Abstract
Two-dimensional polymers and three-dimensional covalent organic frameworks are crystalline, permanently porous macromolecules. Removing guest molecules from the pores of these materials reliably is challenging because extreme capillary forces are generated when evacuating nanoporous structures. Herein, we outline the most common approaches for isolating and activating these materials. We also note how some methods, such as thermal activation under vacuum, introduce significant challenges with reproducibly activating porous organic materials. Throughout this discussion, we highlight emerging approaches that are most reliable for activating a wide range of materials without damaging their porosity or crystallinity. We also discuss what material features lead to more robust scaffolds towards activation. We hope this review can serve as a resource for experts and newcomers in this field as they determine what activation protocol to use on as-synthesized materials.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Two-dimensional polymers (2DPs) and three-dimensional covalent organic frameworks (3D COFs) are organic materials with directional covalent bonding, highly ordered periodic structures and large permanent porosities1,2,3,4. These materials’ predictable and well-defined structural characteristics set them apart from other porous organic materials, such as polymers of intrinsic microporosity5,6, hypercrosslinked polymers7, and conjugated microporous polymers8. This structural definition has led to 2DPs and 3D COFs being explored for a wide range of applications, including gas storage, catalysis, chemical sensing, ionic conduction, drug delivery, energy storage, optoelectronics, and separation membranes9,10,11,12,13,14,15,16,17,18,19. In each of these applications, reliably retaining the ordered and nanoporous structure is perceived to be critical.
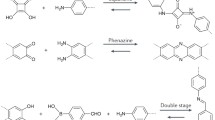
One can target crystalline lattices by judiciously selecting appropriate topological monomer systems (Fig. 1). Recent advances in polymerizing and processing 2DPs and 3D COFs have led to reliable protocols for obtaining many materials with a large number of linkage chemistries1,20,21,22. Frequently, these strategies will produce densely cross-linked and insoluble materials with the correct bond-forming chemistry as evaluated by vibrational spectroscopy. However, despite the use of directionally multi-topic linkers that polymerize in the desired way, practitioners in this field will attest that these materials are not always reproducibly crystalline and highly porous.
The observation that dimensional polymerization does not always yield well-defined materials has been a focal point in the study of crystalline organic systems. Many studies have speculated about the nature of the amorphous structures obtained in these cases. Some researchers have postulated that disordered network polymers are created when the formation dynamics are not well-controlled1,23,24,25. Others have speculated that randomly oriented linear polymers form, leaving dangling chemical functionality that fails to react26. Others contend that the desired crystalline lattice is present, but nanoscale deformations related to interpenetration or interlayer stacking make the materials appear disordered by conventional characterization methods27. These different scenarios are almost certainly system-dependent and are still challenging to resolve using even advanced characterization tools28,29. The situation is further complicated by the fact that researchers have now identified other routes to produce crystalline 3D COFs and 2DPs. For example, mechanochemical and reconstruction-based strategies are now both known to yield high-quality materials, but may not operate by the same mechanisms as those identified in solution-based condensation syntheses30,31,32,33,34. Moreover, newly developed synthetic approaches can yield highly crystalline systems in various morphologies, including single-crystals19,35,36,37, polycrystalline powders38,39, thin films22,40,41, or monoliths42,43. While significant improvements in material quality have been realized over the past two decades, the process of optimizing the synthesis of new material to avoid these amorphous forms is still largely empirical. We contend that one of the major reasons for this is that the post-synthetic processing of these materials is still subject to significant reproducibility challenges.
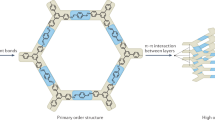
Following the polymerization of these materials and preceding their characterization, the materials must be isolated from their reaction mixture, which may contain monomeric species, oligomers, solvents, and residual catalysts. The insoluble nature of extended organic solvents makes it straightforward to collect them by filtration and wash them with fresh solvent, which typically removes these contaminating species. However, the nanoporous structures of 2DPs and 3D COFs make them especially prone to retaining guest molecules in their pores. For this reason, following their isolation and before their characterization, these materials are subjected to additional processing known as activation that evacuates the nanoporous structure. A common misconception is that the strong covalent bonds that drive the assembly of these species make them robust to activation. However, the rapid removal of solvent from nanoporous materials leads to extreme capillary forces (Fig. 2), which have been shown to degrade the porosity of other crystalline materials, such as metal-organic frameworks (MOFs)44,45. A variety of studies have shown how different isolation and workup protocols influence (and sometimes entirely disrupt) the crystallinity of these materials46,47,48. Perhaps unsurprisingly, workups' influence on material quality also depends on the exact material being investigated. Below, we describe how the activation protocols and specific structural features concurrently influence the reliability of obtaining high-quality materials. While 2DPs and 3D COFs are typically not studied together in the same report, we find that the structural and chemical similarities of these materials lead us to conclude that the workup and activation findings may generalize between them. By considering these two material classes together, we aim to answer the question of what methods are most reliable for the activation of structurally diverse 2DPs and 3D COFs.
