Abstract
In a multicenter case–control investigation, we assessed the efficacy of the Electrooculogram Signal Analysis (EOG-SA) method, which integrates attention-related visual evocation, electrooculography, and nonlinear analysis, for distinguishing between intellectual and developmental disabilities (IDD) and typical development (TD) in children. Analyzing 127 participants (63 IDD, 64 TD), we applied nonlinear dynamics for feature extraction. Results indicated EOG-SA’s capability to distinguish IDD, with higher template thresholds and Correlation Dimension values correlating with clinical severity. The template threshold proved a robust indicator, with higher values denoting severe IDD. Discriminative metrics showed areas under the curve of 0.91 (template threshold) and 0.85/0.91 (D2), with sensitivities and specificities of 77.6%/95.9% and 93.5%/71.0%, respectively. EOG-SA emerges as a promising tool, offering interpretable neural biomarkers for early and nuanced diagnosis of IDD.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Intellectual and developmental disabilities (IDD) encompass a spectrum of neurodevelopmental disorders that emerge during developmental phases, hallmarked by intellectual impairments and the inability to attain key developmental milestones1,2,3,4. These disorders frequently present comorbidly with behavioral conditions, including autism spectrum disorder (ASD) and cerebral palsy (CP), and exert significant societal, economic, and emotional impacts5,6,7,8. With a prevalence rate of 1–3% and an upward trend correlating with societal progress9,10,11, comprehensive assessments and tailored interventions are essential for optimal development.
Despite the importance of early identification and intervention for individuals with IDD12, current clinical practice relies heavily on parental questionnaires and expert clinical assessment rather than direct child observation13. Although advancements in gene microarray analysis and imaging studies14,15,16, the complex etiology and heterogeneous clinical manifestations of IDD make early and differential diagnosis from typical development (TD) challenging17,18,19.
Studies affirm the link between early infant gaze and communication and social abilities, with eye-gaze avoidance, a common feature in IDD, potentially affecting social engagement20,21,22,23. Eye movement analysis, reflecting brain function and behavior, is further emphasized by the intricate communication between nearly all cerebral regions and the ocular motor control system24,25,26,27,28. Benjamin et al.’s study supports this, indicating eye behavior as a reflection of attention deficits in fragile X syndrome, ASD, and TD individuals29. Thus, eye-movement patterns and visual attention may serve as promising biomarkers for cognitive and developmental disparities, aiding diagnosis and treatment of various diseases30,31,32,33,34.
Eye-tracking technology, including EOG, eye-tracking glasses, and remote eye trackers, is typically used to capture eye movement patterns such as saccades and fixations35,36,37,38. Among them, EOG, a non-invasive method quantifying electrical potential changes between the cornea and retina from eyeball movement, has been a key tool since the 1970s39,40,41. Its applications extend to diagnosing retinal diseases and facilitating EOG-based eye-writing communication42,43,44. Consequently, this study proposes EOG’s critical role in capturing visual attention allocation dynamics, reflecting attention processes in real-world scenarios. Nonetheless, the automatic, rapid, and non-repetitive nature of this phenomenon may hinder comprehensive EOG signal (EOGs) analysis45,46.
Our team has previously developed the Attention-related Visual Evocation System to address the challenge of EOG signal analysis. This system elicits attentional activation in children while simultaneously recording their EOGs during horizontal smooth pursuit with eyes viewing. The aim was to investigate attention-related phenomena in a controlled standard setting47. Given the suggested correlation between eye movement patterns and EOG waveforms48, we utilized the template matching technique to capture eye movements. The method’s efficacy was tested on a cohort of 4 TD individuals and 4 individuals with ASD, showing reliability and initial indications of promise in identifying specific subgroups within the IDD population49.
This study aimed to quantify attention in order to elucidate significant attentional disparities in individuals with IDD. We employed the Attention-related Visual Evocation System for precise EOG recordings and applied nonlinear dynamics methodologies for feature extraction, forming the EOG Signal Analysis (EOG-SA) method. We expanded our child participant sample and compared visual attention patterns in children with IDD and a matched control group using controllable standard visual stimuli, then contrasted these results with conventional assessment scales. We also explored various scenarios to understand real-life visual attention difficulties. This comprehensive approach underscored the potential of EOG-SA markers as a significant biobehavioral measure for IDD, promising to advance early detection, risk assessment, and personalized medicine progression (Fig. 1).
Conceptual Framework of the Study. The images were created by Shi-yi Qi, one of the co-authors as the copyright holder. A schematic representation delineating the methodological approach and investigative workflow employed in the study, encapsulating the essential phases of investigation, data analysis procedures, and overarching research objectives.
Methods
Participants
This investigation presents a multicenter case–control study. Participants were recruited from the Affiliated People’s Hospital of Fujian University of Traditional Chinese Medicine, Fujian Maternity and Child Health Hospital, and the Third People’s Hospital Affiliated to Fujian University of Traditional Chinese Medicine. The study protocol was approved by the Clinical Research Ethics Committees of the participating hospitals (approval numbers: 2021-016-02, 2021YJ042, 2020-kl-042). Registration of the study with the Chinese Clinical Trial Registry was completed, bearing the identifier ChiCTR-TRC-2200058387, and the initial trial registration date was scrupulously logged as 08/04/2022. Adhering to the stringent ethical standards of research, all study procedures were conducted in strict compliance with the updated Declaration of Helsinki, and we affirm that all methods were carried out in accordance with the relevant guidelines and regulations.
Participants in the IDD group met DSM-5 diagnostic criteria2, were aged 3–9, of either gender, and had guardian consent. Exclusion criteria included epilepsy, severe mental disorders, significant cerebral involvement (confirmed by CT or MRI), ocular surgery or disease history, and current medication use. The control group (TD) comprised age and sex-matched, clinically healthy children with typical development.
Child neurodevelopment assessment
The Infants-Junior Middle School Students’ Social-Life Abilities Scales (S-M scale), adapted from the Japanese S-M social life ability test50, was used to assess developmental and social maturity in children aged 6 months to 14 years. It covers six domains: self-dependence, locomotion, work skills, communication, socialization, and self-management51,52. For children under 6, the Gesell Developmental Schedule (GDS) was used for subgroup analysis. The GDS, a global child development scale developed by Yale University53, assesses neurological and intellectual growth in infants aged 1 month to 6 years across five domains: gross motor, fine motor, linguistic, adaptive, and personal-social skills. Results were expressed as a developmental quotient (DQ)54.
Apparatus
EOGs collecting procedure, attention-related visual evocation system
Following our previous work47,55, we devised an Attention-related Visual Evocation System to evoke attentional responses. This system included a display screen, visual stimuli, and a dynamic interface (Fig. 2A). Participants, either independently or with their parents, were positioned approximately 0.5 m from the screen to view video sequences. The procedure began with a cartoon image moving leftward and rightward in each 6-s cycle. The image was replaced in each cycle, with the entire process lasting for 60 s (Fig. 2 B/C). The system aims to provide a reliable tool for investigating attention-related phenomena in a controlled standard setting.
