Abstract
Chiral benzo five-membered heterocyclic spirocyclopropanes are an important class of parent core structures with pharmacological activity. A novel organocatalytic one-pot cascade ether oxidation iminium-ion activation strategy for the asymmetric spirocyclopropylation of benzofuran-2-ones and indolin-2-ones from allyl tert-butyl ethers/ pent-2,4-dienyl ethyl ethers with excellent enantioselectivity (ee% up to > 99) and diastereoselectivity(dr.% up to 91:9) has been developed. This process involves the successful dynamic kinetic resolution of racemic 3-bromobenzofuran-2-ones or 3-bromoindolin-2-ones. Its synthetic application will provide a new aminocatalytic cascade tool for the efficient synthesis of complex molecules.
Similar content being viewed by others
Introduction
Organocatalytic enal-derived iminium-enamine activated asymmetric transformations have been demonstrated as powerful approaches for constructing enantiomerically enriched and functionalized aldehydes1,2,3,4,5,6,7,8,9,10,11. Oxidative cascade strategies, in which the iminium cation is generated in situ, are an attractive alternative to previous investigations in iminium catalysis and have been what developed because of its convenient one-pot-access, decreasing time and cost12,13.
In this context, oxidative cascade procedures involving the release of unstable enals in situ have been developed. In 2011, Rueping and co-workers reported enantioselective oxidative iminium activation for the functionalization of aldehydes from allylic alcohols14. Hayashi and Wang have independently described a new transformation, oxidative enamine catalysis for the direct asymmetric β-functionalization of simple aldehydes, in which direct oxidation of an enamine occurs to generate an iminium species followed by subsequent reaction with a nucleophile15,16.
Ethers are ubiquitous in nature and synthetic chemistry, and importantly, the stability of the C-O bond of ethers renders it a starting material that is easy to store. In particular, the oxidative cleavage of ether bonds to generate chemically reactive aldehydes or ketones should be useful for organic synthesis because it occurs under neutral and mild conditions and remains a topic of interest17,18,19.
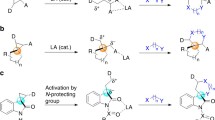
To our knowledge, the development of the oxidative cleavage of cinnamyl alkyl ethers to access enals-derived iminium ions should be an attractive synthetic strategy in which ethers can be used as “masked” aldehydes for further iminium catalysis. Therefore, we wondered whether an amino-catalyzed strategy using ether as a substrate could be devised for the cationic activation of ether oxidation of unsaturated imines for oxidative cascade processes (Fig. 1).
3-spirocyclopropyl -oxindole and -benzofuranone derivatives bearing a quaternary stereogenic center at the 3-position are known to exhibit remarkable biological and pharmaceutical activities20,21,22,23,24,25,26,27 and interest in synthetic methodologies for construction of these frameworks remains undiminished (Figs. 2 and 3)23,24,25,26,27.
Although some catalytic asymmetric methods can be used for the chemical synthesis of these scaffolds, the development of spiral rings with multiple stereocenters provided by ether-oxidation cascade strategies has not been realized. Motivated by the work of Wang and co-workers, who published a preliminary demonstration that organocatalytic asymmetric cascade reactions of enals with bromomalonates serve as a powerful approach to the preparation of highly enantio- and dia-stereoselective cyclopropanes28, we hypothesized that if racemic 3-bromobenzofuran-2-ones and 3-bromoindolin-2-ones29,30,31 with both electrophilicity and nucleophilicity could be successfully applied the interconversion between enantiomers properties in novel ether oxidation following asymmetric Michael addition initial cascade reaction, then stereoconvergent construction of 3-spirocyclopropyl-oxindoles and -benzofuranones could be accessed via the successful dynamic kinetic resolution of racemic 3-bromobenzo-five-membered-lactone/-lactam.
