Abstract
The actual prevalence of diabetic kidney disease (DKD) in patients with type 2 diabetes (T2D) in Malaysia is unknown. We aimed to determine the prevalence of DKD and its associated risk factors among T2D patients in Malaysia. An analytical cross-sectional study was conducted using the year 2022 clinical audit dataset from the National Diabetes Registry. DKD was defined as albuminuria, a decreased glomerular filtration rate, or both. Among 80,360 patients, 62.2% were female, 68.4% were Malay, and the mean age was 61.4 years. A total of 56.7% (95% CI 56.4–57.1%) of patients were found to have DKD. Increasing age, male sex, Malay ethnicity, longer duration of diabetes, overweight, obesity, hypertension, diabetic retinopathy, diabetic foot ulcer, nontraumatic lower-extremity amputation, ischaemic heart disease, stroke, insulin, higher numbers of antihypertensive agents, antiplatelet agents, poorer HbA1c control, higher systolic blood pressure, non-achievement of triglyceride target, and non-attainment of HDL-cholesterol goal were independent risk factors associated with DKD. Clinicians, program managers, and health policymakers should target modifiable factors to manage DKD and prevent its progression to end-stage kidney disease in Malaysia.
Similar content being viewed by others
Introduction
Chronic kidney disease (CKD) imposes enormous disease burdens in terms of financial costs and adverse health outcomes, such as frailty, end-stage kidney disease (ESKD), decreased quality of life, and premature mortality1. CKD is one of the most important causes of morbidity and mortality globally2. This progressive condition is estimated to affect over 10% or more than 800 million of the general population worldwide2.
Diabetes mellitus is the main cause of CKD and ESKD in developed and developing countries3. CKD among patients with diabetes is known as diabetic kidney disease (DKD)4, and DKD is responsible for the majority of the excess cardiovascular and all-cause mortality in them5. Although kidney biopsy is the gold standard to diagnose DKD, it is not routinely done in real-world clinical settings6. DKD manifests clinically as albuminuria, decreased glomerular filtration rate (GFR), or both4. Estimates of DKD prevalence vary widely between nations, from 27.1% in China to 83.7% in Tanzania5,7,8.
Malaysia, a multi-ethnic upper-middle-income country in Southeast Asia, has the highest prevalence of diabetes in the region9. DKD was consistently responsible for more than 50% of ESKD among new patients requiring dialysis in Malaysia between 2011 and 202110. The prevalence of DKD among type 2 diabetes (T2D) patients in the country was reported to be only 14.6% in 201911. As the reported prevalence was relatively lower than that reported elsewhere across the globe1,5, underreporting of the condition and under-screening for DKD were postulated as potential explanations11,12. For perspective, the prevalence of CKD among the general adult population in Malaysia was 15.5% in 201813.
Hence, a more realistic estimate of DKD prevalence in T2D patients in Malaysia is required. Moreover, in the recent World Health Organization (WHO) guidelines on the monitoring of noncommunicable diseases at health facilities, the proportion of patients with diabetes who are diagnosed with DKD is one of the recommended indicators14. Thus, we aimed to determine the prevalence of DKD and its associated factors among T2D patients in Malaysia.
Methods
Study design
An analytical cross-sectional study was conducted using the year 2022 clinical audit dataset from the National Diabetes Registry. This secondary dataset was routinely collected every year since the registry’s inception in 2009 as a surveillance platform to track the quality of diabetes care in public health clinics, aid policymaking, and used for research purposes11. The clinical audit, also known as the Quality Assurance Study, is under the National Quality Assurance Programme to ensure that the care given by the Ministry of Health Malaysia follows regional and international standards11,15.
Study population
The target population included patients with T2D who received care from public health clinics in Malaysia. Around 70% of patients with diabetes in the country were treated in public health clinics16. The inclusion criteria were adult patients with T2D aged 18 years and above. Patients without documented serum creatinine or urine albumin levels were excluded. Figure 1 shows the flow diagram detailing the number of individuals at each stage of the dataset management, together with inclusion and exclusion criteria. The final number of eligible patients for analysis was 80,360. We compared the proportions of selected and non-selected patients by demographic factors in Supplementary Table S1. A higher proportion of those selected in the study were in the younger age category, females, and of Malay ethnicity.