Experimental Apparatus and Setup Configuration. The images were created by Shi-yi Qi, one of the co-authors as the copyright holder. A comprehensive visual array depicting the experimental tools and environment utilized in the study. (A) Electrooculography (EOG) acquisition system for monitoring eye movements. (B) User interface of the Attention-related Visual Evocation System, designed for stimulus presentation. (C) Sample video sequence as displayed by the Attention-related Visual Evocation System. (D) Diagrammatic representation detailing the specific placements of electrodes for EOG data collection. (E) Illustrative overview of the three experimental scenarios employed.
We also developed an EOGs collection system utilizing a 24-bit A/D conversion chip ADS1299 and STM32F407 control chip. This system incorporated three additional electrodes for EOG collection, designed to study smooth pursuit eye movements. The horizontal EOGs were recorded by placing two electrodes at the outer canthus of the eyes and a reference electrode on the forehead (Fig. 2D). The impedance of all electrodes was maintained below 5 kΩ and the sampling was programmed using LabVIEW 7.1 (National Instruments, Austin, TX USA), with a sampling rate of 1000 Hz.
Evaluate the discrimination degree of application for EOG-SA across three different scenarios
To assess the applicability of EOG-SA across varied scenarios, we conducted three daily behavior tests (Fig. 2E) representing standard, guided, and freeplay scenarios. Each scenario represents a different meaning, with the standard scenario requiring participants to follow a predetermined eye movement trajectory, the guide scenario involving social interaction, and the freeplay scenario involving an object of interest. The specific set-up for each test was as follows:
Standard scenario Participants watched video sequences from the Attention-related Visual Evocation System for a duration of 5 min.
Guide scenario Participants were instructed by trained researchers to engage in a 5-min reading session with a children’s book.
Freeplay scenario Participants engaged in unstructured play with toys within the testing room for 5 min.
To ensure reliability, pediatricians were extensively trained and certified at the Affiliated People’s Hospital of Fujian University of Traditional Chinese Medicine, and regular quality-control examinations were conducted.
Ethics declarations
This investigation presents a multicenter case–control study. Participants were recruited from the Affiliated People’s Hospital of Fujian University of Traditional Chinese Medicine, Fujian Maternity and Child Health Hospital, and the Third People’s Hospital Affiliated to Fujian University of Traditional Chinese Medicine. The study protocol was approved by the Clinical Research Ethics Committees of the participating hospitals (Approval Numbers: 2021-016-02, 2021YJ042, 2020-kl-042). Registration of the study with the Chinese Clinical Trial Registry was completed, bearing the identifier ChiCTR-TRC-2200058387, and the initial trial registration date was scrupulously logged as 08/04/2022. Adhering to the stringent ethical standards of research, all study procedures were conducted in strict compliance with the updated Declaration of Helsinki, and we affirm that all methods were carried out in accordance with the relevant guidelines and regulations.
Data preprocessing, and feature extraction
Data preprocessing
EOGs span an amplitude range of 50–3500 µV and a frequency range of 0.1–10 MHz56,57. As in previous work47, preprocessing of raw data was required to eliminate noise, using a 6-order Butterworth filter as a 50 Hz notch filter and a Chebyshev low-pass filter to remove frequencies above 10 Hz. Further analysis was performed utilizing Matlab (Matlab R2022a, The MathWorks Inc., Natick, Massachusetts). Unconscious blinking, an inevitable artifact during EOG recording, was eliminated using the Moving-Average Method, which calculates the mean and standard deviation of the instantaneous spike rate within a moving interval58. It is capable of effectively removing jitter, resulting in smoother EOGs that facilitate the identification of targeted eye movements. A sliding window with length of 500 sampling points and a step size of 1 sampling point was employed47.
Feature extraction
Dynamic time warping (DTW) for EOG waveforms
In this study, we combined human-labeling and computer vision techniques to monitor attentional state and calibrate eye movements, using a hybrid approach. Initially, eye movement data (EOGs) representing the subject’s attentional state were generated, and subsequently used as templates in Dynamic Time Warping (DTW) to measure similarity with test EOGs47. This process involved expert clinicians assessing attentional state during template construction. Attention-related Visual Evocation System was used to engage participants in observing video sequences while clinicians recorded and marked time intervals. Three marker intervals were necessary, as illustrated in Fig. 2B. If test EOG waveforms resembled template waveforms within a 6-s timeframe, they were identified as target EOGs for that interval, indicating focused attention.
The DTW algorithm calculated the similarity metric, functioning as a distance measure capable of handling non-linear distortion and phase errors59,60,61. This algorithm uses a dynamic programming approach to determine minimal distance between two time series, improving waveform alignments62. Typically, the distances between data points reflect similarity, with closer points indicating higher similarity63. The computation of DTW distance is carried out as follows: the proximity of data points is indicative of their level of similarity, the template and test EOGs are denoted by M = (m1, m2, m3 . . . mn) and T = (t1, t2, t3 . . . tq), respectively, where n and q represent the length of the signals. Firstly, the matrix D with i rows and j columns is established as the sequence distance matrix, where D (i, j) = (mi − tj)base. Generally, the squared Euclidean distance is used as the base distance are the Euclidean distances from the point to point of the M and T sequences in the corresponding rows and columns. This approach facilitates the generation of a cost matrix C (i, j), wherein C (1,1) corresponds to D (1,1), and C-Values are calculated using Eq. (1):
The computation of distances between M and T yielded C (n, q).
To enhance identification and evaluation robustness, we refined the Template threshold determination using the mentioned techniques. Clinicians annotated three to five attentional time intervals, indicating the corresponding system boxes. The template threshold test was performed thrice, each lasting 60 s. Subsequently, we calculated the DTW distances between the template and EOG waveforms within these intervals, using the average distance as the Template threshold.
Furthermore, our system interface provided real-time DTW values every 6 s, which we will refer to as RT-DTW in the following sections (Fig. 2B).
Approximate entropy (ApEn) for EOG signals
Introduced by Pincus in 199164, Approximate entropy (ApEn) is a nonlinear statistic quantifying regularity and complexity in time series data, particularly suitable for short, noisy series65,66. Higher ApEn values represent increased fluctuations or irregularities, while lower values signify reduced fluctuation67. This methodology has been successfully applied to physiological time series analysis68,69. The formula for ApEn is defined as follows:
In our study, we acquired 5000 longitudinal data sets and calculated a single ApEn value (m = 2, R = 0.2sd) for every 1000 data points using the “pracma” command in R programming language (version 3.6.3). The average of the five computed results was then determined.
Correlation Dimension (D2) for EOG signals
The correlation dimension (D2), another method to measure time series complexity, was introduced by Grassberger and Procaccia in 198370. It characterizes chaotic attractors by evaluating their estimated geometric structure71.
Consider \(X_{n}\), a vector of length m, given by:
Among these variables, m represents the embedding dimension, τ denotes the time delay, and \(N_{m}\) signifies the dimension of the phase-space.
Let \(C\left( r \right)\), , referred to as the correlation integral, be ascertained as follows:
The D2 can be defined as the limit value:
In this study, we quantified D2 values by determining the regression curve slope using Matlab.
