dalton echinoderms and HMC cements from mixed and plant-only coal balls. c Calhoun echinoderms and HMC cements from mixed and plant-only coal balls. For all box plots, the boxes extend from the upper and lower quartiles (interquartile range, Q3 to Q1), the horizontal solid black line is the median. Whiskers extend from boxes out to 1.5 times the interquartile range. Data points are shown in small, black circles. Any data past the ends of the whiskers are outliers and shown as large, black circles.
Sr/CaC and Mg/CaC for coal-ball and echinoderm calcite are shown in comparison to modern marine cement (ref. 25). Dashed line at 55 Mg/Ca separates LMC values to the left and HMC values to the right. Calhoun values are green and Dalton values are orange. a Coal-ball echinoderms and interstereom cement in mixed echinoderm-plant coal balls (circles) Mg/CaC of Pennsylvanian marine echinoderms (blue bar) (ref. 15). b Plant calcite in mixed echinoderm-plant coal balls (diamonds). c Plant calcite in plant-only coal balls (triangles). All ratios were reported in mmol/mol.
The dashed line at 55 Mg/Ca separates LMC values to the left and HMC values to the right. Calhoun values are green and Dalton values are orange. a Coal-ball echinoderms and interstereom cement in mixed echinoderm-plant coal balls (circles) Mg/CaC of Pennsylvanian marine echinoderms (blue bar) (ref. 15). b Plant calcite in mixed echinoderm-plant coal balls (diamonds). c Plant calcite in plant-only coal balls (triangles). All ratios were reported in mmol/mol.
For individual ossicles from Dalton coal balls, the average range in Mg/CaC analytic point values (i.e. the difference between the highest and lowest Mg/CaC analytic points measured on each ossicle) is 35 Mg/CaC mmol/mol (N = 12 ossicles; Table 2). For individual ossicles from Calhoun coal balls, the average range in analytic point values is 50 Mg/CaC mmol/mol (N = 14 ossicles; Table 2). The range in average Mg/CaC mmol/mol per ossicle for the Dalton and Calhoun coals are respectively 21 and 68.
Mg, Sr and Na in early HMC and second-generation LMC cement of coal balls
Early HMC cement in Dalton mixed coal balls has significantly more Mg than echinoderm stereom from the same coal balls, ~1.9 mole% MgCO3. However early HMC cement in Dalton mixed coal balls and Dalton echinoderm stereom contain similar amounts of Mg (Table 1 and Figs. 3e, 4, 5a, b). In the Calhoun, early HMC cement in both mixed and plant-only coal balls has significantly more Mg than echinoderms, respectively 2.5 and 3.5 mole% MgCO3 (Table 1 and Figs. 3f, 4, 5). In each deposit, echinoderm stereom has significantly more Sr than early HMC cement from both types of coal balls (Table 1 and Fig. 5). Early HMC cement in mixed and plant-only coal balls has significantly higher Na than second-generation LMC (Fig. 6b, c; Supplementary Data).
LMC, most ferroan, some non-ferroan, permineralizes echinoderm stereom in Dalton and Calhoun coal balls (Fig. 3). At each locality, second-generation LMC cement associated with echinoderms and plant debris has a similar amount of Mg (Table 1 and Fig. 5). However, second-generation LMC in plant debris from Dalton plant-echinoderm coal balls has much more Sr than other second-generation LMC (Table 1 and Fig. 5).
Echinoderm seawater Mg/CaSW ratios
The predicted Mg/CaSW ratio of Pennsylvanian seawater based on echinoderms in coal balls is 2.8–3.6 mol/mol for the Dalton coal in the Midland Basin and 3.4–4.2 mol/mol for the Calhoun Coal in the Illinois Basin15 (Table 1).
Discussion
Echinoderms in coal balls
As previously discussed, storms likely transported echinoderm ossicles into the swamp, where they became incorporated into coal balls. The depositional setting and processes that led to the inclusion of echinoderm ossicles in Pennsylvanian coal balls may be similar to the ones that led to the inclusion of Cretaceous crinoids in Burmese amber35. Cementation of the stereom probably occurred in the swamp. The interstereom cement matches the composition of the second generation of coal-ball cement, with the exception of high Sr/CaC, which may be due to aragonite dissolution in fresh pore water. Further, echinoderms in both deposits show evidence of infiltration by peat organic fluids into the interstereom pore space prior to cementation (Figs. 3a–c and 7d). These ossicles have coal-filled interstereom pores. The LMC cement of other coal-ball echinoderm ossicles has void spaces, also suggesting the presence of peat organic fluid in the interstereom pores prior to cementation (Figs. 3a–c and 7c). Coal-ball ossicles frequently have borings, and ossicle fragments in coal balls may result from the breakage of bored ossicles (Fig. 7b, d). Nonetheless, some echinoderm ossicles in coal balls have diagenetic histories similar to those preserved in marine facies. For example, a number of coal-ball ossicles have Dickson’s type 1 preservation on the edge, and type 2 in the middle15 (Fig. 7e). Although we use analytical points collected from the edge of these ossicles, we did not use analytical points from regions with type 2 preservation (Figs. 3a–c and 7).