Thermal activation
The most widely used activation method for conventional porous materials, such as porous carbons or zeolites49,50,51, is known as “thermal activation”45. In this process, materials are heated to remove volatile solvents, sometimes under negative applied pressure. Thermal or vacuum activation protocols are widely used because the equipment needed to perform this activation is readily available in unspecialized chemical and material laboratories. However, this process is inherently challenging to systematically reproduce because controlling the rate of solvent evacuation is difficult to standardize with common laboratory equipment.
Porous framework materials and dimensional polymers are generally synthesized using high boiling solvents, which facilitate the high reaction temperatures that are sometimes invoked as necessary to produce high-quality materials9,47,52,53. This presents a challenge for thermal activation because high temperatures and reduced pressure are then required to remove residual solvent molecules in the pores. These high temperatures may be incompatible with the thermal or chemical stability of 2DPs or 3D COFs. High boiling solvents also generally have higher surface tensions than lower boiling point solvents. This produces another degradation mechanism where capillary forces collapse the initially crystalline lattice (Fig. 2). Because capillary pressures are positively correlated with the surface tension of the solvent, thermal activation from higher boiling solvents is more likely to lead to pore collapse and amorphization. Capillary pressure is also inversely related to the pore size of the pore, which means that nanoporous materials experience high contraction forces when even moderate surface tension solvents are removed. These capillary forces are the main cause of pore collapse during activation and are important to minimize for reproducible activation.
It has been widely observed for MOFs, COFs, and 2DPs that attempting to isolate high boiling points directly by thermal activation of the reaction solvent is unsuccessful. Farha, Matzger, and coworkers reported a strategy to address this in MOF crystallites by exchanging high boiling solvents for low boiling solvents prior to vacuum drying at elevated temperatures46,54. Using this strategy for different classes of MOFs, they found that the crystallinity, high surface area, and narrow pore size distribution could be retained when using this solvent exchange strategy. A similar strategy was employed by Yaghi and coworkers in their seminal report of a 2DP, which they call COF-5, where the solvent exchange the dioxane:mesitylene mixture used for synthesis with acetone prior to activation (Fig. 3a)55. This approach yielded crystalline materials with narrow pore size distributions, as determined by powder X-ray diffraction and nitrogen porosimetry (Fig. 3b). Exchanging solvent for a slightly lower boiling point solvent during washing prior to vacuum activation is widely used in the porous organic materials community.
a Synthesis and activation of a boronate ester-linked 2DP and its b post-activation nitrogen isotherm. Blue- and orange-colored circles represent adsorption and desorption. The graph was reproduced with permission from ref. 55 (copyright American Association for the Advancement of Science, 2005).
Thermal activation is not reliable for a large number of 2DP and 3D COF structures. For instance, while vacuum activation is known to be successful for COF-5 activation, it has been experimentally deleterious for a common imine-linked 2DP (TAPB–PDA, TAPB = 1,3,5-Tris(4-aminophenyl)benzene and PDA = phenylene dialdehyde, Fig. 4a). Although the reason behind the fragility of 2DPs to workup is still not entirely understood, it is often assumed that interlayer non-covalent reinforcement acts to stabilize these materials during activation. In the context of the previous observations regarding COF-5 and TAPB–PDA (Fig. 4a), it may be concluded that the more planar, rigid, conjugated structure of COF-5 improves its stability to thermal activation. Other reports reveal that enhanced π–π interactions (Fig. 4b)56, strong H-bonding between layers (Fig. 4c)57, incorporation of arene-perfluoroarene interactions (Fig. 4d)58, molecular docking (Fig. 4e)59,60, synergistic dipolar attractions (Fig. 4f)61 can all improve 2DP stability as evaluated by X-ray diffraction (XRD) or porosimetry measurements62,63. In some cases, these interactions can be introduced by mixing the monomers used to produce these crystalline networks. For example, 2DPs that incorporate self-complementary arene–perfluoarene interactions by mixing arene and perfluoroarene linkers were found to be more robust than those comprised of only arene or perfluoroarene linkers, respectively. The authors note that this is due to the fact that these interactions enhance the crystal packing energy of 2DPs and reduce their effective unit cell size64. This was confirmed by monitoring the intensity of the XRD profile and the nitrogen porosimetry isotherms, both of which showed higher material quality when 2,3,5,6-tetrafluoroterephthalaldehyde:terephthalaldehyde was combined in a 1:1 ratio with a 5,10,15,20-tetrakis(p-tetraphenylamine)porphyrin node56. Similarly, Smaldone and coworkers achieved improved crystallinities and larger surface areas (2000 m2 g−1) in 2DPs with arene-perfluoroarene interactions than those that did not have these interactions (760 m2 g−1)65. This agrees with in situ X-ray scattering observations by Dichtel and coworkers that identified 2DPs with stronger supramolecular interactions had higher thermal stabilities62,63. In these reports, the authors also identified amorphous phases that emerge prior to covalent bond breaking, which suggests that meta-stable structural phases might exist in crystalline organic materials more generally. Similar trends have been identified in 3D COFs when molecular engineering is used to fill nanopores with bulky substituents that increase the robustness of these materials66. Presumably, this occurs because fewer solvent molecules and, thus, weaker capillary forces are present to disrupt the crystalline structure upon desolvation. This is consistent with similar observations that more densely interpenetrated networks are generally more stable than their less interpenetrated counterparts67,68,69,70,71,72.
a TAPB–PDA material that is known to be sensitive to activation. Supramolecular reinforcement has been shown to lead to more stable and more reproducibly activatable crystalline networks, including. b π–π stacking interactions. c H-bonding (black colored dashed lines represent H-bonding). d Arene–perfluoroarene interactions. e Docking interactions, inset illustrates COF lattice with the red circle representing the rotation of tetraphenyl-ethene moiety (C, orange; N, blue; H, white). f Dipole–dipole interactions. This graphic was reproduced with permission from ref. 59 (copyright Nature Portfolio, 2016).