To sum up, the signals extracted subsequent to the aforementioned process, referred to as Electrooculogram Signal Analysis (EOG-SA) in this context, will be subjected to an exploration of their correlation with disease scales.
Statistical analysis
The statistical analysis was conducted using SPSS (version 22 for Windows, SPSS Inc., USA). Continuous variables were presented as mean ± standard deviation (SD), while categorical variables were detailed as frequencies (percentages). Upon confirming the non-normal distribution of the data through normality tests, we employed Spearman coefficients for correlation assessments and utilized either the Independent t-test or Mann–Whitney rank-sum test for comparisons between two groups. For comparisons involving more than two groups, Kruskal Wallis with Dunn’s multiple comparison post-test was utilized. Linear regression analysis was applied for continuous outcome variables, while logistic regression was used to model associations with Visual Attention Patterns for binary outcome variables. The ROC curve was employed for further validation. All tests were two-tailed, with P < 0.05 denoting statistical significance.
Results
Demographic and clinical characteristics of participants
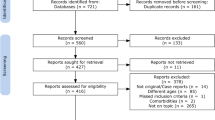
From January 2021 to March 2023, we recruited participants from respected medical institutions including the Affiliated People’s Hospital of Fujian University of Traditional Chinese Medicine, Fujian Maternity and Child Health Hospital, and the Third People’s Hospital Affiliated to Fujian University of Traditional Chinese Medicine. Of the 140 enrolled families, 13 were excluded due to data loss, comorbid conditions, or other reasons, leaving 127 participants. The study was successfully completed by 63 patients with IDD and 64 healthy adolescents, all of whom were right-handed Asians. Parents provided informed consent and no adverse events were reported. Details on participant selection and demographics are illustrated in Fig. 3 and Table 1, respectively.
Comparative analysis of EOG signal analysis (EOG-SA) in two distinct developed profile groups of children
In our study, we extract features from EOGs recorded in the Attention-related Visual Evocation System, hereby denoted as EOG Signal Analysis (EOG-SA), specifically referring to the template threshold, ApEn, and D2 values. As shown in Table 2, the IDD group displayed significantly higher template threshold and D2 values than the TD group (P < 0.001), but the ApEn values did not significantly differ between the groups (P > 0.05).
Examination of the relationship between scale and EOG-SA
We further probed the association between scales and EOG-SA, encompassing comparative analysis across different scales in two groups, and the relationship of EOG-SA with developmental levels and disease severity.
The IDD group exhibited significantly higher template threshold and D2 values on both the S-M scale and GDS compared to the TD group (P < 0.001), but not ApEn (P > 0.05) (Fig. 4 A/D). Moreover, significant associations were observed between template threshold/D2 values and individual items and total score of the scales (P < 0.001), while ApEn values correlated with S-M scale items, excluding total score and work skills (P > 0.05), but not with GDS (P > 0.05) (Fig. 4 B/E).
Correlating Intellectual and Developmental Disabilities (IDD) with Electrooculogram Signal Analysis (EOG-SA) Metrics. A comprehensive analysis illustrating the association between IDD and various EOG-SA parameters. (A) Illustrates significant differences in template threshold, approximate entropy (ApEn), and correlation dimension (D2) values between control and IDD groups, as gauged by the total S-M scale score (***P < 0.001). (B) Depicts the results from Spearman’s correlation analysis linking template threshold, ApEn, and D2 values with the S-M scale. (C) Presents a comparative analysis of the aforementioned EOG-SA parameters across different severity grades of IDD, as delineated by the S-M scale score (**P < 0.01, ***P < 0.001 versus borderline; #P < 0.05 versus mild). (D) Highlights differences in EOG-SA metrics between groups based on the total GDS scale score (***P < 0.001). (E) Shows Spearman’s correlation analysis outcomes for template threshold, ApEn, and D2 values in relation to the GDS. (F) Compares EOG-SA parameters across IDD severity grades as defined by the GDS score, indicating significant variations (***P < 0.001 versus borderline; ##P < 0.01 versus mild or moderate).
Finally, upon stratifying disease severity according to total S-M or GDS scale score (Table 3), we examined EOG-SA’s correlation with disease severity in TD/DD individuals, aiming to identify visual attention patterns associated with varying developmental delay degrees. Severity of the condition was determined using the total S-M score72,73, with our data indicating a negative correlation between severity scores on the S-M scale and both thresholds and D2 (r = − 0.77, P < 0.001; r = − 0.63, P < 0.001), whereas ApEn did not show significant correlation (P > 0.05). Notably, individuals with moderate and severe or profound IDD displayed significantly higher template thresholds than those with borderline IDD (P < 0.001). However, no significant differences were found in template thresholds between patients with borderline, mild, and moderate IDD (P > 0.05), nor in terms of ApEn and D2 (P > 0.05) (Fig. 4C). Furthermor, when severity was classified based on the total GDS score54,74, a clear negative correlation was found between severity scores on the GDS scale and both thresholds and D2 (r = − 0.81 P < 0.001; r = − 0.71 P < 0.001). Patients with severe or profound IDD had significantly higher template thresholds compared to those with borderline or mild or moderate IDD (P < 0.001, P < 0.01). Yet, no significant differences were observed in other indicators (P > 0.05) (Fig. 4F).
Linear regression analyses
We conducted a series of linear regression analyses to evaluate the influence of participant age, gender, and developmental outcomes on EOG-SA. Age, gender, or each subscale of the S-M scale or GDS were used as independent variables, with EOG-SA as the dependent variable. The results revealed a significant effect of age on ApEn (β = − 0.001, P = 0.02). Furthermore, personal-social skills significantly impacted template threshold (β = − 0.81, P = 0.03), while both gross motor and personal-social skills influenced D2 significantly (β = − 0.03, P = 0.01; β = − 0.83, P = 0.02). These positive findings were visually represented using scatter plots and regression lines (Fig. 5A). Further details are provided in Table 4.
Evaluating Electrooculogram Signal Analysis (EOG-SA) Across Diverse Scenarios Using Linear Regression and Discrimination Metrics. (A) Depicts the outcomes of linear regression analysis applied to EOG-SA data. (B) Provides a comparative assessment of real-time Dynamic Time Warping (DTW), approximate entropy (ApEn), and correlation dimension (D2) values across three distinct scenarios, revealing significant disparities (***P < 0.001 versus standard scenario; ###P < 0.001, ##P < 0.01, comparisons between TD individuals and those with IDD).
Analysis results on discrimination degree of EOG-SA application across scenarios
Our subsequent analysis sought to discern potential variations in EOG-SA application across different groups and scenarios. The IDD group showed significantly higher real-time DTW(RT-DTW) and D2 values than the TD group across scenarios (###P < 0.001, ##P < 0.01), but no significant ApEn differences were observed (P > 0.05). Both TD and IDD groups displayed significantly increased RT-DTW and ApEn in guide and freeplay scenarios compared to the standard scenario (P < 0.001), with no significant D2 variation (P > 0.05) (Fig. 5B).
Results of binary logistic regression analysis and ROC curve evaluation
Finally, binary logistic regression analysis was employed to examine the relationship between EOG-SA and IDD, followed by ROC curve analysis for further evaluation. Independent variables included EOG-SA measures (template threshold, ApEn, and D2 values), while the dependent variable was the presence or absence of IDD, diagnosed using the S-M scale or GDS.