Each image is from an echinoderm used in this study unless noted. a Well-preserved Calhoun ossicle showing HMC stereom in medium grey and LMC interstereom cement in light grey. The scale is 100 μm. b Relatively well-preserved Dalton ossicle fragment outlined in white, surrounded by LMC coal-ball cement at the top of the image. HMC stereom medium grey, LMC interstereom cement light grey, pyrite (P) white. The scale is 200 μm. c Relatively well-preserved Calhoun ossicle fragment outlined in white surrounded by LMC coal-ball cement. HMC stereom medium grey, LMC interstereom cement light grey, V, vugs in LMC interstereom cement. The scale is 200 μm. d Broken Dalton ossicle cut by gypsum vein (Gyp), not used in this study. HMC stereom (medium grey) filled with organic carbon (black), pyrite (P) (white) and small carbonate grains. The lower cross shows the location of EDS (energy-dispersive spectroscopy) analysis indicating HMC stereom. The upper cross shows the location of EDS analysis of a second ossicle fragment indicating HMC stereom. Strontium sulfate (Cel) dark grey. Scale is 300 μm. e Calhoun ossicle with Dickson type 1 (Gorzelak type 0) preservation on the rim and Dickson type 2 (Gorzelak type 2) preservation in the middle, HMC stereom medium grey, LMC interstereom cement light grey, dolomite (Dolo) dark grey (refs. 15,18). Scale is 200 μm; f Bored Dalton ossicle with stereom not used in this study. White Arrow indicates HMC stereom, cross shows location of EDS analysis indicating HMC stereom. The scale is 300 μm.
Mg, Sr and Na in LPMS echinoderms
The echinoderms from marine facies used in this study come from the LPMS15 and contain 9.9–12.5 mole% MgCO3 (Table 1 and Fig. 1b). The amount of Mg incorporated into HMC increases with temperature and decreases with freshwater influx14,19. Variations in temperature, depth and proximity to the shoreline, as well as diet and vital effects, could contribute to this range in Mg content18,36,37,38.
The average Mg content of both Dalton and Calhoun echinoderms is statistically indistinguishable from values reported for echinoderms from Pennsylvanian LPMS marine facies15 (Table 1). This result supports our first hypothesis, echinoderms in coal balls retain the Mg/Ca ratio of the seawater in which they grew, even when transported to, and preserved in, peat swamps. However, Calhoun echinoderms from the Illinois Basin contain significantly more Mg than Dalton echinoderms from the Midland Basin (Figs. 1b, 4, 5a and Table 1). Vital effects or environmental variation tied to basin depth, evaporation and higher water temperature could contribute to more Mg in Calhoun echinoderms14,18,19,36,37,38.
Samples of echinoderm ossicles from a single locality yield similar ranges in Mg/CaC (~21–85 mmol/mol), regardless of geological age (Table 2). The Dalton coal has a narrow range in average ossicle Mg/CaC, 21 mmol/mol, whereas the Calhoun Coal and samples of Recent crinoids from the Bahama Banks and Suruga Bay, Japan have larger ranges in average ossicle Mg/CaC between 65–85 mmol/mol39 (Table 2). Data from Recent crinoids suggest that we should expect a wide range in estimates of ancient seawater Mg/Ca based on the Mg/CaC of echinoderm stereom.
One modern crinoid ossicle yields a wide estimated range of Mg/CaC values (154 mmol/mol Mg/CaC, 1.44 wt% Mg), possibly due to variations in the composition of the associated organic matrix37,39. Because very few fossil crinoids retain biomolecules40,41, the range of Mg/CaC values observed in Dalton and Calhoun ossicles probably results from both diagenesis and the vital effects rather than variation in the composition of the associated organic matrix. Both freshwater and marine diagenesis of HMC results in loss of Mg and Sr ions, producing LMC42. Thus, Mg/CaC values of ancient echinoderm stereom likely underestimate the Mg/CaSW of the water in which the stereom formed.
Variable Mg/CaC values in fossil echinoderms are consistent with BSE images of ancient echinoderm stereom that show a mottled pattern of darker (>Mg) and lighter (<Mg) carbonates43 (Fig. 3b, c). Modern echinoderm stereom has narrow edges of lower Mg carbonate 1–2 μm wide adjacent to the interstereom pores and fairly uniform Mg content in the middle18,43. Dickson hypothesized that early interstereom cement acted as a ‘crystal casket’, inhibiting loss of Mg ions from HMC stereom43. Although HMC calcite readily transforms into dolomite and LMC, Dickson proposed that the Mg content of the original HMC could be modeled if the volume and Mg/Ca of the dolomite and calcite were known43,44. Nonetheless, we did not collect points from stereom with nearby non-stochiometric dolomite crystals.
The average Sr/CaC mmol/mol of coal-ball echinoderms, 1.34 for in the Dalton and 1.54 in the Calhoun, is significantly higher than the Sr/Ca ratio of early HMC cement in mixed and plant-only coal balls (Table 1; Fig. 6a; Supplementary Data). High rates of stereom formation relative to abiotic precipitation rates may facilitate the incorporation of minor and trace elements in skeletal calcite24,45. Modern echinoderms have higher Sr/CaC than LPMS echinoderms, ranging from 1.6 in echinoids from the Arctic and the NW Atlantic to nearly 5 in holothurians from the NE Atlantic46,47. Although coal-ball echinoderms have significantly higher Na/CaC than their interstereom cement, coal-ball echinoderms have low Na/CaC compared to modern echinoderms. Whereas Dalton and Calhoun echinoderms have average Na/CaC of 3.7 and 3.3 mmol/mol, respectively, Na/CaC of modern Arctic echinoderms ranges from 57 to 93 mmol/mol47. Loss of Na during burial diagenesis may account for low Na/CaC ratios in Pennsylvanian echinoderms48.