The unique stability to thermal activation of a select number of easily synthesized materials raises an intriguing possibility that early mechanistic work aimed at optimizing the synthesis of 2DPs and 3D COFs may not have been selected for optimal synthesis conditions. Instead, these early reports may have been unintentionally optimizing for materials that were most robust to thermal activation protocols of the time. Nonetheless, we infer that thermal activation is likely among the most challenging approaches to perform reliably on crystalline organic materials and may not be amenable to materials without meaningful supramolecular reinforcement.
Fluorinated solvent exchange
One approach to combat the unreliability of vacuum activation is to exchange the reaction solvent for extremely low surface tension solvents that generate reduced capillary forces upon their removal (Table 1). Verduzco pioneered an approach to activate crystalline organic materials by exchanging them with low surface tension fluorinated solvents73. In their initial efforts, they considered six different 2DPs and attempted to activate them using variable surface tension solvents (11.9–72.8 mN m−1). They found that even an extremely fragile TAPB–PDA could be activated by exchanging the pore solvent for perfluorinated hexane (surface tension 11.9 mN m−1) to yield highly crystalline and high surface area materials (>2100 m2 g−1). In this same study, these authors also showed how increasing the surface tension of the activating solvents led to significant decreases for TAPB–PDA, with the lowest surface area of 172 m2 g−1 in polar DMF (37.1 mN m−1). For more robust systems, such as TAPB–OMe–PDA, TAPT–OMe–PDA, and TAPB–BTCA, the crystalline and porous nature were negligibly impacted by using high-surface tension solvents. This observation undeniably demonstrates that while activation methods have differences in their reliability, some systems may be insensitive to activation variability. The authors go on to conclude that activating nanoporous materials with fluorinated solvent exchange could be a universal method to activate 2DPs and 3D COFs without the need for specialized equipment. We expect systematic studies into activation, such as those performed by Verduzco and coworkers will provide important guidance in the field of reticular organic materials for years to come.
Identifying the structure-property relationships pertinent to crystalline organic materials activation is critically important. Verduzco and coworkers demonstrated that TAPB–PDA’s surface area upon workup is linearly related to the surface tension of the solvent infiltrated into the pores prior to activation (Fig. 5)74. This experiment was performed by exposing the porous 2DP to mixtures of n-hexane (18.4 mN m−1) and ethanol (22.1 mN m−1) prior to vacuum activation (1 Torr). When pure n-hexane was used, a high surface area of >2100 m2 g−1 was observed by nitrogen porosimetry. In contrast, when a 70:30 (vol) hexane:EtOH mixture was used, the surface area was significantly decreased (<200 m2 g−1). These porosimetry results are consistent with powder X-ray diffraction patterns of materials activated under these conditions, which showed that less defined Bragg features are obtained when higher surface tension solvents are used for activation. The authors also measured how other potentially convoluting characteristics, such as isolation under reduced pressure or elevated temperature, impacted the measured surface area. They found that these factors did not predict the measured surface area, leading the authors to conclude that the surface tension of the solvent was the dominant predictor of successful activation. Similar trends were observed with other 2DP systems in the same study. Together, these systematic studies by Verduzco and coworkers reveal that low surface tension solvents, such as perfluoroalkanes, can be used to reliably isolate crystalline and high surface area nanoporous materials without the need for specialized equipment73. We encourage newcomers to this area without access to more complex experimentation to consider this approach as a method to improve the reliability of thermal activation.
a Variable levels of structural damage during activation with variable solvent mixtures. b Powder X-ray diffraction. c Nitrogen porosimetry measurements (orange circles) show how more polar solvents (surface tension shown as blue squares) lead to more pronounced damage. This graphic was reproduced with permission from ref. 74 (copyright American Chemical Society, ACS 2022).
Despite the success of using perfluoroalkanes for lab-scale activation, these materials are likely not suited for large-scale or industrial activation of 2DPs or 3D COFs. The environmental drawbacks of perfluorinated substances are well-documented. The hazards associated with the materials used by Verduzco and coworkers have led to them being labeled as “emerging pollutants”75,76. For this reason, researchers have explored how to activate 2DPs and 3D COFs with other low-vapor pressure solvents. For example, ionic liquids were used as an alternative to the typical synthesis solvents by Qiu and coworkers for 3D COFs and Jiang and coworkers for 2DPs77,78,79. Because of ionic liquids' low vapor pressure, miscibility with both organic and inorganic solvents, and non-flammability, they have several advantages over more conventional solvents. Moreover, because they are readily miscible in many solvents, they can be easily removed by low-surface tension solvents (e.g. MeOH). This strategy represents a unique approach to synthesizing high-quality porous organic materials and activating them with low-surface tension solvents.