Binary logistic regression results, based on the S-M scale, indicated significant influences of both template threshold and D2 values on the presence of IDD (β = 0.06, P = 0.000; β = 1.12, P = 0.001), with ApEn showing no effect (β = 2.30, P = 0.25). Furthermore, ROC curve analysis revealed AUCs of 0.91 and 0.85 for the template threshold and D2, respectively (Fig. 6A). Using the Youden method, threshold values for template threshold and D2 were determined as 79 and 2.19, respectively, with sensitivities of 74.2% and 90.3%, and specificities of 95.0% and 63.3%.
Performance Analysis of Disease Prediction Models Using Electrooculogram Signal Analysis (EOG-SA). This figure presents the analytical performance of EOG-SA-derived metrics in disease prediction models. Receiver Operating Characteristic (ROC) curve analyses are illustrated, with template threshold depicted in blue and Correlation Dimension (D2) in red, across both the S-M scale and Global Deterioration Scale (GDS). The diagnostic sensitivity and specificity are represented by paired columns in shades of blue and purple, respectively, for Dynamic Time Warping (DTW) and D2 within these assessment scales. A. Within the S-M scale context, DTW achieved a sensitivity of 74.2% and a specificity of 95.0%, whereas D2 reached a sensitivity of 90.3% and a specificity of 63.3%. B. Within the GDS framework, DTW’s sensitivity and specificity stood at 77.6% and 93.5%, respectively, while D2 displayed sensitivity and specificity of 95.9% and 71.0%, correspondingly.
Further analysis based on GDS revealed significant effects of both template threshold and D2 values on the presence of IDD (β = 0.06, P = 0.002; β = 1.64, P = 0.003), with no effect from ApEn (β = 2.19, P = 0.45). ROC curve analysis yielded AUCs of 0.91 for both measures, with thresholds of 79 and 2.19, respectively. Sensitivity and specificity at these thresholds were 77.6% and 95.9% for template threshold, and 93.5% and 71.0% for D2 (Fig. 6B).
Discussion
This study evaluated cognitive and developmental quotient using the S-M scale, and GDS for certain age-specific participants. The findings revealed higher template threshold and D2 values in individuals with IDD than those with TD, indicating excellent discriminant validity. Both values correlated well with both scales, demonstrating robust criterion validity. The template threshold showed a stronger association with all parameters than D2 and could discriminate the severity of IDD. Despite no statistical significance between borderline and mild IDD, similar trends were observed across both scales, possibly due to the limited sample size. Consequently, the internal consistency of the template threshold was deemed acceptable, affirming its exceptional properties.
The linear regression results showed a significant effect of personal-social skills on template threshold, suggesting that higher personal-social skills correlate with lower template thresholds. This aligns with prior research linking visual patterns to social disability27,75. Similar effects were observed for D2, with both gross motor and personal-social skills significantly impacting. Gross motor variables unexpectedly emerged from the analyses, providing external validity to our findings, and supporting previous studies linking gross motor development to cognition76,77 and development78,79. Scholars have found that patients with developmental dyscalculia exhibit deficits in visual perception and motor integration, as well as mildly delayed gross motor milestones80,81. Current research focuses on links between visual characteristics, social and cognitive deficits, and developmental disorders in IDD patients. Our study highlights the potential value of EOG analysis with D2 for assessing gross motor development. The lack of a relationship in the S-M scale remains unclear, possibly due to the age specificity of the GDS, used for children under six82. This age-related difference may be more pronounced at younger ages, a view supported by our findings with ApEn. Future age-stratified studies are planned to test this hypothesis.
Furthermore, to integrate eye-movement and attentional state recognition into social interactions, we evaluated the performance of EOG-SA in three distinct scenarios. Each task, from tracking a pre-defined eye movement trajectory in the standard scenario, engaging in social interaction in the guide scenario, to focusing on an object within an unbounded setting in the freeplay scenario, conveyed a unique connotation.
Here, employing real-time DTW (RT-DTW) calculations at 6-s intervals, we observed distinct visual attention patterns between individuals with IDD and TD across various scenarios, demonstrating the efficacy of RT-DTW and D2 in discerning individuals with TD from those with IDD. Despite higher RT-DTW and ApEn values in the guide and freeplay scenarios compared to the standard,, suggesting that these scenarios require more attention and cognitive effort, no significant differences were noted between the guide and freeplay scenarios. This may be due to the heightened variability of eye movements in real-world settings, whereas the templates utilized in our analysis were derived from the standard scenario, thereby limiting their ability to differentiate between these scenarios. Our results underscore the need for individualized attention pattern consideration in designing interventions for IDD. While D2 analysis effectively differentiated between IDD and TD, it failed to distinguish between scenarios. Interestingly, ApEn analysis, though less effective in disease classification, successfully distinguished between scenarios While no significant differences were detected between the guide and freeplay scenarios, a slight inclination towards higher values in the freeplay scenarios was observed in both groups.
In IDD clinical trials, precise discernment of attentional alterations is crucial for early disease diagnosis and effective monitoring83,84. Hence, enhancing the sensitivity and objectivity of outcome measures has been a paramount pursuit for researchers85,86.
Eye movement patterns encompass the minute, involuntary ocular motions that transpire during diverse visual tasks. Traditional devices, such as eye-tracking glasses or remote eye trackers, employ infrared light to meticulously track the eyes’ position and record their intricate movements87,88,89. These investigations commonly assess the duration, frequency, and dwell time of gaze on different elements of a visual scene, including facial features and areas of interest (AOIs)90,91,92,93. While these conventional methods provide valuable insights into information processing94,95,96, they are not typically utilized in social contexts97. Given the complex and dynamic nature of visual attention98, relying solely on fixation patterns and absolute percent fixation times may fail to capture subtle eye movement patterns, ultimately leading to misleading conclusions that lack sensitivity and objectivity99. Thus, while eye-tracking studies are promising, their outcomes often apply more to groups than individuals100,101, offering limited clinical insights.
EOG, with its high signal-to-noise ratio, objectivity, sensitivity, and ease of recording, is valuable in situations where conventional eye-tracking devices may be impractical102,103,104. It allows for the evaluation of time-dependent visual attention dynamics and the transformation of eye-tracking metrics into practical biomarkersrather than mere proportional distribution measures100. Expanding this research to encompass natural social interactions and settings is crucial, as it can provide quantifiable indicators of cognitive processes and attentional focus associated with eye movements105, complementing psychophysical and neural cognition measurements (Fig. 7).
Comparative Analysis of Measured Parameters in Electrooculography and Eye-Tracking Technologies. The schematic delineates a side-by-side comparison of the principal parameters measured by electrooculography (EOG) and eye-tracking technologies, illustrating the distinct performance characteristics and methodological differences of each technique in monitoring ocular movements.