Early HMC cements
The echinoderms in Dalton and Calhoun coal balls likely grew in marine, shallow-shelf communities adjacent to the peat swamp and washed on to the peat surface during storms. Well-preserved stereom in coal-ball echinoderms reflects the Mg/Ca of the LPMS seawater in which they grew. The Mg/CaC of early HMC cement in Dalton and Calhoun coal balls suggests this cement formed from marine pore water49 (Fig. 5b, c and Table 1). This HMC cement has higher Mg/CaC than well-preserved echinoderm stereom from the same coal balls, consistent with echinoderm growth and coal-ball formation from marine water with similar Mg/CaSW (Figs. 4, 5 and Table 1). The relationship between Mg/CaC in early HMC coal-ball cement and echinoderm stereom from the same coal balls (higher in coal balls, lower in echinoderms) supports our second hypothesis, the early HMC cement of coal balls formed from marine pore water. In the Calhoun deposit, both mixed and plant-only coal balls likely formed from marine pore water (Figs. 4, 5 and Table 1). In the Dalton deposit, mixed coal balls probably formed from marine water, whereas plant-only coal balls, which have HMC cement with Mg/CaC matching that of Dalton echinoderms, likely formed from brackish water (Figs. 4, 5 and Table 1). Nonetheless, both types of Dalton coal balls yield similar predicted seawater Mg/CaSW (Table 1).
Early HMC cements in coal balls have been attributed to microbial precipitation of HMC in freshwater peat due to carbonate springs4. However, with averages of 10.5–15.8 mole% MgCO3, the early HMC cements of Dalton and Calhoun coal balls have far more Mg than most freshwater calcite (≤5 mole% MgCO3; ref. 26), with the exception of HMC formed in hypersaline, alkaline lakes, or lakes in carbonate terrains with seasonally dry climate50,51,52. None of these modern lakes is currently the site of peat accumulation. Further, the columnar crystal habit of early HMC cement in all coal balls suggests an abiotic origin4,53.
Second-generation LMC cements
Second-generation carbonate cement permineralizing echinoderm stereom and plants in coal balls is usually ferroan LMC. This cement may indicate the presence of fresh, low-Mg pore water in the wetland during coal-ball formation3,5, supporting our third hypothesis, fresh pore water replaced marine pore water relatively early in coal-ball formation. This would place the environment of coal-ball formation in the marine/freshwater mixing zone2 (Table 1). LMC in coal balls results from both cementation and diagenesis of HMC polycrystals3 (Figs. 2 and 3d). A coal ball from the Herrin Coal in the Illinois Basin shows evidence of three generations of early HMC cement, separated by thin layers of ferroan, LMC cement4, suggesting that fresh pore water replaced marine pore water twice during its formation.
Low Na/CaC values in second-generation LMC cement also support a freshwater origin for this calcite, or freshwater diagenesis of marine carbonate (Fig. 6). Experimental results suggest that the Na content of calcite increases rapidly with increasing salinity of the aqueous solution up to ~10 psu54. Above 10 psu, the Na content of calcite increases slowly with increasing salinity54. In addition to the Na content of water, the growth rate of calcite crystals also influences their Na/Ca ratio, such that rapidly growing crystals have higher Na/CaC than slowly growing crystals in seawater of the same salinity55. Thus while calcite with high Na/CaC ratios indicates precipitation in brackish or marine water, Na/CaC values do not allow precise salinity predictions within this range54,55. Likewise, freshwater diagenesis of marine carbonate also decreases its Na content27,56, which may account for low Na/CaC values in second-generation LMC.
Aragonite dissolution during coal-ball formation
The Sr/CaC in coal-ball cements may reflect aragonite dissolution during coal-ball formation. Aragonite formed in equilibrium with seawater contains ~10,000 ppm Sr; whereas calcite formed in equilibrium with seawater contains ~1000 ppm Sr27. Thus, high Sr/CaC ratios in calcite cement may signal precipitation in the presence of dissolving aragonite49. Plant-only coal balls from the Dalton and Calhoun localities show no evidence of aragonite dissolution during their formation. In these coal balls, both the early HMC cement and the second-generation LMC have modest Sr/CaC, ranging below the detection limit (0.02 wt% Sr) to 2.2 mmol/mol (Fig. 6c).
Mixed coal balls from both deposits show an interesting pattern of Sr/CaC. In both, early HMC cement has modest Sr/CaC, ranging from 0.1 mmol/mol, which is above the detection limit for Sr, to 1.6 mmol/mol, suggesting no dissolution of aragonite occurred during the formation of this cement, consistent with precipitation from marine pore water (Fig. 6b). However, LMC cement from both deposits has elevated Sr/CaC, slightly so in the case of Calhoun (0.7–3.1 mmol/mol; Fig. 6b), greatly so in the case of the Dalton (0.3–6.1 mmol/mol; Fig. 6b). Elevated Sr/CaC values in second-generation LMC cement, may indicate aragonite dissolution in the swamp during precipitation of LMC cement. Both processes could result from the presence of fresh pore water in the swamp. All the LMC cement in Calhoun mixed coal balls (i.e. LMC cement in plant debris and the interstereom pores of ossicles) has a similar range of Sr/CaC (Fig. 6b). However, in Dalton mixed coal balls, a single sample of LMC from the interstereom pore space has elevated Sr/CaC (Fig. 6b).
Elevated Sr/CaC (>1.5 mmol/mol) in second-generation LMC may indicate the presence of marine aragonite in the swamp prior to coal-ball formation even in the absence of marine fossils. Many discussions of coal-ball formation suggested that the dissolution of marine carbonate shells contributed to the formation of coal balls5,6,12,57,58. Sr/CaC in coal-ball cement provides a test of this hypothesis.
Late Pennsylvanian midcontinent seawater Mg/CaSW ratios
All of the echinoderm fossils that Dickson used to predict Pennsylvanian Mg/CaSW came from the LPMS, and Pennsylvanian echinoderms from LPMS coal balls yield significantly similar Mg/CaSW values, suggesting that fossil echinoderms from the same time interval and epicontinental sea may enable us to trace relative changes in Phanerozoic Mg/CaSW15 (Table 1; Supplementary data). However, echinoderm stereom and coal-ball HMC cement from the Midland Basin (Dalton coal) contain significantly less Mg than comparable populations from the Illinois Basin (Calhoun Coal; Table 1, Supplementary Data). Vital effects or differences in water depth leading to more evaporation, or higher water temperature could lead to more Mg in HMC from the Illinois Basin. Understanding whether we can use fossil echinoderms to track the history of Mg/Ca in seawater may require studies that incorporate multiple samples from the same basin, and which track Mg/CaC in more than one basin from each time interval21.