Identifying activation stability structure–property relationships is important to the continued development of nanoporous organic materials. In their efforts to identify a reliable activation method, Verduzco and coworkers also measured the stability of several imine-linked 2DPs to different activation protocols80. For example, they measured how pore-functionalizing TAPB–PDA impacted the stability of this parent lattice. They found that functionalizing these materials with small motifs (such as F-atoms) did little to improve the stability of activation. However, functionalizing with larger motifs (such as Br-atoms) provided a significant enhancement to their activation, as evaluated by nitrogen porosimetry and X-ray diffraction measurements (Fig. 6). Smet and coworkers identified a similar trend by comparing methylated and unmethylated forms of the same imine-linked 2DP81. Methylation enhanced the stability of two 2DPs to thermal activation and yielded improved surface areas as evaluated by nitrogen porosimetry. The trends were even more pronounced upon vacuum activation with Brunauer–Emmett–Teller surface areas (SBET) of 2100, 2100, 400, and 1000 m2 g-1 for Me3TFB–PA, Me3TFB–BD, TFB–PA, and TFB–BD, respectively (Fig. 7). Similarly, integrating methoxy units into 2DPs framework led to significant improvement in crystallinity, surface area and their chemical stability (e.g. acid–base treatment)82,83. This enhanced material quality is hypothesized to originate from the dipolar interactions between cofacial aromatic rings. Note that choosing such prochiral starting materials leads to forming local isomers, such that the dipolar interactions between layers are likely incomplete. Moreover, the propensity for imine condensation/hydrolysis is also impacted by substituting the benzene ring with OMe. So, it is challenging to disentangle the importance of workup from the impact of in-plane polymerization and cross-plane stacking19. While these measurements demonstrate that steric pore functionalities help to stabilize the framework, it cannot be ruled out that these functionalizations may also provide some additional supramolecular reinforcement that is challenging to characterize. Verduzco and coworkers partially addressed this ambiguity by assessing a pyrene-based rhombic 2DPs80. They found that larger pore sizes led to decreased activation stability in these systems. This suggests that whatever features lead to stability with one family of 2DP may be possible to generalize to a broader class of materials. Going forward, isolating the influence of individual structural features will be a valuable addition to the field of nanoporous materials activation.
Supercritical CO2 activation
As has been reported across many materials, decreasing the surface tension of solvent in the pore is a straightforward method to prevent pore collapse. This realization has encouraged exchanging residual solvents for supercritical fluids, namely CO2 (scCO2), which have no surface tension84,85,86. The lack of surface tension ensures that the nanoporous material experiences no capillary forces upon activation. For this reason, scCO2 activation is the most reliable method to isolate near-pristine nanoporous organic materials. As one example, Marder, Dichtel, and coworkers observed that isolating a variety of 2DPs from virtually any solvent miscible with scCO2 led to near-theoretical isotherms87. For example, TAPB–PDA (previously reported as fragile) activated by scCO2 had a surface area of >2500 m2 g−1, whereas all vacuum-activated samples were less than 300 m2 g−1 with poorly defined isotherms at low partial pressure. Similarly, Verduzco and coworkers showed how nanoporous 3D COFs with pto and mhq-z topologies could be isolated by solvent exchanging with THF, acetone, and ethanol, followed by supercritical CO2 activation to afford yellow crystalline powders88. Interestingly, Wei and coworkers have recently performed the synthesis and activation of 3D COFs in supercritical CO2, which allowed them to isolate single-crystalline 3D COFs with sizes of up to 110 µm directly from the polymerization mixture89. The use of scCO2 activation has driven the expansion of isolable 2DP and 3D COF systems and, in part, addresses the challenges with reproducibility in nanoporous materials activation. However, scCO2 activation requires specialized equipment and is time-consuming. This realization motivates a continued exploration of straightforward and efficient nanoporous material activation protocols.
Nitrogen drying
The rate of solvent removal is related to the capillary forces experienced by a nanoporous medium. It stands to reason that removing solvent slowly could preserve the nanoporous structure, even in the presence of high surface tension solvents. However, this is challenging to control by applying negative pressures with conventional vacuum setups in material synthesis laboratories. Another approach is to remove solvent by slow evaporation. Marder, Dichtel, and coworkers explored this strategy by solvent-exchanging several hydrated materials in MeOH and then putting the wetted powders in a vial under constant dry nitrogen flow (Fig. 8)87. After several hours under gentle nitrogen flow, surface areas similar to those obtained by scCO2 activation were obtained for a variety of 2DP structures (Fig. 9). While these authors found no dependence on the solvents used in this protocol, it is conceivable that in some cases higher surface tension solvents may be desirable because they will evaporate more slowly. We find MeOH exchange followed by extended drying by N2 flow to be a reliable method to activate nanoporous materials that might otherwise be unstable to vacuum activation.
The structures of 2DPs were reported to be successfully activated by the dry nitrogen flow method. This graphic was reproduced with permission from ref. 87 (copyright Wiley VCH, 2020).