Finally, to further validate the robustness of our study, specifically the assessment of the template threshold and D2 as valuable methods for distinguishing between TD and IDD, we employed Binary Logistic Regression Analysis Results to prevent overfitting. The classifier modeling performance was appraised via the ROC curve. The AUCs of the template threshold were 0.91 on both scales, whereas the AUCs of D2 were 0.85 (S-M scale) and 0.91 (GDS), respectively, yielding the largest Youden indexes and optimal cut-off points of the elastic strain ratio (79 for template threshold and 2.19 for D2). These cutoffs were evaluated utilizing the S-M scale for the former and the GDS for the latter, resulting in a sensitivity of 74.2% and 77.6% for the template threshold, and a specificity of 95.00% and 93.5%, respectively. As for D2, the sensitivities were 90.3% and 95.9%, and the specificities were 63.3% and 71.0%. The high specificity rates of template threshold indicate low false-positive rates106, thereby enabling accurate identification of IDD107 and reducing the likelihood of misdiagnosis among participants108. Therefore, individual EOG waveforms in the attentional state serve as a template for individual diagnoses, which can be defined as a description of the observed behavior of visual attention. This approach emphasizes behavioral analysis at the individual level, rather than comparing differences at the population level.
Naturally, it is common for certain diagnostic methods to exhibit specificity but lack sensitivity, resulting in the possibility of missed diagnoses109,110. This shortfall can be mitigated by utilizing D2. Sensitivity, which measures the capacity to accurately identify true positives, becomes crucial in this context111. A high sensitivity value for D2 implies a lower probability of missed diagnoses112, thus minimizing the misclassification of individuals with IDD as TD. The template threshold and D2 exhibit high specificity and sensitivity, respectively, in IDD diagnosis, effectively curtailing both misdiagnoses and missed diagnoses. Consequently, these parameters could serve as potential diagnostic markers for IDD, with thresholds set at 79 (template threshold) and 2.19 (D2).
Performance disparities observed may be due to the distinct meanings conveyed by the data. To pinpoint individual attentional change patterns, we utilized a template matching algorithm based on DTW47. Customizing the template threshold to each individual was crucial for state recognition. Furthermore, ApEn, a measure of information production rate113, denotes the randomness and irregularity within an interactive ensemble114, while D2, a complexity metric, represents the strange attractor’s magnitude in the phase space and characterizes an individual’s overall state115. ApEn and D2, thus, reflect different physiological aspects. While ApEn characterizes dynamic changes during the process of information integration, D2 is more apt for distinguishing between health and disease states. These findings concur with previous EMG116 or EEG117,118 studies.
Limitations
Despite the encouraging nature of the findings presented in this study, it is imperative to acknowledge the limitations that warrant attention. The primary limitation pertains to the absence of a formal and direct measurement of the subjects’ IQ, with information on the degree of IDD derived solely from scales and clinical notes. A second limitation concerns the lack of stratification in the study group, such as age stratification, and the absence of an assessment of diagnoses such as ASD. Furthermore, a third limitation is attributed to the relatively abbreviated duration of the three scenarios, which imposes constraints on the comprehensive interpretation of the results. Nonetheless, in light of these limitations, we contend that the EOG-SA presented in this study hold the potential to function as an early biomarker of IDD.
Conclusions
Our study highlights the efficacy of integrating EOG, Attention-related Visual Evocation System, and nonlinear analysis techniques (EOG-SA) to discern Visual Attention Patterns between individuals with IDD and their TD counterparts. The template threshold based on DTW demonstrated superior discrimination and criterion validity, effectively distinguishing the severity of the condition. Notably, the template threshold and D2 demonstrated high specificity and sensitivity, respectively, for the diagnosis of IDD, thereby mitigating the risk of misdiagnosis and missed diagnosis. These tools also show potential for assessing personal-social skills and gross motor development. Meanwhile, ApEn effectively differentiates diverse application scenarios. This individualized matching technology, which requires no frequent calibration or prior training, is particularly suitable for the IDD population, including young children. It accurately monitors attention over time and identifies pattern changes. This study is the most comprehensive application of EOG in IDD to date, with the EOG-SA presented offering a quantifiable measure to detect subtle changes in visual attention patterns, aiding early diagnosis and disease monitoring.
Data availability
The original contributions presented in the study are included in the Supplementary material, further inquiries can be directed to the corresponding author.
References
Delobel-Ayoub, M. et al. Socioeconomic disparities and prevalence of autism spectrum disorders and intellectual disability. PLoS ONE 10, e141964 (2015).
American Psychiatric Association, Diagnostic and statistical manual of mental disorders: DSM-5™, 5th edn, CODAS vol. 25, 191 (2013).
Vasudevan, P. & Suri, M. A clinical approach to developmental delay and intellectual disability. Clin. Med. 17, 558 (2017).
Peralta, V., Moreno-Izco, L., Jalón, E., Sánchez-Torres, A. M. & Gil-Berrozpe, G. J. Prospective long-term cohort study of subjects with first-episode psychosis examining eight major outcome domains and their predictors: Study protocol. Front. Psychiatry 12, 643112 (2021).
Zhang, Y., Liu, X., Gao, H., He, R. & Zhao, Y. Identifying of 22q11.2 variations in Chinese patients with development delay. BMC Med. Genomics 14, 26 (2021).
Jeoung, B. Motor proficiency differences among students with intellectual disabilities, autism, and developmental disability. J. Exerc. Rehabil. 14, 275 (2018).
van Karnebeek, C. D., Houben, R. F., Lafek, M., Giannasi, W. & Stockler, S. The treatable intellectual disability APP www.treatable-id.org: A digital tool to enhance diagnosis & care for rare diseases. Orphanet. J. Rare Dis. 7, 47 (2012).
Ervin, D. A. & Merrick, J. Intellectual and developmental disability: Healthcare financing. Front. Public Health 2, 160 (2014).
Chen, J., Yu, W., Tsai, M., Hung, P. & Tu, Y. Comorbidities associated with genetic abnormalities in children with intellectual disability. Sci. Rep. 11, 6563 (2021).
Moeschler, J. B., Shevell, M., Committee on Genetics. Comprehensive evaluation of the child with intellectual disability or global developmental delays. Pediatrics 134, e903 (2014).
Boggula, V. R. et al. Clinical utility of multiplex ligation-dependent probe amplification technique in identification of aetiology of unexplained mental retardation: A study in 203 Indian patients. Indian J. Med. Res. 139, 66 (2014).
Rydz, D., Shevell, M. I., Majnemer, A. & Oskoui, M. Developmental screening. J. Child Neurol. 20, 4 (2005).
Vermeirsch, J. et al. Diagnosing autism spectrum disorder in toddlers born very preterm: Estimated prevalence and usefulness of screeners and the autism diagnostic observation schedule (ADOS). J. Autism Dev. Disord. 51, 1508 (2021).
Lin, Y. et al. Diagnosis of Arboleda-Tham syndrome by whole genome sequencing in an Asian boy with severe developmental delay. Mol. Genet. Metab. Rep. 25, 100686 (2020).
Vickers, R. R. & Gibson, J. S. A review of the genomic analysis of children presenting with developmental delay/intellectual disability and associated dysmorphic features. Cureus J. Med. Sci. 11, e3873 (2019).
de Ligt, J. et al. Diagnostic exome sequencing in persons with severe intellectual disability. New Engl. J. Med. 367, 1921 (2012).