Results and discussion
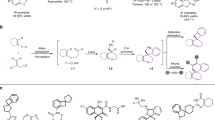
In order to explore the potential of the disclosed oxidative cascade methodology, we initiated our work by investigating the reaction of 3-bromooxindole (1a) and cinnamyl alkyl ether (2, n = 1) using 2,3-dichloro-5,6-dicyano-1, 4-benzoquinone (DDQ) as an oxidant in the presence of a secondary amine catalyst for promoting conversion of allyl ethers to α, β-spirocyclopropyl enals (Table 1, entries 1-16). Although DDQ has been shown to be an effective oxidant for the cleavage of ether bonds, a certain amount of water was necessary for the hydrolysis of the oxocarbenium-ion intermediate to complete the transformation to access aldehydes or ketones32,33
To our delight, after the introduction of a tert-butyl ether, an excellent yield and stereoselectivity could be achieved in the presence of DDQ and TMS-protected diphenyl prolinol catalyst (I) (entry 4). Other exploited alkyl ethers such as methyl, ethyl, and iso-propyl ethers were obtained with poor yields owing to the occurrence of excessive oxidation of the ether (entries 1-3). It was found that some oxidants such as IBX, MnO2 and TEMPO are ineffective for the ether oxidation process (entries 5-7). None of the chiral secondary amines IV-VI were suitable for this reaction in CHCl3 while II and III were found to be effective with good stereoselectivity although gave lower yields (entries 8-12). The effect of bases on the processes is evaluated next. In general, the reaction was found to be promoted by bases to furnish the products with good results, whereas NaOAc was the best choice as base for affording the product in the best result (entries 4, 13 and 14). It is also found that reactions performed well in less polar solvents such as CHCl3 rather than EtOH and DMF (entries 4 vs 15 and 16). This precedent pointed us toward the possibility of the Michael acceptor pent-2, 4-dienyl ethers (2, n = 2) for the oxidative vinylogous iminium activation strategy (entries 17–20). In contrast to cinnamyl tert-butyl ether, the unsaturated iminium ion intermediates were effectively generated in situ from pent-2, 4-dienyl ethyl ether without excessive oxidation by-products (entry 18). Typically, the π-orbital calculation of the LUMO of deconjugated iminium-ion mediated activation indicates that the β-C undergoing a nucleophilic attack is favoured over δ-C enabling 1, 4-addition instead of 1, 6-addition.
Subsequently, some of 3-spirocyclopropyloxindoles and 3-spirocyclopropyl-2-coumaranone 3 were prepared by the general method described above. As revealed in Table 2, remarkably, significant structural variation of allyl ethers, 3-bromooxindoles and 3-bromo-2-coumaranone could be applicable to the powerful one-pot oxidative cascade process to furnish the highly functionalized chiral cyclopropanes 3 in good yields with high levels of enantioselectivity and excellent diastereoselectivity. The electronic nature of the substituents of aromatic systems of 2 has some influence on the outcome. When it comes to cinnamyl tert-butyl ethers, higher reaction yields could be achieved for those with neutral and electron-withdrawing groups than heterocyclic and electron-donating groups (3a-3c, 3d-3e, 3g-3j and 3k-3l). Furthermore, when the reactant 3-bromooxindoles were changed to 3-bromo-2-coumaranone, the slightly lower reaction yields were observed but with good enantioselectivities (3a-3f, 3g-3l, 3m-3t and 3 u).
In this paper, the use of ethers as raw materials for the cyclopropanation has the dual advantages of reaction conditions and yields. Wang and co-workers28 used aldehyde compounds to realize the cyclopropanation. We found that the yield difference of this type reaction with ether or aldehyde as the substrate is not obvious, because the ether must be oxidized to aldehyde in the reaction process to participate in the reaction. Aldehydes are more reactive and less stable, and the reaction needs to be carried out at 0 °C. The method in this paper can complete the reaction at room temperature. The method of Chen and co-workers27 uses α, β-unsaturated acylphosphonates to complete the organocatalytic cyclopropanation reaction. The experimental results show that the acylphosphonates need a higher temperature (90 °C) to participate in the similar cyclopropanation, and the yield of this method is higher when the spiro structures of compounds 3a, 3c, 3d, and 3 h are synthesized.
A plausible reaction mechanism of the amino catalyst catalysed α, β-spirocyclopropanation reactions of alkyl allyl ether is illustrated in Fig. 4. The formation of oxocarbenium ion 4a can be initiated by the DDQ-mediated ether oxidation from 2a, we reason that the generated oxocarbenium ion was active enough and enal can be generated in situ to form the iminium ion intermediate 6a promoted by aminocatalysis. Then, intermolecular Michael addition happens, then the enamine intermediate 7a undergoes an intramolecular SN2 α-alkylation to produce α, β-spriocyclopropane 3a.
The isomers R-1a and S-1a of feedstock 1a can be interconverted by the enol tautomer 9a of 1a. Meanwhile, R-1a is the dominant isomer in the reaction with 6a, and the reaction between R-1a and 6a will lead to the continuous transformation of S-1a to R-1a, so as to generate the product 3a with a single configuration (Fig. 4).