Amorphization under activation is generally perceived to be irreversible. However, recent efforts have shown how this assumption may not be accurate in all cases. Bein and coworkers explored the stability of several 2DPs towards activation86. Similar to other reports, they found that unsubstituted TAPB-based systems were exceptionally sensitive to thermal activation, whereas more functionalized or supramolecularly reinforced systems could be isolated without optimizing the activation and isolation protocols. In all cases, the authors could isolate highly crystalline and porous materials using scCO2 activation. In an unexpected finding, the authors also found that exposing 2DPs made amorphous by vacuum activation to subsequent scCO2 activation protocols led to a near full recovery surface area and crystallinity recovery (Fig. 10). This interconversion between amorphous and crystalline states could be performed several times reliably for several materials. This observation raises important questions about the structure accessed in the amorphous state upon vacuum activation. It seems to indicate that the bonding environment is preserved upon activation, but a metastable collapsed structure is generated. The ability to toggle between these two states reliably may provide a unique opportunity to remove guest molecules that might otherwise be strongly adsorbed to the nanoporous material. More investigations into reversibly toggling porosity will be required to fully evaluate the potential of these transformations. Ultimately, accessing reproducible protocols for the isolation and activation of nanoporous materials will be a prerequisite to these investigations.
Outlook
Crystalline organic lattices with high crystallinity and porosity have been shown as promising for many applications and as a desirable platform to interrogate fundamental scientific questions. However, to realize the promise of these materials, their production must be reliable. While significant improvements have been made in optimizing the design and synthesis of these materials, the reproducibility challenges associated with their activation are often overlooked. Here, we discuss the different activation protocols and their associated advantages and disadvantages. We identify supercritical CO2 activation as the most reproducible method to obtain crystalline/porous materials but note that this method requires specialized equipment that may not be accessible to all researchers. Newly developed methods, including slow drying with a N2 stream or solvent exchanging with perfluorinated alkanes are more straightforward to perform with standard materials chemistry equipment and give similar levels of reproducibility to supercritical CO2 activation. For this reason, we encourage newcomers to this area to consider using these emerging activation approaches, which are considerably more reproducible than thermal activation protocols.
It is now clear that some 2DPs and 3D COFs are more stable to harsh activation protocols than others. Research from several groups has shown how smaller pore sizes, the inclusion of bulky substituents, or the reinforcement of layers by supramolecular interactions all lead to more stable crystalline lattices. However, an exact understanding of how these features stabilize these materials is not yet well-developed. The system-dependent reproducibility to workup also introduces challenges when trying to compare the viability of general synthetic methods, which might be confounded by the particular model systems used to develop these approaches. Going forward, more systematic investigations of activation reproducibility should be performed for materials with various topologies, pore sizes, functionalizations, defect densities, grain sizes, and morphologies. We expect that these investigations will be complemented by emerging characterization tools that allow researchers to better understand what structural evolutions occur during activation. As these investigations are performed, it will be of substantial interest to identify whether the trends for 2DPs can be generalized to 3D COFs and vice versa. A more robust understanding of activation reproducibility may also provide a route to switchable porosities and meta-stable framework architectures, which we find to be an exciting possibility.
We hope that the suggestions made in this review will be valuable to new researchers in this area and expedite their ability to reliably obtain new crystalline organic materials. Widespread adoption of best activation practices will ultimately unlock the potential for meaningful comparisons between diverse materials and data, both experimental and computational, across different laboratories. This paves the way for significant research advancements by ensuring data consistency and replicability. Importantly, crystalline organic polymers are an exciting and rapidly developing frontier, and we expect that best practices may evolve as new developments become available.
References
Evans, A. M. et al. Two-dimensional polymers and polymerizations. Chem. Rev. 122, 442–564 (2022).
Sakamoto, J., van Heijst, J., Lukin, O. & Schlüter, A. D. Two-dimensional polymers: just a dream of synthetic chemists? Angew. Chem. Int. Ed. 48, 1030–1069 (2009).
Colson, J. W. & Dichtel, W. R. Rationally synthesized two-dimensional polymers. Nat. Chem. 5, 453–465 (2013).
Springer, M. A., Liu, T.-J., Kuc, A. & Heine, T. Topological two-dimensional polymers. Chem. Soc. Rev. 49, 2007–2019 (2020).
Cheng, G. et al. Conjugated polymers of intrinsic microporosity (C-PIMs). Adv. Funct. Mater. 24, 5219–5224 (2014).
McKeown, N. B. & Budd, P. M. Polymers of intrinsic microporosity (PIMs): organic materials for membrane separations, heterogeneous catalysis and hydrogen storage. Chem. Soc. Rev. 35, 675–683 (2006).
Tan, L. & Tan, B. Hypercrosslinked porous polymer materials: design, synthesis, and applications. Chem. Soc. Rev. 46, 3322–3356 (2017).
Lee, J.-S. M. & Cooper, A. I. Advances in conjugated microporous polymers. Chem. Rev. 120, 2171–2214 (2020).
Lohse, M. S. & Bein, T. Covalent organic frameworks: structures, synthesis, and applications. Adv. Funct. Mater. 28, 1705553 (2018).
Guan, X., Chen, F., Fang, Q. & Qiu, S. Design and applications of three dimensional covalent organic frameworks. Chem. Soc. Rev. 49, 1357–1384 (2020).