Howard, J. et al. Brief report: Classifying rates of students with autism and intellectual disability in north Carolina—Roles of race and economic disadvantage. J. Autism Dev. Disord. 51, 307 (2021).
Miller, L. E., Burke, J. D., Robins, D. L. & Fein, D. A. Diagnosing autism spectrum disorder in children with low mental age. J. Autism Dev. Disord. 49, 1080 (2019).
Kim, S. H. & Lord, C. Combining information from multiple sources for the diagnosis of autism spectrum disorders for toddlers and young preschoolers from 12 to 47 months of age. J. Child Psychol. Psychiatry 53, 143 (2012).
Hernik, M. & Broesch, T. Infant gaze following depends on communicative signals: An eye-tracking study of 5- to 7-month-olds in Vanuatu. Dev. Sci. 22, e12779 (2019).
Pons, F., Bosch, L. & Lewkowicz, D. J. Twelve-month-old infants’ attention to the eyes of a talking face is associated with communication and social skills. Infant Behav. Dev. 54, 80 (2019).
Klusek, J., Moser, C., Schmidt, J., Abbeduto, L. & Roberts, J. E. A novel eye-tracking paradigm for indexing social avoidance-related behavior in fragile X syndrome. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 183, 5 (2019).
Li, R. et al. Aberrant brain network and eye gaze patterns during natural social interaction predict multi-domain social-cognitive behaviors in girls with fragile X syndrome. Mol. Psychiatry 27, 3768–3776 (2022).
Wolf, A. & Ueda, K. Contribution of eye-tracking to study cognitive impairments among clinical populations. Front. Psychol. 12, 590986 (2021).
Glaholt, M. G. & Reingold, E. M. Eye movement monitoring as a process tracing methodology in decision making research. J. Neurosci. Psychol. Econom. 4, 125 (2011).
Nayar, K., Shic, F., Winston, M. & Losh, M. A constellation of eye-tracking measures reveals social attention differences in ASD and the broad autism phenotype. Mol. Autism 13, 18 (2022).
Hall, S. S. et al. Quantifying naturalistic social gaze in fragile X syndrome using a novel eye tracking paradigm. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 168, 564 (2015).
Kasneci, E. et al. Do your eye movements reveal your performance on an IQ test? A study linking eye movements and socio-demographic information to fluid intelligence. PLOS One 17, e264316 (2022).
Benjamin, D. P. et al. Effects of labeling and pointing on object gaze in boys with fragile X syndrome: An eye-tracking study. Res. Dev. Disabil. 35, 2658 (2014).
Frank, M. C., Vul, E. & Saxe, R. Measuring the development of social attention using free-viewing. Infancy 17, 355 (2012).
Wilkinson, K. M., O’Neill Zimmerman, T. & Light, J. Visual attention to cued targets in simulated aided augmentative and alternative communication displays for individuals with intellectual and developmental disabilities. J. Speech Lang. Hear. Res. 64, 1726 (2021).
Yang, Y. et al. Brain responses to biological motion predict treatment outcome in young adults with autism receiving virtual reality social cognition training: Preliminary findings. Behav. Res. Ther. 93, 55 (2017).
Vargason, T., Grivas, G., Hollowood-Jones, K. L. & Hahn, J. Towards a multivariate biomarker-based diagnosis of autism spectrum disorder: Review and discussion of recent advancements. Semin. Pediatr. Neurol. 34, 100803 (2020).
Latrèche, K., Kojovic, N., Franchini, M. & Schaer, M. Attention to face as a predictor of developmental change and treatment outcome in young children with autism spectrum disorder. Biomedicines 9, 942 (2021).
Sumner, E., Hutton, S. B., Kuhn, G. & Hill, E. L. Oculomotor atypicalities in developmental coordination disorder. Dev. Sci. 21, e12501 (2018).
Marandi, R. Z. & Gazerani, P. Aging and eye tracking: In the quest for objective biomarkers. Future Neurol. 14, FNL33 (2019).
Jian, Y. C. Influence of science text reading difficulty and hands-on manipulation on science learning: An eye-tracking study. J. Res. Sci. Teach. 59, 358 (2022).
Bruno, J. L., Garrett, A. S., Quintin, E., Mazaika, P. K. & Reiss, A. L. Aberrant face and gaze habituation in fragile X syndrome. Am. J. Psychiatry 171, 1099 (2014).
Imaoka, Y., Flury, A. & de Bruin, E. D. Assessing saccadic eye movements with head-mounted display virtual reality technology. Front. Psychiatry 11, 572938 (2020).
Perez Reynoso, F. D. et al. A custom EOG-based HMI using neural network modeling to real-time for the trajectory tracking of a manipulator robot. Front. Neurorobot. 14, 578834 (2020).
Merino, M., Gomez, I. M. & Molina, A. J. Envelope filter sequence to delete blinks and overshoots. Biomed. Eng. Online 14, 48 (2015).
Wu, S. L. et al. Controlling a human-computer interface system with a novel classification method that uses electrooculography signals. IEEE T Bio-med. Eng. 60, 2133 (2013).
Valleix, S. et al. H244R VSX1 is associated with selective cone ON bipolar cell dysfunction and macular degeneration in a PPCD family. Invest. Ophthalmol. Vis. 47, 48 (2006).
Chang, W., Cha, H., Kim, D. Y., Kim, S. H. & Im, C. Development of an electrooculogram-based eye-computer interface for communication of individuals with amyotrophic lateral sclerosis. J. Neuroeng. Rehabil. 14, 1–13 (2017).
Heo, J., Yoon, H. & Park, K. A novel wearable forehead EOG measurement system for human computer interfaces. Sensors 17, 1485 (2017).
Pettersson, K. et al. Algorithm for automatic analysis of electro-oculographic data. Biomed. Eng. Online 12, 110 (2013).
Yuan, D. et al. A closed-loop electrical stimulation system triggered by EOG for acupuncture therapy. Syst. Sci. Control Eng. 8, 128 (2020).
Choi, V. & Priebe, N. J. Interocular velocity cues elicit vergence eye movements in mice. J. Neurophysiol. 124, 623 (2020).
Zhang, S. J., Lin, D., Qi, S. Y. & Gong, M. Clinical application of acupuncture-moxibustion for the treatment of spirit based on approximate entropy of electrooculogram signal. Zhongguo Zhen Jiu 43, 79 (2023).
Zhixiang, Z. & Zhenwu, L. Re-standardization of social life ability scale for Chinese infant-junior school student. Chin. J. Clin. Psychol. 3, 12 (1995).
Suzuki, H., Kobayasi, C. & Ikeda, Y. The developmental characteristics of soical maturity on children with down syndrome: Using social maturity scale-reviced (S-M). Bull. Defectol. 21, 139 (1997).
Tatebe, H. et al. Quantification of plasma phosphorylated tau to use as a biomarker for brain Alzheimer pathology: Pilot case-control studies including patients with Alzheimer’s disease and down syndrome. Mol. Neurodegener. 12, 1–11 (2017).