In conclusion, we have developed an umpolung ether oxidation iminium-ion activation process to afford spirocyclopropyl benzo-five-membered -lactone and -lactam cores with three adjacent stereo centres and an active aldehyde group which can be transferred to bioactive molecules with warhead targeting to the special drug targets, from 3-bromooxindoles or 3-bromo-2-coumaranone with allyl tert-butyl ethers or pent-2, 4-dienyl ethyl ethers. Compared with the similar methods in the previous literature using aldehyde and acyl phosphonate as the substrate, this method uses ether as the substrate, the reaction conditions are milder, and the synthesis of some compounds has an advantage in yield. And put forward a hypothesis that the product is almost a single configuration. This hypothesis assumes that the racemic benzo five-membered heterocycle undergoes enol tautomerization under the induction of a chiral secondary amine resulting in a single configuration of the product. This study and synthetic application of this strategy provides a new amino catalytic cascade strategy tool for the efficient synthesis of complex drug molecules.
Methods
General procedure for synthesis of 3a-3u
A solution of 1 (1.0 mmol) in CHCl3 (6 mL) was add of 2 (1.2 mmol), DDQ (1.2 mmol), NaOAc (2.0 mmol) and (S)-diphenyl prolinol trimethylsilyl ether (0.2 mmol) following stirred at rt for 24 h. After the reaction completed, the mixture was filtered and the filtrate was removed by vacuum distillation. The crude product was purified by silica gel chromatography to obtain 3a-3u. See Supplementary Notes 1, 2 for experimental details and compound characterization data. See Supplementary Figs. 1–45 for 1H NMR and 13 C NMR and Supplementary Figs. 46–87 for HPLC spectra.
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its Supplementary Information file.
References
Marson, C. M. Multicomponent and sequential organocatalytic reactions. Diversity with atom-economy and enantiocontrol. Chem. Soc. Rev. 41, 7712–7722 (2012).
Albrecht, L., Jiang, H. & Jørgensen, K. A. A Simple recipe for sophisticated cocktails: organocatalytic one-pot reactions—concept, nomenclature, and future perspectives. Angew. Chem., Int. Ed. 50, 8492–8509 (2011).
Moyano, A. & Rios, R. Asymmetric organocatalytic cyclization and cycloaddition reactions. Chem. Rev. 111, 4703–4832 (2011).
Grondal, C., Jeanty, M. & Enders, D. Organocatalytic cascade reactions as a new tool in total synthesis. Nat. Chem. 2, 167–178 (2010).
Danijel, G. & Matija, G. Organocatalytic asymmetric transformations of 3-Substituted 3-Hydroxyisoindolinones. Synlett 28, 889–897 (2017).
Sun, L., Chen, X. Y. & Dieter, E. Aldehyde catalysis: new options for asymmetric organocatalytic reactions. Chem 4, 2026–2028 (2018).
List, B. & Yang, J. W. The organic approach to asymmetric catalysis. Science 313, 1584–1586 (2006).
MacMillan, D. W. C. The advent and development of organocatalysis. Nature 455, 304–308 (2008).
Erkkilä, A., Majander, I. & Pihko, P. M. Iminium catalysis. Chem. Rev. 107, 5416–5470 (2007).
Tsogoeva, S. B. Recent Advances in asymmetric organocatalytic 1,4-conjugate additions. Eur. J. Org. Chem. 11, 1701–1716 (2007).
Dominika, K., Maria, M., Erik, R. & Radovan, S. Mechanochemically activated asymmetric organocatalytic domino mannich reaction-fluorination. ACS Sustain. Chem. Eng. 8, 14417–14424 (2020).
Taylor, R. J. K., Reid, M., Foot, J. & Raw, S. A. Tandem oxidation processes using manganese dioxide: discovery, applications, and current studies. Acc. Chem. Res. 38, 851–869 (2005).
Tietze, L. F. Domino reactions in organic synthesis. Chem. Rev. 96, 115–136 (1996).
Rueping, M., Sundén, H., Hubener, L. & Sugiono, E. Asymmetric oxidative lewis base catalysis—unifying iminium and enamine organocatalysis with oxidations. Chem. Commun. 48, 2201–2203 (2012).
Hayashi, Y., Itoh, T. & Ishikawa, H. Oxidative and enantioselective cross-ccoupling of aldehydes and nitromethane catalyzed by diphenylprolinol silyl ether. Angew. Chem., Int. Ed. 50, 3920–3924 (2011).
Zhang, S. L. et al. Organocatalytic enantioselective β-functionalization of aldehydes by oxidation of enamines and their application in cascade reactions. Nat. Commun. 2, 211–218 (2011).
Kamijo, S., Matsumura, S. & Inoue, M. CCl3CN: A crucial promoter of mCPBA-mediated direct ether oxidation. Org. Lett. 12, 4195–4197 (2010).