Coal-ball carbonates as environmental archives
Coal-ball cements are environmental archives of wetland conditions during formation and early diagenesis; as for all carbonates, the trace and minor elemental composition of coal-ball cement provides clues to pore water salinity and oxygen availability during carbonate precipitation and diagenesis3,10. Stopes and Watson were among the first to propose that Pennsylvanian coal balls with HMC or dolomite cement formed in marine swamps3,8,10. The relative Mg/CaC values of early HMC cement and echinoderm fossils in Dalton and Calhoun coal balls are consistent with the formation of these coal balls in marine or brackish swamps. In all HMC or dolomitic coal balls, the first carbonate cements formed in oxic environments, whereas second-generation cements usually formed in anoxic conditions3,4,10.
The presence of marine sediments overlying nearly all coal beds that contain coal balls is consistent with their formation in marine peat (dolomitic coal balls) or in marine or brackish peat, close to the marine-freshwater mixing zone (early HMC, second-generation LMC coal balls; Supplementary Data). However, more paragenetic sequence data are needed to determine whether this is a widespread pattern. Coal balls with LMC or siderite as the first widespread carbonate cement, may have formed in freshwater. For example, sideritic coal balls from the Foord seam in the Stellarton Basin of Canada, overlain by lacustrine sediments, may have formed in freshwater swamps59,60,61,62. The first widespread carbonate cement in these coal balls is low-Mg siderite, indicative of anoxic, freshwater environments59,62. Subsequent generations of carbonate cement are Mg-carbonates, dolomite and ankerite, although the source of the Mg-laden pore water in a lacustrine basin remains unexplored62.
Retallack11 described carbonate nodules with permineralized plants and freshwater snails from the Hitchcox limey (fen) peat on the south coast of Australia. These nodules formed in Recent peat that has accumulated behind coastal dunes, underlain by Miocene limestone and limestone soils on the landward side. The Miocene limestone and carbonate nodules from this site reveal similar stable isotopic patterns, consistent with both pedogenic carbonate and freshwater diagenesis of marine limestone11. The carbonate mineralogy and paragenetic sequence of these nodules, which would enable us to test hypotheses of their formation, has not been reported11.
Although coal balls with early HMC or dolomite cement indicate the presence of marine or brackish peat swamps, even in coals with coal balls, their occurrence is sporadic1. Most Late Paleozoic paleotropical coal accumulated in freshwater. Nonetheless, our results suggest that equatorial mangrove swamps may be a more appropriate environmental analogue for coal-ball peat than tropical freshwater swamps. This idea has implications for our understanding of late Paleozoic land-plant communities. Whereas freshwater peat swamps are among the least productive land-plant communities in the tropics today, mangroves are among the most63,64,65,66. In addition to gradients in water depth, nutrients and climate, gradients in salinity probably affected species distributions in coal balls. If coal balls preserve brackish and marine plant communities, yet most Late Paleozoic paleotropical coal formed from freshwater peat, this may help to explain why few coal-ball communities match the palynomorph diversity of their source coal67,68,69,70.
Conclusions
The Mg content of early HMC cement in Dalton and Calhoun coal balls (8.8–17.8 mole% MgCO3) matches or exceeds the Mg content of echinoderm ossicles from the same localities (9.4–13.2 mole% MgCO3), suggesting that coal-ball formation began in marine or brackish water. Echinoderm stereom in coal balls is permineralized by ferroan and non-ferroan LMC, suggesting that second-generation coal-ball calcite formed from fresh pore water, usually anoxic but occasionally oxic, confirming freshwater presence in the swamp relatively early in coal-ball formation, and supporting coal-ball formation close to the peat surface. Coal balls likely formed in coastal swamps, in the mixing zone between marine and fresh pore water. Elevated Sr levels in the second-generation LMC of plant-echinoderm coal balls may derive from dissolution of aragonitic shell material and sediment, and could be a way to trace the presence of marine aragonite during coal-ball formation.
Methods
Sample preparation and structural analysis
Initially, we characterised samples using optical and electron (SEM/EDS) microscopy. In order to obtain Mg/CaC ratio data, we sliced Dalton and Calhoun coal balls and prepared thin sections from these slices. We identified echinoderm ossicles in thin sections using a Zeiss Axioplan 2 microscope and attached AxioCam HRc digital camera. We used a Phenom XL scanning electron microscope (SEM) equipped with energy-dispersive spectroscopy (EDS) to acquire secondary- and backscattered-electron (BSE) images, as well as EDS spectra on selected calcites (typical operating conditions consisted of an accelerating voltage of 15 kV and a working distance of 4 to 8 mm). The images and spectra enabled us to identify targets for electron microprobe (EMP) analysis.
Echinoderm ossicles in coal balls often display patchy preservation, with areas of well-preserved stereom composed of HMC and poorly-preserved areas with indistinct stereom (Figs. 3a–c and 7). All echinoderm analytical points came from stereom with type 1 diagenetic transformation, in which stereom may appear speckled in backscattered-electron (BSE) images due to the formation of dolomite microcrystals, but is distinct from the permineralizing cement18,43 (Figs. 3a–c, 7). This preservation type encompasses the type 0 and 1 transformations of Gorzelak and collaborators18. Early HMC cement in coal balls displays a similar speckled texture in BSE images3 (Fig. 2).