Amatruda, C. S. & Gesell, A., Developmental diagnosis;normal and abnormal child development, clinical methods and pediatric applications, 2d ed., rev. and enl. ed. (Hoeber, 1947).
Wu, L. et al. Physical, language, neurodevelopment and phenotype-genotype correlation of Chinese patients with Mowat-Wilson syndrome. Front. Genet. 13, 1016677 (2022).
Acharya, D., Rani, A. & Agarwal, S., International Conference on Reliability, 2015 (unpublished).
Champaty, B., Jose, J., Pal, K. & Thirugnanam, A., in 2014 Annual International Conference on Emerging Research Areas: Magnetics, Machines and Drives (AICERA/iCMMD) 2014 Annual International Conference on Emerging Research Areas: Magnetics, Machines and Drives (AICERA/iCMMD) 1 (2014).
Tonin, A. et al. Auditory electrooculogram-based communication system for ALS patients in transition from locked-in to complete locked-in state. Sci. Rep. 10, 8452 (2020).
Koepcke, L., Hildebrandt, K. J. & Kretzberg, J. Online detection of multiple stimulus changes based on single neuron interspike intervals. Front. Comput. Neurosci. 13, 69 (2019).
Jafarpour, A., Piai, V., Lin, J. J. & Knight, R. T. Human hippocampal pre-activation predicts behavior. Sci. Rep. 7, 5959 (2017).
Jing, J. et al. Rapid annotation of interictal epileptiform discharges via template matching under dynamic time warping. J. Neurosci. Methods 274, 179 (2016).
Keogh, E. & Ratanamahatana, C. A. Exact indexing of dynamic time warping. Knowl. Inf. Syst. 7, 358 (2005).
Van Strien, J. et al. COmplexome Profiling ALignment (COPAL) reveals remodeling of mitochondrial protein complexes in Barth syndrome. Bioinformatics 35, 3083 (2019).
Chiang, P., Chien, T., Chen, C., Yanagawa, Y. & Lien, C. ASIC-dependent LTP at multiple glutamatergic synapses in amygdala network is required for fear memory. Sci. Rep. 5, 10143 (2015).
Pincus, S. M. Approximate entropy as a measure of system complexity. Proc. Natl. Acad. Sci. USA 88, 2297 (1991).
Rabbi, A. F. & Fazel-Rezai, R. A fuzzy logic system for seizure onset detection in intracranial EEG. Comput. Intell. Neurosci. 2012, 1 (2012).
Sartori, M. et al. Association between diastolic dysfunction with inflammation and oxidative stress in females ob/ob mice. Front. Physiol. 8, 572 (2017).
Talebi, N., Nasrabadi, A. M. & Curran, T. Investigation of changes in EEG complexity during memory retrieval: The effect of midazolam. Cogn. Neurodyn. 6, 537–546 (2012).
Martínez-Rodrigo, A., García-Martínez, B., Zunino, L., Alcaraz, R. & Fernández-Caballero, A. Multi-lag analysis of symbolic entropies on EEG recordings for distress recognition. Front. Neuroinform. 13, 40 (2019).
Kilicarslan, A. & Contreras-Vidal, J. L. Characterization and real-time removal of motion artifacts from EEG signals. J. Neural Eng. 16, 056027 (2019).
Melillo, P., Bracale, M. & Pecchia, L. Nonlinear Heart Rate Variability features for real-life stress detection. Case study: Students under stress due to university examination. Biomed. Eng. Online 10, 96 (2011).
Grassberger, P. Measuring the strangeness of strange attracters. Physica 9, 189–208 (1983).
Ji, C., Yao, D., Chen, W., Li, M. & Zhao, Z. Adaptive behavior in Chinese children with Williams syndrome. BMC Pediatry 14, 90 (2014).
Li, B. et al. The characteristics of social maturity in infants and children with cochlear implants in China. Int. J. Pediatr. Otorhinolaryngol. 131, 109887 (2020).
Wang, Y. et al. Metachromatic leukodystrophy: Characterization of two (p.Leu433Val, p.Gly449Arg) arylsulfatase A mutations. Exp. Ther. Med. 18, 1738 (2019).
Dindar, K. et al. Social-pragmatic inferencing, visual social attention and physiological reactivity to complex social scenes in autistic young adults. J. Autism Dev. Disord. 52, 73 (2022).
van der Fels, I. M. et al. The relationship between motor skills and cognitive skills in 4–16 year old typically developing children: A systematic review. J. Sci. Med. Sport 18, 697 (2015).
Houwen, S., Visser, L., Putten, A. V. D. & Vlaskamp, C. The interrelationships between motor, cognitive, and language development in children with and without intellectual and developmental disabilities. Res. Dev. Disabil. 53–54, 19 (2016).
Oliveira, L., Julio-Costa, A., Dos, S. F., Carvalho, M. & Haase, V. G. Numerical processing impairment in 22q11.2 (LCR22-4 to LCR22-5) microdeletion: A cognitive-neuropsychological case study. Front. Psychol. 9, 2193 (2018).
Wuang, Y. P., Huang, C. L. & Tsai, H. Y. Sensory integration and perceptual-motor profiles in school-aged children with autistic spectrum disorder. Neuropsychiatr. Dis. Treat. 16, 1661 (2020).
Valerie, et al. Behavioral abnormalities are common and severe in patients with distal 22q11.2 microdeletions and microduplications. Mol. Genet. Genom. Med. 3, 346 (2015).
Ben-Shachar, S. et al. 22q11.2 distal deletion: A recurrent genomic disorder distinct from DiGeorge syndrome and velocardiofacial syndrome. Am. J. Hum. Genet. 82, 214 (2008).
Tian, Y. et al. Influencing factors of the neurodevelopment of high-risk infants. Gen. Psychiatry 31, e100034 (2018).
Sweere, D. J. J. et al. Clinical utility of eye tracking in assessing distractibility in children with neurological disorders or ADHD: A cross-sectional study. Brain Sci. 12, 1369 (2022).
Luna, B., Velanova, K. & Geier, C. F. Development of eye-movement control. Brain Cognit. 68, 293 (2008).
Bradshaw, J. et al. The use of eye tracking as a biomarker of treatment outcome in a pilot randomized clinical trial for young children with autism. Autism Res. 12, 779 (2019).
McPartland, J. C. Considerations in biomarker development for neurodevelopmental disorders. Curr. Opin. Neurol. 29, 118 (2016).
Braff, D. L. Information processing and attention dysfunctions in schizophrenia. Schizophr. Bull. 19, 233 (1993).
Rommelse, N. N., Van der Stigchel, S. & Sergeant, J. A. A review on eye movement studies in childhood and adolescent psychiatry. Brain Cognit. 68, 391 (2008).
Borji, A. & Itti, L. Defending Yarbus: Eye movements reveal observers’ task. J. Vis. 14, 29 (2014).
Vargas-Cuentas, N. I. et al. Developing an eye-tracking algorithm as a potential tool for early diagnosis of autism spectrum disorder in children. PLOS One 12, e188826 (2017).