Wang, Z., Zhang, S., Chen, Y., Zhang, Z. & Ma, S. Covalent organic frameworks for separation applications. Chem. Soc. Rev. 49, 708–735 (2020).
Liu, R. et al. Covalent organic frameworks: an ideal platform for designing ordered materials and advanced applications. Chem. Soc. Rev. 50, 120–242 (2021).
Meng, Z. & Mirica, K. A. Covalent organic frameworks as multifunctional materials for chemical detection. Chem. Soc. Rev. 50, 13498–13558 (2021).
Jhulki, S. et al. Humidity sensing through reversible isomerization of a covalent organic framework. J. Am. Chem. Soc. 142, 783–791 (2020).
Rashid, R. B. et al. A semiconducting two-dimensional polymer as an organic electrochemical transistor active layer. Adv. Mater. 34, 2110703 (2022).
Fang, Q. et al. 3D Porous crystalline polyimide covalent organic frameworks for drug delivery. J. Am. Chem. Soc. 137, 8352–8355 (2015).
Xu, H., Tao, S. & Jiang, D. Proton conduction in crystalline and porous covalent organic frameworks. Nat. Mater. 15, 722–726 (2016).
He, G., Zhang, R. & Jiang, Z. Engineering covalent organic framework membranes. Acc. Mater. Res. 2, 630–643 (2021).
Natraj, A. et al. Single-crystalline imine-linked two-dimensional covalent organic frameworks separate benzene and cyclohexane efficiently. J. Am. Chem. Soc. 144, 19813–19824 (2022).
Waller, P. J. et al. Chemical conversion of linkages in covalent organic frameworks. J. Am. Chem. Soc. 138, 15519–15522 (2016).
Xue, S. et al. Advanced development of three-dimensional covalent organic frameworks: valency design, functionalization, and applications. Coord. Chem. Rev. 504, 215659 (2024).
Yang, Y., Ratsch, M., Evans, A. M. & Börjesson, K. Layered 3D covalent organic framework films based on carbon–carbon bonds. J. Am. Chem. Soc. 145, 18668–18675 (2023).
Smith, B. J. & Dichtel, W. R. Mechanistic studies of two-dimensional covalent organic frameworks rapidly polymerized from initially homogenous conditions. J. Am. Chem. Soc. 136, 8783–8789 (2014).
Li, H. et al. Nucleation–elongation dynamics of two-dimensional covalent organic frameworks. J. Am. Chem. Soc. 142, 1367–1374 (2020).
Zhang, Z. et al. Micro/nano-scaled covalent organic frameworks: polymerization, crystallization and self-assembly. ChemNanoMat 8, e202100345 (2022).
Zhu, D. et al. Transformation of one-dimensional linear polymers into two-dimensional covalent organic frameworks through sequential reversible and irreversible chemistries. Chem. Mater. 33, 413–419 (2021).
Feriante, C. et al. New mechanistic insights into the formation of imine-linked two-dimensional covalent organic frameworks. J. Am. Chem. Soc. 142, 18637–18644 (2020).
Castano, I. et al. Mapping grains, boundaries, and defects in 2D covalent organic framework thin films. Chem. Mater. 33, 1341–1352 (2021).
Robison, L. et al. Transient catenation in a zirconium-based metal-organic framework and its effect on mechanical stability and sorption properties. J. Am. Chem. Soc. 143, 1503–1512 (2021).
Biswal, B. P. et al. Mechanochemical synthesis of chemically stable isoreticular covalent organic frameworks. J. Am. Chem. Soc. 135, 5328–5331 (2013).
Das, G., Balaji Shinde, D., Kandambeth, S., Biswal, B. P. & Banerjee, R. Mechanosynthesis of imine, β-ketoenamine, and hydrogen-bonded imine-linked covalent organic frameworks using liquid-assisted grinding. Chem. Commun. 50, 12615–12618 (2014).
Emmerling, S. T. et al. In situ monitoring of mechanochemical covalent organic framework formation reveals templating effect of liquid additive. Chem 7, 1639–1652 (2021).
Brown, N. et al. Green mechanochemical synthesis of imine-linked covalent organic frameworks for high iodine capture. Green Chem. 25, 6287–6296 (2023).
Zhang, W. et al. Reconstructed covalent organic frameworks. Nature 604, 72–79 (2022).
Zhang, Y.-B. et al. Single-crystal structure of a covalent organic framework. J. Am. Chem. Soc. 135, 16336–16339 (2013).
Kang, C. et al. Growing single crystals of two-dimensional covalent organic frameworks enabled by intermediate tracing study. Nat. Commun. 13, 1370 (2022).
Evans, A. M. et al. Seeded growth of single-crystal two-dimensional covalent organic frameworks. Science 361, 52–57 (2018).
Qian, C. et al. Polycrystalline covalent organic frameworks. Mater. Today 71, 91–107 (2023).
Matsumoto, M. et al. Rapid, low temperature formation of imine-linked covalent organic frameworks catalyzed by metal triflates. J. Am. Chem. Soc. 139, 4999–5002 (2017).
Kumar Mahato, A. et al. Crystallizing sub 10 nm covalent organic framework thin films via interfacial–residual concomitance. J. Am. Chem. Soc. 143, 20916–20926 (2021).