Electron microprobe (EMP) geochemical analysis
We perform chemical characterisation of carbonates using wavelength dispersive (WDS) analyses with a Cameca SX-5, housed in the Texas A&M - the Materials Characterization Facility (MCF) Core Facility (RRID:SCR_022202). These analyses allow us to characterise the Calcium, Magnesium, Strontium, Iron, Sodium, Manganese, Sulfur, Silicon and Aluminum contents of echinoderm ossicles and plant cements. Operating conditions consisted of an accelerating voltage of 15 kV and a beam current of 10 nA. The diameter of the electron beam at the sample (spot size) was 5 μm, and counting times ranged from 20–30 s for most elements and 40 s for strontium. Detection limits are Mg – 200 ppm, Sr – 260 ppm, Fe – 400 ppm, and Mn – 360 ppm. We used natural and synthetic mineral standards for calibration and time-dependent intensity (TDI) corrections were applied to standards and unknowns71.
EMP characterisation of calcite chemistries targets echinoderm ossicles with preserved stereom microstructure in polished, carbon-coated thin sections: all echinoderm analytical points in this study come from areas of the ossicle with clear stereom microstructure (Figs. 3a–c and 7). We analyze HMC cement and echinoderm ossicles from the same coal ball to create a dataset of paired echinoderm ossicle and coal-ball HMC cement mole% MgCO3 values. Following previous work15, in mixed coal-balls from each locality, we choose 12–15 ossicles, collecting 3–12 analytical points from each ossicle (seven ossicles with 3–12 data points for the Dry Canyon, NM crinoid study43). In the same coal balls from each locality, we identify 6–8 plant fragments and collect 2–12 analytical points from each, depending on the size of the fragment. In mixed coal balls, we target Psaronius (tree-fern) root fragments and small pieces of wood for microprobe analysis of calcite cement. In plant-only coal balls from each locality, we identify 10–12 locations and collect 2–10 analytical points from each. In plant-only coal balls, we targeted roots and void spaces between pieces of plant debris for microprobe analysis of calcite cement. Typically, analytic points were selected based on greyscale BSE images where HMC and LMC were identified respectively as “dark” and “light”. Early HMC cements in coal balls are polycrystals that display similar speckled texture in BSE images (Fig. 3). Ca, Mg, Sr and Na elemental data (wt%) appear in the Supplementary Data.
We discarded analytic points with weight percent (wt%) totals outside the 98–102% window71,72,73. We also discarded analytic points in echinoderm stereom and early HMC cement with >5000 ppm Fe (>0.5 wt% Fe). Modern echinoderm skeletons have from 100–2300 ppm Fe; the crinoid Antedon mediterranea has from 800–1800 ppm Fe46. Likewise, the early HMC cement of coal balls is non-ferroan3,4. More than 5000 ppm Fe in either type of HMC signals diagenetic alteration, which may have affected the Mg/CaC74.
Mg/Ca ratio nomenclature
We follow previous workers in reporting the Mg/Ca ratio of seawater (Mg/CaSW) as a molar (mol/mol) ratio15,17,18,21,23,28,75. However, methods of reporting the Mg/Ca ratio of calcite (Mg/CaC) vary. Here, we report the Mg content of calcite both as mole% MgCO3, to facilitate comparisons to previous work by Dickson15, and as the ratio of mmol Mg to mol Ca (Mg/CaC) to simplify comparison of our results to those of other workers19,49. We report mole% MgCO3, Mg/CaSW (mmol/mol), Sr/CaC (mmol/mol), and Na/CaC (mmol/mol), in Table 1 and Supplementary Data.
A number of definitions have been proposed for LMC and HMC, LMC <4 mol % MgCO3 or LMC <5 mol % MgCO3, and HMC >8 mol % MgCO324,26,76. Because mol % MgCO3 values for calcite depend on knowing the concentrations of other important minor and trace elements in calcite, these do not easily translate into Mg/Ca mmol/mol values. For the purpose of this contribution, we consider Mg/Ca ≥55 mmol/mol as HMC.
Statistical analysis
We test for statistically significant differences in Mg and Sr content among sample populations of echinoderm stereom using the Mann–Whitney U-test77. To establish the Mg/CaC content of mid-to-late Pennsylvanian echinoderms from the LPMS, we use four locality averages (Pittsburg, KS; Pontotoc Co., OK; Palo Pinto Co., TX; Holder Fm., Dry Canyon, NM; Fig. 1b, c) from existing Pennsylvanian echinoderm stereom data15. We determine coal-ball ossicle averages for both the Dalton coal and Calhoun Coal separately. For each coal-ball locality, we test for statistically significant differences and similarities in Mg, Sr and Na among three populations of HMC (stereom in mixed coal balls, and early HMC cement in mixed and plant-only coal balls), and three populations of second-generation calcite (in echinoderm stereom, and in plant debris from mixed and plant-only coal balls). A summary of results appears in the Supplementary Data.
Mg seawater partitioning
We use the partition coefficient DCMg = 0.03757 for echinoderms15, and power partition functions Mg/CaC = 0.0516Mg/CaSW0.668 for echinoderms and Mg/CaC = 0.0482Mg/CaSW0.898 for calcite cement28, to calculate the Mg/CaSW ratio of Late Pennsylvanian seawater on the Midcontinent Shelf and in the Illinois Basin from HMC in echinoderms and coal-ball cement75. Results for Mg/CaSW (mol/mol) estimates appear in Table 1 and Supplementary Data.
Data availability
Data for this study are accessible at figshare: https://doi.org/10.6084/m9.figshare.22812749.