Eckstein, M. K., Guerra-Carrillo, B., Miller Singley, A. T. & Bunge, S. A. Beyond eye gaze: What else can eyetracking reveal about cognition and cognitive development?. Dev. Cogn. Neurosci. 25, 69 (2017).
Krasotkina, A., Götz, A., Höhle, B. & Schwarzer, G. Infants’ gaze patterns for same-race and other-race faces, and the other-race effect. Brain Sci. 10, 331 (2020).
Geeraerts, S. B. et al. Individual differences in visual attention and self-regulation: A multimethod longitudinal study from infancy to toddlerhood. J. Exp. Child Psychol. 180, 104 (2019).
Riddiford, J. A., Enticott, P. G., Lavale, A. & Gurvich, C. Gaze and social functioning associations in autism spectrum disorder: A systematic review andmeta-analysis. Autism Res. 15, 1380 (2022).
Reani, M., Peek, N. & Jay, C. How different visualizations affect human reasoning about uncertainty: An analysis of visual behaviour. Comput. Hum. Behav. 92, 55 (2019).
Tian, L. H. et al. Mapping the relationship between dysmorphology and cognitive, behavioral, and developmental outcomes in children with autism spectrum disorder. Autism Res. 13, 1227 (2020).
Nag, A. et al. Toward continuous social phenotyping: Analyzing gaze patterns in an emotion recognition task for children with autism through wearable smart glasses. J. Med. Internet Res. 22, e13810 (2020).
Gordon, U., Marom, S. & Brenner, N. Visual detection of time-varying signals: Opposing biases and their timescales. PLOS One 14, e224256 (2019).
Rice, K., Moriuchi, J. M., Jones, W. & Klin, A. Parsing heterogeneity in autism spectrum disorders: Visual scanning of dynamic social scenes in school-aged children. J. Am. Acad. Child Adolesc. Psychiatry 51, 238 (2012).
Pierce, K. et al. Eye tracking reveals abnormal visual preference for geometric images as an early biomarker of an autism spectrum disorder subtype associated with increased symptom severity. Biol. Psychiatry 79, 657 (2016).
Oliveira, J. S. et al. Computer-aided autism diagnosis based on visual attention models using eye tracking. Sci. Rep. 11, 10131 (2021).
Barea, R., Boquete, L., Rodriguez-Ascariz, J. M., Ortega, S. & Lopez, E. Sensory system for implementing a human-computer interface based on electrooculography. Sensors 11, 310 (2011).
Wolfe, B. A., Kosovicheva, A. A., Leib, A. Y., Wood, K. & Whitney, D. Foveal input is not required for perception of crowd facial expression. J. Vis. 15, 11 (2015).
Hu, D., Cheng, T., Xie, K. & Lam, R. Microengineered conductive elastomeric electrodes for long-term electrophysiological measurements with consistent impedance under stretch. Sensors 15, 26906 (2015).
López, A., Ferrero, F., Villar, J. R. & Postolache, O. High-performance analog front-end (AFE) for EOG systems. Electronics 9, 970 (2020).
Guo, J., Hong, Y., Wang, Z. & Li, Y. Prognostic value of thyroid hormone FT3 in general patients admitted to the intensive care unit. Biomed. Res. Int. 2020, 1 (2020).
Aoe, J. et al. Automatic diagnosis of neurological diseases using MEG signals with a deep neural network. Sci. Rep. 9, 5057 (2019).
Cicalese, P. A. et al. An EEG-fNIRS hybridization technique in the four-class classification of Alzheimer’s disease. J. Neurosci. Methods 336, 108618 (2020).
Maley, J. H., Worsham, C. M., Landon, B. E. & Stevens, J. P. Association between palliative care and end-of-life resource use for older adults hospitalized with septic shock. Ann. Am. Thorac. Soc. 17, 974 (2020).
Liang, F., Wang, S., Zhang, K., Liu, T. & Li, J. Development of artificial intelligence technology in diagnosis, treatment, and prognosis of colorectal cancer. World J. Gastro Oncol. 14, 124 (2022).
Janaki, C., Gowri, V. S. & Srinivasan, N. Master blaster: An approach to sensitive identification of remotely related proteins. Sci. Rep. 11, 8746 (2021).
Fu, L. et al. Hybrid network with attention mechanism for detection and location of myocardial infarction based on 12-lead electrocardiogram signals. Sensors 20, 1020 (2020).
Shourie, N., Firoozabadi, M. & Badie, K. Analysis of EEG signals related to artists and nonartists during visual perception, mental imagery, and rest using approximate entropy. Biomed. Res. Int. 2014, 1 (2014).
Veldhuis, J. D., Roelfsema, F., Keenan, D. M. & Pincus, S. Gender, age, body mass index, and IGF-I individually and jointly determine distinct GH dynamics: Analyses in one hundred healthy adults. J. Clin. Endocrinol. Metab. 96, 115 (2011).
Karakatsanis, L. P. et al. Spatial constrains and information content of sub-genomic regions of the human genome. Iscience 24, 102048 (2021).
Chen, W., Wang, Z. & Ren, X. Characterization of surface EMG signals using improved approximate entropy. J. Zhejiang Univ. B. Sci. 7, 844 (2006).
Jiang, Z., Feng, H., Liu, D. & Wang, T. Analyzing sleep EEG using correlation dimension and approximate entropy. Sheng Wu Yi Xue Gong Cheng Xue Za Zhi 22, 649 (2005).
Qi, S. et al. Using nonlinear dynamics and multivariate statistics to analyze EEG signals of insomniacs with the intervention of superficial acupuncture. Evid.-BASED Complement. Altern. 2020, 1 (2020).
Acknowledgements
This research received financial support from the National Natural Science Foundation of China (Grant No. 82074521), the 2024 Fujian University of Traditional Chinese Medicine Acupuncture and Moxibustion Discipline Open Project, and the Fujian Provincial Key Discipline Construction Project of Traditional Chinese Medicine—“Acupuncture and Moxibustion Engineering”. We extend our heartfelt thanks to the editorial team for their meticulous and diligent efforts in the peer-review process. Your valuable input has been instrumental in refining our manuscript.
Author information
Authors and Affiliations
Contributions
D.L., S.Y.Q., S.J.Z., and L.L.L. had full access to the data and ensured the integrity and accuracy of the analysis. D.L. and L.L.L. conceptualized and designed the study. S.Y.Q., S.J.Z., J.Y.W., S.L., M.G., and L.L.H. were responsible for data collection. Data interpretation was conducted by Y.R.L., J.G.C., Y.C.N., and X.D. The statistical analysis was carried out by S.Y.Q. and S.J.Z. S.Y.Q. drafted the initial manuscript. Critical revisions for intellectual content were made by D.L., S.J.Z., and L.L.L. D.L. and L.L.L. secured the funding. Administrative and technical support was provided by J.Z., P.G., G.H.L., and L.F.C. D.L. led the overall study supervision. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Qi, Sy., Zhang, Sj., Lin, Ll. et al. Quantifying attention in children with intellectual and developmental disabilities through multicenter electrooculogram signal analysis. Sci Rep 14, 22186 (2024). https://doi.org/10.1038/s41598-024-70304-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-70304-x
- Springer Nature Limited