Yang, Y. et al. A self-standing three-dimensional covalent organic framework film. Nat. Commun. 14, 220 (2023).
Liu, Z., Wang, H., Ou, J., Chen, L. & Ye, M. Construction of hierarchically porous monoliths from covalent organic frameworks (COFs) and their application for bisphenol A removal. J. Hazard. Mater. 355, 145–153 (2018).
Huang, L. et al. Monolithic covalent organic frameworks with hierarchical architecture: attractive platform for contaminant remediation. Chem. Mater. 35, 2661–2682 (2023).
Mondloch, J. E., Karagiaridi, O., Farha, O. K. & Hupp, J. T. Activation of metal-organic framework materials. CrystEngComm 15, 9258–9264 (2013).
Zhang, X. et al. A historical overview of the activation and porosity of metal-organic frameworks. Chem. Soc. Rev. 49, 7406–7427 (2020).
Howarth, A. J. et al. Best practices for the synthesis, activation, and characterization of metal-organic frameworks. Chem. Mater. 29, 26–39 (2017).
Farha, O. K. & Hupp, J. T. Rational design, synthesis, purification, and activation of metal-organic framework materials. Acc. Chem. Res. 43, 1166–1175 (2010).
Nelson, A. P., Farha, O. K., Mulfort, K. L. & Hupp, J. T. Supercritical processing as a route to high internal surface areas and permanent microporosity in metal-organic framework materials. J. Am. Chem. Soc. 131, 458–460 (2009).
Wang, Q. et al. Functional zeolitic-imidazolate-framework-templated porous carbon materials for CO2 capture and enhanced capacitors. Chem. Asian J. 8, 1879–1885 (2013).
Ivanova, E. N., Averin, A. A., Alekhina, M. B., Sokolova, N. P. & Kon’kova, T. V. Thermal activation of type X zeolites in the presence of carbon dioxide. Prot. Met. Phys. Chem. Surf. 52, 267–272 (2016).
Lee, J., Kim, J. & Hyeon, T. Recent progress in the synthesis of porous carbon materials. Adv. Mater. 18, 2073–2094 (2006).
Geng, K. et al. Covalent organic frameworks: design, synthesis, and functions. Chem. Rev. 120, 8814–8933 (2020).
Li, X. et al. Expeditious synthesis of covalent organic frameworks: a review. J. Mater. Chem. A 8, 16045–16060 (2020).
Wright, K. R., Nath, K. & Matzger, A. J. Superior metal-organic framework activation with dimethyl ether. Angew. Chem. Int. Ed. 61, e202213190 (2022).
Côté, A. P. et al. Porous, crystalline, covalent organic frameworks. Science 310, 1166–1170 (2005).
Chen, X., Addicoat, M., Irle, S., Nagai, A. & Jiang, D. Control of crystallinity and porosity of covalent organic frameworks by managing interlayer interactions based on self-complementary π-electronic force. J. Am. Chem. Soc. 135, 546–549 (2013).
Chen, X. et al. Locking covalent organic frameworks with hydrogen bonds: general and remarkable effects on crystalline structure, physical properties, and photochemical activity. J. Am. Chem. Soc. 137, 3241–3247 (2015).
Braunecker, W. A. et al. Phenyl/perfluorophenyl stacking interactions enhance structural order in two-dimensional covalent organic frameworks. Cryst. Growth Des. 18, 4160–4166 (2018).
Ascherl, L. et al. Molecular docking sites designed for the generation of highly crystalline covalent organic frameworks. Nat. Chem. 8, 310–316 (2016).
Auras, F. et al. Synchronized offset stacking: a concept for growing large-domain and highly crystalline 2D covalent organic frameworks. J. Am. Chem. Soc. 138, 16703–16710 (2016).
Salonen, L. M. et al. A supramolecular strategy based on molecular dipole moments for high-quality covalent organic frameworks. Chem. Commun. 52, 7986–7989 (2016).
Evans, A. M. et al. Trends in the thermal stability of two-dimensional covalent organic frameworks. Faraday Discuss. 225, 226–240 (2021).
Evans, A. M. et al. Buckling of two-dimensional covalent organic frameworks under thermal stress. Ind. Eng. Chem. Res. 58, 9883–9887 (2019).
Li, W.-B., Cheng, Y.-Z., Yang, D.-H., Liu, Y.-W. & Han, B.-H. Fluorine-containing covalent organic frameworks: synthesis and application. Macromol. Rapid Commun. 44, 2200778 (2023).
Alahakoon, S. B., McCandless, G. T., Karunathilake, A. A. K., Thompson, C. M. & Smaldone, R. A. Enhanced structural organization in covalent organic frameworks through fluorination. Chem. Eur. J. 23, 4255–4259 (2017).
Wang, Y. et al. Three-dimensional mesoporous covalent organic frameworks through steric hindrance engineering. J. Am. Chem. Soc. 142, 3736–3741 (2020).
Hanna, S. L. et al. Discovery of spontaneous de-interpenetration through charged point-point repulsions. Chem 8, 225–242 (2022).
Kang, X. et al. Rational synthesis of interpenetrated 3D covalent organic frameworks for asymmetric photocatalysis. Chem. Sci. 11, 1494–1502 (2020).