References
Raymond, A., Lambert, L. L. & Costanza, S. H. Are coal balls rare? A cyclostratigraphic analysis of coal-ball occurrence in North America. Int. J. Coal Geol. 206, 65–79 (2019).
Scott, A. C., Mattey, D. P. & Howard, R. New data on the formation of Carboniferous coal balls. Rev. Palaeobot. Palynol. 93, 317–331 (1996).
Raymond, A., Guillemette, R., Jones, C. P. & Ahr, W. M. Carbonate petrology and geochemistry of Pennsylvanian coal balls from the Kalo Formation of Iowa. Int. J. Coal Geol. 94, 137–149 (2012).
Siewers, F. D. & Phillips, T. L. Petrography and microanalysis of Pennsylvanian coal-ball concretions (Herrin Coal, Illinois Basin, USA): Bearing on fossil plant preservation and coal-ball origins. Sediment. Geol. 329, 130–148 (2015).
Breecker, D. O. & Royer, D. L. The pedogenic formation of coal balls by CO 2 degassing through the rootlets of arborescent lycopsids. Am. J. Sci. 319, 529–548 (2019).
Anderson, T. F., Brownlee, M. E. & Phillips, T. L. A stable isotope study on the origin of permineralized peat zones in the Herrin Coal. J. Geol. 88, 713–722 (1980).
Demaris, P. J. Formation and distribution of coal balls in the Herrin Coal (Pennsylvanian), Franklin County, Illinois Basin, USA. J. Geol. Soc. 157, 221–228 (2000).
Stopes, M. C. & Watson, D. M. S. On the present distribution and origin of the calcareous concretions in coal seams, known as “Coal Balls”. Philos. Trans. R. Soc. Lond. Ser. B Contain. Pap. Biol. Character 200, 167–218 (1909).
Garrels, R. & Wollast, R. Equilibrium criteria for two-component solids reacting with fixed composition in an aqueous phase; example, the magnesian calcites; discussion. Am. J. Sci. 278, 1469–1474 (1978).
Richter, D. K. et al. First description of Phanerozoic radiaxial fibrous dolomite. Sediment. Geol. 304, 1–10 (2014).
Retallack, G. Modern analogs reveal the origin of Carboniferous coal balls. Palaeogeogr. Palaeoclimatol. Palaeoecol. 564, 110185 (2021).
Mamay, S. H. & Yochelson, E. L. Occurrence and Significance of Marine Animal Remains in American Coal Balls. U.S. geol. Surv. Prof. Pap. 354, 193–224 (1962).
Perkins, T. W. Textures and conditions of formation of Middle Pennsylvanian coal balls, central United States. Univ. Kans. Paleontol. Contrib. 82, 1–13 (1976).
Mackenzie, F. T. et al. in Carbonates (ed. Reeder, R. J.) Ch. 4 (De Gruyter, 1983).
Dickson, J. A. D. Echinoderm skeletal preservation: calcite-aragonite seas and the Mg/Ca ratio of phanerozoic oceans. J. Sediment. Res. 74, 355–365 (2004).
Dickson, J. A. D. Paleozoic Mg calcite preserved: Implications for the Carboniferous ocean. Geology 23, 535 (1995).
Dickson, J. A. D. Fossil echinoderms as monitor of the Mg/Ca ratio of Phanerozoic oceans. Science 298, 1222–1224 (2002).
Gorzelak, P., Krzykawski, T. & Stolarski, J. Diagenesis of echinoderm skeletons: constraints on paleoseawater Mg/Ca reconstructions. Glob. Planet. Change 144, 142–157 (2016).
Lebrato, M. et al. Global variability in seawater Mg:Ca and Sr:Ca ratios in the modern ocean. Proc. Natl. Acad. Sci. USA 117, 22281–22292 (2020).
Gorzelak, P. et al. A Devonian crinoid with a diamond microlattice. Proc. R. Soc. B Biol. Sci. 290, 20230092 (2023).
Kołbuk, D., Dubois, P., Stolarski, J. & Gorzelak, P. Impact of seawater Mg2+/Ca2+ on Mg/Ca of asterozoan skeleton – Evidence from culturing and the fossil record. Chem. Geol. 584, 120557 (2021).
Moore, L. R. Some Sediments Closely Associated with Coal Seams in Coal and Coal-Bearing Strata (eds. Murchison, D. G. & Westoll, T. S.) 105–123 (Oliver and Boyd, 1968).
Ries, J. B. Review: geological and experimental evidence for secular variation in seawater Mg/Ca (calcite-aragonite seas) and its effects on marine biological calcification. Biogeosciences 7, 2795–2849 (2010).
Carpenter, S. J. & Lohmann, K. C. Sr/Mg ratios of modern marine calcite: Empirical indicators of ocean chemistry and precipitation rate. Geochim. Cosmochim. Acta 56, 1837–1849 (1992).
Major, R. P. & Wilber, R. J. Crystal habit, geochemistry, and cathodoluminescence of magnesian calcite marine cements from the lower slope of Little Bahama Bank. Geol. Soc. Am. Bull 103, 461–471 (1991).
Burton, E. A. & Walter, L. M. Relative precipitation rates of aragonite and Mg calcite from seawater: temperature or carbonate ion control? Geology 15, 111 (1987).
Veizer, J. Chemical diagenesis of carbonates: theory and application of trace element technique, in Stable Isotopes in Sedimentary Geology SEPM Short Course Notes (Eds. Arthur, M. A., Anderson, T. F., Kaplan, I. R., Veizer, J. & Land. L. S.) 10, 3.1–3.100 (SEPM, 1983).
Ries, J. B. Effect of ambient Mg/Ca ratio on Mg fractionation in calcareous marine invertebrates: a record of the oceanic Mg/Ca ratio over the Phanerozoic. Geology 32, 981 (2004).