Li, X. B. et al. Ipso-oxidation of allyl ether/decarboxylative aromatization cascade strategy via oxocarbenium activation: A novel approach for synthesis of N-Alkyl Pyrroles. Chin. J. Org. Chem. 39, 2906–2911 (2019).
Li, T. T., Gerald, B. H. & Xu, B. Cobalt-catalyzed aerobic oxidative cleavage of alkyl aldehydes: synthesis of ketones, esters, amides, and α-ketoamides. Chem. Eur. J. 38, 9737–9741 (2021).
Jiang, T. et al. Design, synthesis and biological evaluations of novel oxindoles as HIV-1 non-nucleoside reverse transcriptase inhibitors. Part I. Biorg. Med. Chem. Lett. 16, 2105–2108 (2006).
Jiang, T. et al. Design, synthesis, and biological evaluations of novel oxindoles as HIV-1 non-nucleoside reverse transcriptase inhibitors. Part 2. Biorg. Med. Chem. Lett. 16, 2109–2112 (2006).
Kapure, J. S. et al. Diastereoselective synthesis of spiro[cyclopropane-1,3’-indolin]-2’-ones through metal-free cyclopropanation using tosylhydrazone salts.RSC Advances 72, 38425–38432 (2014).
Hong, L. & Wang, R. Recent advances in asymmetric organocatalytic construction of 3,3′-Spirocyclic oxindoles. Adv. Synth. Catal. 355, 1023–1052 (2013).
Noole, A. et al. 3-Chlorooxindoles: versatile starting materials for asymmetric organocatalytic synthesis of spirooxindoles. Adv. Synth. Catal. 355, 829–835 (2013).
Marti, C. & Carreira, E. M. Total synthesis of (-)-spirotryprostatin B: synthesis and related studies. J. Am. Chem. Soc. 127, 11505–11515 (2005).
Wood, J. L. et al. Application React. enols Synth.: A versatile, Effic., stereoselective Constr. welwitindolinone carbon Skelet. 121, 6326–6327 (1999).
Chen, L. & He, J. DABCO-catalyzed michael/alkylation cascade reactions involving α‑substituted ammonium ylides for the construction of spirocyclopropyl oxindoles: access to the powerful chemical leads against HIV‑1. J. Org. Chem. 85, 5203–5219 (2020).
Xie, H., Zu, L., Li, H., Wang, J. & Wang, W. Organocatalytic enantioselective cascade michael-alkylation reactions: synthesis of chiral cyclopropanes and investigation of unexpected organocatalyzed stereoselective ring opening of cyclopropanes. J. Am. Chem. Soc. 129, 10886–10894 (2007).
Zheng, W., Zhang, Z., Kaplan, M. J. & Antilla, J. C. Chiral calcium VAPOL phosphate mediated asymmetric chlorination and michael reactions of 3-substituted oxindoles. J. Am. Chem. Soc. 133, 3339–3341 (2011).
Movassaghi, M., Ahmad, O. K. & Lathrop, S. P. Directed heterodimerization: stereocontrolled assembly via solvent-caged unsymmetrical diazene fragmentation. J. Am. Chem. Soc. 133, 13002–13005 (2011).
Zhao, M. X., Zhang, Z. W., Chen, M. X., Tang, W. H. & Shi, M. Cinchona alkaloid catalyzed enantioselective chlorination of 3-aryloxindoles. Eur. J. Org. Chem. 16, 3001–3008 (2011).
Bai, H. T., Lin, H. C. & Lui, T. Y. Phenanthrene-tethered furan-containing cyclophenes: synthesis and photophysical properties. J. Org. Chem. 75, 4591–4595 (2010).
Tohyama, S., Choshi, T., Azuma, S., Fujioka, H. & Hibino, S. A new synthetic route to the 1-oxygenated carbazole Aalkaloids, mukonine and clausine E (clauzoline I). Heterocy-cles 79, 955–965 (2009).
Acknowledgements
We gratefully acknowledge financial support from the National Science Foundation of China (20972051 and 21476078, X.-H.Y.), (08431901800 and 08430703900, X.-H. Y.).
Author information
Authors and Affiliations
Contributions
Y.X. and Z.J. designed the experiments. H.Y. and Y.J. performed the experiments, and prepared this manuscript and the supplementary information. L.Z. and Z.L. analysed the data.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Chemistry thanks Ciaran Seath and Wanbin Zhang for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hu, Y., Yuan, J., Li, Z. et al. Asymmetric α-spirocyclopropanation of oxindoles and benzofuranones via dynamic kinetic resolution. Commun Chem 5, 106 (2022). https://doi.org/10.1038/s42004-022-00695-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-022-00695-3
- Springer Nature Limited