Zhu, Q. et al. 3D cage COFs: a dynamic three-dimensional covalent organic framework with high-connectivity organic cage nodes. J. Am. Chem. Soc. 142, 16842–16848 (2020).
Martínez-Abadía, M. et al. π-Interpenetrated 3D covalent organic frameworks from distorted polycyclic aromatic hydrocarbons. Angew. Chem. Int. Ed. 60, 9941–9946 (2021).
Shan, Z. et al. 3D Covalent organic frameworks with interpenetrated PCB topology based on 8-connected cubic nodes. J. Am. Chem. Soc. 144, 5728–5733 (2022).
Fischbach, D. M., Rhoades, G., Espy, C., Goldberg, F. & Smith, B. J. Controlling the crystalline structure of imine-linked 3D covalent organic frameworks. Chem. Commun. 55, 3594–3597 (2019).
Zhu, D. & Verduzco, R. Ultralow surface tension solvents enable facile COF activation with reduced pore collapse. ACS Appl. Mater. Interfaces 12, 33121–33127 (2020).
Zhu, D. et al. Solvent-induced incremental pore collapse in two-dimensional covalent organic frameworks. ACS Mater. Lett. 4, 2368–2374 (2022). The authors show how higher surface tension solvents are more likely to lead to pore collapse.
Zhang, C. et al. Biological utility of fluorinated compounds: from materials design to molecular imaging, therapeutics and environmental remediation. Chem. Rev. 122, 167–208 (2022).
Schlaich, C., Yu, L., Cuellar Camacho, L., Wei, Q. & Haag, R. Fluorine-free superwetting systems: construction of environmentally friendly superhydrophilic, superhydrophobic, and slippery surfaces on various substrates. Polym. Chem. 7, 7446–7454 (2016).
Gao, Y. et al. Synthesis of two-dimensional covalent organic frameworks in ionic liquids. Chem. Eur. J. 25, 15488–15492 (2019).
Guan, X. et al. Fast, ambient temperature and pressure ionothermal synthesis of three-dimensional covalent organic frameworks. J. Am. Chem. Soc. 140, 4494–4498 (2018).
Zhao, L. et al. An ionic liquid as a green solvent for high potency synthesis of 2D covalent organic frameworks. New J. Chem. 44, 15410–15414 (2020).
Zhu, D. et al. Understanding fragility and engineering activation stability in two-dimensional covalent organic frameworks. Chem. Sci. 13, 9655–9667 (2022). This work highlights how activation fragility of 2DPs is influenced by their pore size, pore functionality, and pore architecture.
Dautzenberg, E., Lam, M., Li, G. & de Smet, L. C. P. M. Enhanced surface area and reduced pore collapse of methylated, imine-linked covalent organic frameworks. Nanoscale 13, 19446–19452 (2021). The authors demonstrate how methylation of 2DP core significantly improves the stability of these materials to activation.
Halder, A. et al. Ultrastable imine-based covalent organic frameworks for sulfuric acid recovery: an effect of interlayer hydrogen bonding. Angew. Chem. Int. Ed. 57, 5797–5802 (2018).
Zhai, L. et al. Constructing stable and porous covalent organic frameworks for efficient iodine vapor capture. Macromol. Rapid Commun. 42, 2100032 (2021).
Cooper, A. I. Porous materials and supercritical fluids. Adv. Mater. 15, 1049–1059 (2003).
Matsuyama, K. Supercritical fluid processing for metal-organic frameworks, porous coordination polymers, and covalent organic frameworks. J. Supercrit. Fluids 134, 197–203 (2018).
Sick, T. et al. Switching on and off interlayer correlations and porosity in 2D covalent organic frameworks. J. Am. Chem. Soc. 141, 12570–12581 (2019). The authors depict how fragile non-porous 2DPs can be reconverted into highly crystalline and porous frameworks using supercritical CO2 activation.
Feriante, C. H. et al. Rapid synthesis of high surface area imine-linked 2D covalent organic frameworks by avoiding pore collapse during isolation. Adv. Mater. 32, 1905776 (2020). The authors demonstrate dry N2 flow as an alternative and efficient method to activate 2DPs.
Zhu, D. et al. Three-dimensional covalent organic frameworks with pto and mhq-z topologies based on tri- and tetratopic linkers. Nat. Commun. 14, 2865 (2023).
Peng, L. et al. Ultra-fast synthesis of single-crystalline three-dimensional covalent organic frameworks and their applications in polarized optics. Chem. Mater. 34, 2886–2895 (2022).
Acknowledgements
We thank the Department of Defence for a Multidisciplinary University Research Initiative Award (W911NF2310260) for supporting our work.
Author information
Authors and Affiliations
Contributions
A.M.E. and R.R. prepared the manuscript. All authors have read and approved the final version of the manuscript for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Materials thanks Donglin Jiang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Natalia Shustova and Jet-Sing Lee.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Roy, R., Evans, A.M. Reproducibility challenges in activating two-dimensional polymers and three-dimensional covalent organic frameworks. Commun Mater 5, 102 (2024). https://doi.org/10.1038/s43246-024-00536-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43246-024-00536-x
- Springer Nature Limited