Boardman, D. R. & Heckel, P. H. Glacial-eustatic sea-level curve for early Late Pennsylvanian sequence in north-central Texas and biostratigraphic correlation with curve for midcontinent North America. Geology 17, 802 (1989).
Heckel, P. H. Pennsylvanian stratigraphy of Northern Midcontinent Shelf and biostratigraphic correlation of cyclothems. Stratigraphy 10, 3–39 (2013).
Lowenstein, G. R. The Environment of Deposition of the Dalton Coal (Upper Pennsylvanian), Palo Pinto Co., TX (Texas A&M University, 1986).
Willard, D. A., Phillips, T. L., Lesnikowska, A. D. & DiMichele, W. A. Paleoecology of the Late Pennsylvanian-age Calhoun coal bed and implications for long-term dynamics of wetland ecosystems. Int. J. Coal Geol. 69, 21–54 (2007).
Phillips, T. L. Stratigraphic occurrences and vegetational patterns of Pennsylvanian pteridosperms in Euramerican coal swamps. Rev. Palaeobot. Palynol. 32, 5–26 (1981).
Scotese, C. PALEOMAP PaleoAtlas for GPlates and the PaleoData Plotter Program, PALEOMAP Project. https://doi.org/10.1130/abs/2016NC-275387 (2016).
Salamon, M. A. et al. Paleoenvironmental and biostratigraphic implications of echinoderm ossicles trapped within Burmese amber. PALAIOS 34, 652–656 (2019).
Kołbuk, D. et al. Effects of seawater Mg 2+ /Ca 2+ ratio and diet on the biomineralization and growth of sea urchins and the relevance of fossil echinoderms to paleoenvironmental reconstructions. Geobiology 18, 710–724 (2020).
Hermans, J., André, L., Navez, J., Pernet, P. & Dubois, P. Relative influences of solution composition and presence of intracrystalline proteins on magnesium incorporation in calcium carbonate minerals: Insight into vital effects. J. Geophys. Res. 116, G01001 (2011).
Weber, J. N. The incorporation of magnesium into the skeletal calcites of echinoderms. Am. J. Sci. 267, 537–566 (1969).
Gorzelak, P., Stolarski, J., Mazur, M. & Meibom, A. Micro- to nanostructure and geochemistry of extant crinoidal echinoderm skeletons. Geobiology 11, 29–43 (2013).
O’Malley, C. E., Ausich, W. I. & Chin, Y.-P. Isolation and characterization of the earliest taxon-specific organic molecules (Mississippian, Crinoidea). Geology 41, 347–350 (2013).
O’Malley, C. E., Ausich, W. I. & Chin, Y.-P. Deep echinoderm phylogeny preserved in organic molecules from Paleozoic fossils. Geology 44, 379–382 (2016).
Tucker, M. E. & Wright, V. P. Carbonate Sedimentology (Blackwell Publishing Ltd., 1990).
Dickson, J. A. D. Diagenesis and crystal caskets: echinoderm Mg calcite transformation, Dry Canyon, New Mexico, U.S.A. J. Sediment. Res. 71, 764–777 (2001).
Frank, T. D. & Lohmann, K. C. Diagenesis of fibrous magnesian calcite marine cement: implications for the interpretation of δ18O and δ13C values of ancient equivalents. Geochim. Cosmochim. Acta 60, 2427–2436 (1996).
Levin, L., Hönisch, B. & Frieder, C. Geochemical proxies for estimating faunal exposure to ocean acidification. Oceanography 25, 62–73 (2015).
Lebrato, M. et al. From the Arctic to the Antarctic: the major, minor, and trace elemental composition of echinoderm skeletons. Ecology 94, 1434–1434 (2013).
Iglikowska, A., Humphreys-Williams, E., Przytarska, J., Chełchowski, M. & Kukliński, P. Minor and trace elements in skeletons of Arctic echinoderms. Mar. Pollut. Bull. 158, 111377 (2020).
Yoshimura, T. et al. Altervalent substitution of sodium for calcium in biogenic calcite and aragonite. Geochim. Cosmochim. Acta 202, 21–38 (2017).
Laya, J. C. et al. Dissolution of ooids in seawater‐derived fluids – an example from Lower Permian re‐sedimented carbonates, West Texas, USA. Sedimentology 68, 2671–2706 (2021).
Gierlowski-Kordesch, E. H. Lacustrine carbonates. Dev. Sedimentol. 61, 1–101 (2010).
Tompa, É., Nyirő-Kósa, I., Rostási, Á., Cserny, T. & Pósfai, M. Distribution and composition of Mg-calcite and dolomite in the water and sediments of Lake Balaton. Cent. Eur. Geol. 57, 113–136 (2014).
Chagas, A. A. P., Webb, G. E., Burne, R. V. & Southam, G. Modern lacustrine microbialites: towards a synthesis of aqueous and carbonate geochemistry and mineralogy. Earth Sci. Rev. 162, 338–363 (2016).
Jones, B., Renaut, R. W. & Rosen, M. R. Trigonal dendritic calcite crystals forming from hot spring waters at Waikite, North Island, New Zealand. J. Sediment. Res. 70, 586–603 (2000).
Ishikawa, M. & Ichikuni, M. Uptake of sodium and potassium by calcite. Chem. Geol. 42, 137–146 (1984).
Devriendt, L. S. et al. Sodium incorporation into inorganic CaCO3 and implications for biogenic carbonates. Geochim. Cosmochim. Acta 314, 294–312 (2021).
Land, L. S. & Hoops, G. K. Sodium in carbonate sediments and rocks: a possible index to the salinity of diagenetic solutions. J. Sediment. Res. 43 (1973).
Hooker, J. D. & Binney, E. W. On the structure of certain limestone nodules enclosed in seams of bituminous coal, with a description of some Trigonocarpons contained in them. Philos. Trans. R. Soc. Lond. 145, 149–156 (1855).
Weber, J. N. & Keith, M. L. Carbon-isotope composition and the origin of calcareous coal balls. Science 138, 900–902 (1962).
Mozley, P. S. Relation between depositional environment and the elemental composition of early diagenetic siderite. Geology 17, 704 (1989).
Lyons, P. C., Millay, M. A., Zodrow, E. L., Cross, A. T. & Gillis, K. S. Discovery of permineralized plant fossils (coal balls) in the Bolsovian (e.g., Westphalian C) (Middle Pennsylvanian, Upper Carboniferous), Stellarton Basin, Nova Scotia, Canada. Can. J. Bot. 73, 1407–1416 (1995).
Lyons, P. C. et al. Coal-ball floras of Maritime Canada and palynology of the Foord seam: geologic, paleobotanical and paleoecological implications. Rev. Palaeobot. Palynol. 95, 31–50 (1997).
Zodrow, E. L. & Cleal, C. J. Anatomically preserved plants in siderite concretions in the shale split of the Foord Seam: mineralogy, geochemistry, genesis (Upper Carboniferous, Canada). Int. J. Coal Geol. 41, 371–393 (1999).
Whittaker, R. H. Communities and Ecosystems (Macmillan, 1975).
Bruenig, E. F. & Droste, H. J. Structure, Dynamics, and Management of Rainforests on Nutrient-Deficient Soils in Sarawakin in Ecology, Conservation, and Management of Southeast Asian Rainforests (eds. Primack, R. B. & Lovejoy, T. E.) Ch. 3, 41–53 (Yale University Press, 1995).
Cooper, H. V. et al. From peat swamp forest to oil palm plantations: the stability of tropical peatland carbon. Geoderma 342, 109–117 (2019).
Ribeiro, R. de A., Rovai, A. S., Twilley, R. R. & Castañeda‐Moya, E. Spatial variability of mangrove primary productivity in the neotropics. Ecosphere 10, e02841 (2019).
Ravn, R. L. et al. Stratigraphy of the Cherokee Group and revision of Pennsylvanian stratigraphic nomenclature in Iowa Iowa Geol. Surv. Tech. Inform. Ser. 12, 1–76 (1984)
Raymond, A. The paleoecology of a coal-ball deposit from the middle Pennsylvanian of Iowa dominated by cordaitalean gymnosperms. Rev. Palaeobot. Palynol. 53, 233–250 (1988).
DiMichele, W. A. & Phillips, T. L. Paleobotanical and paleoecological constraints on models of peat formation in the Late Carboniferous of Euramerica. Palaeogeogr. Palaeoclimatol. Palaeoecol. 106, 39–90 (1994).
Raymond, A., Lambert, L., Costanza, S., Slone, E. J. & Cutlip, P. C. Cordaiteans in paleotropical wetlands: an ecological re-evaluation. Int. J. Coal Geol. 83, 248–265 (2010).
Lane, S. J. & Dalton, J. A. Electron microprobe analysis of geological carbonates. Am. Mineral. 79, 745–749 (1994).
Macqueen, R. W. & Ghent, E. D. Electron microprobe study of magnesium distribution in some Mississippian echinoderm limestones from western Canada. Can. J. Earth Sci. 7, 1308–1316 (1970).
Reed, S. J. B. Electron Microprobe Analysis and Scanning Electron Microscopy in Geology (Cambridge Univ. Press, 2005).
Richter, D. K. & Fuchtbauer, H. Ferroan calcite replacement indicates former magnesian calcite skeletons. Sedimentology 25, 843–860 (1978).
Hasiuk, F. J. & Lohmann, K. C. Application of calcite Mg partitioning functions to the reconstruction of paleocean Mg/Ca. Geochim. Cosmochim. Acta 74, 6751–6763 (2010).
Alderton, D. Carbonates (Ca, Mg, Fe, Mn) in Encyclopedia of Geology (eds. Alderton, D. & Elias, S. A.) 382–394 (Elsevier Science, 2020).
Campbell, R. C. Statistics for Biologists (Cambridge Univ. Press, 1989).
Acknowledgements
Discussions with R. Guillemette, B. Wilkinson and W. A. DiMichele contributed to this paper. R. Wells, J. Teoh and A. Mott assisted with SEM images and electron microscopy (RRID:SCR_022202). C. Scotese consulted on the paleogeographic reconstruction in Fig. 1. Student research grants to M. Chrpa from GSA and GSA Energy Division (Antoinette Lierman Medlin Scholarship) supported this research. No permissions were required for the Dalton or Calhoun samples. We would like to thank the reviewers and Editors for their thoughtful comments and efforts towards improving our manuscript.
Author information
Authors and Affiliations
Contributions
M.E.C. and A.R. conceived the idea, collected samples, processed data, performed statistical analysis, wrote the paper, and created figures/tables. M.E.C., A.R., and W.M.L. prepared samples. All authors performed geochemical analyses. J.-C.L. and W.M.L. assisted with data interpretation and discussion. W.M.L. assisted with Methods.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Earth & Environment thanks Gregory Retallack, Przemyslaw Gorzelak, Blaine Cecil, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Olivier Sulpis and Joe Aslin.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chrpa, M.E., Raymond, A., Lamb, W.M. et al. A marine origin of coal balls in the Midland and Illinois basins, USA. Commun Earth Environ 4, 214 (2023). https://doi.org/10.1038/s43247-023-00876-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43247-023-00876-5
- Springer Nature Limited
This article is cited by
-
Calcium-rich seawater affects the mechanical properties of echinoderm skeleton
Communications Earth & Environment (2024)