Dependent variable
DKD was defined as having albuminuria, a decreased GFR, or both4. We defined albuminuria as having macroalbuminuria and/or microalbuminuria. Macroalbuminuria and microalbuminuria were captured as categorical variables with positive or negative results, respectively, in the audit dataset. The test for microalbuminuria was routinely done in T2D patients having negative results for urine protein. Common tests used to detect albuminuria in health clinics included automated urine analysis, dipstick, 24-h urine protein, and urine albumin-to-creatinine ratio17.
A decreased GFR was defined as having a GFR < 60 ml/min/1.73 m2, according to the 2020 Kidney Disease Improving Global Outcomes (KDIGO) guidelines18. The GFR categories were G1, G2, G3a, G3b, G4, and G5 with GFR ≥ 90, 60–89, 45–59, 30–44, 15–29, and < 15 ml/min/1.73 m2, respectively18. Hence, GFR < 60 ml/min/1.73 m2 corresponded to G3a to G5. We determined the estimated GFR (eGFR) using the 2009 CKD-Epidemiology Collaboration (CKD-EPI) creatinine equation19, which was the preferred method for the Malaysian population20. A local study reported that the CKD-EPI creatinine equation was more accurate and precise than the Modification of Diet in Renal Disease (MDRD) equation21 The CKD-EPI equation was eGFR = 141 × min (Scr/κ, 1)α × max (Scr/κ, 1)−1.209 × 0.993Age × 1.018 (if female); Scr is serum creatinine, κ is 0.9 for males and 0.7 for females, α is − 0.411 for males and − 0.329 for females, min indicates the minimum of Scr/κ or 1, and max indicates the maximum of Scr/κ or 119.
Independent variables
The independent variables included demographic factors, comorbidities, diabetes-related complications, pharmacological treatments, and metabolic control. The demographic characteristics included age, sex, and ethnic groups. Besides the three major ethnicities (Malay, Chinese, and Indian) in Malaysia, there are indigenous people of Bumiputera Sabah and Bumiputera Sarawak who mainly reside in East Malaysia. ‘Other ethnic groups’ comprised minority ethnicities such as indigenous people of Peninsular Malaysia and those of foreign nationals. Smoking status was defined as the current smoking status.
Overweight and obesity categories were classified as having body mass index (BMI) of 23.0–27.4 and ≥ 27.5 kg/m2, respectively22. These lower cut-offs followed the World Health Organization (WHO) recommendations for Asian populations due to their greater risks of adverse cardiovascular outcomes23. Meanwhile, hypertension and dyslipidaemia followed clinical diagnoses or the use of corresponding pharmacological agents. Diabetes-related complications, such as diabetic retinopathy, diabetic foot ulcer, nontraumatic lower-extremity amputation, ischemic heart disease, and stroke, were based on clinical diagnoses by treating doctors.
Diabetes treatment modality was categorised as ‘lifestyle management only’, ‘oral glucose-lowering drug (OGLD) only’, ‘insulin only’, and ‘OGLD and insulin’. Hypertension treatment was categorised as zero, one, two, and ≥ three blood pressure-lowering drugs. The usage of lipid-lowering and antiplatelet agents was also described.
The metabolic control covered glycosylated haemoglobin A1c (HbA1c), blood pressure (BP), and LDL-cholesterol, the three primary treatment targets in diabetes management20. Most patients with T2D had a HbA1c target < 7.0%, while others, such as elderly patients and those with comorbidities, including DKD, were recommended to achieve a HbA1c goal between 7.0 and 8.0%20. Patients with HbA1c > 8.0% were considered to have poor glycaemic control20. The BP were classified according to stage I (140–159/90–99 mmHg), stage II (160–179/100–109 mmHg), and severe hypertension (≥ 180/ ≥ 110 mmHg)24. An additional systolic BP cut-off of 130 mmHg was used because BP < 130/80 mmHg was the individualised treatment target for patients with DKD or cardiovascular diseases20. Otherwise, a BP < 140/80 mmHg was aimed at those without DKD or cardiovascular diseases20. The LDL-cholesterol < 2.6 mmol/L was the general target, while an intensified LDL-cholesterol < 1.4 mmol/L was recommended for those with target organ damage, including DKD20.
The secondary treatment targets, namely triglyceride and HDL-cholesterol, were also reported20. The triglyceride target was < 1.7 mmol/L, and HDL-cholesterol targets were > 1.0 for males and > 1.3 mmol/L for females20.
Statistical analysis
The analysis was carried out using the IBM SPSS Statistics version 23. Descriptive data were presented as frequencies with percentages for categorical variables. Whereas, mean ± standard deviation or median (interquartile range) were reported for continuous variables. The proportion of patients with DKD was presented as frequency and percentage with a 95% confidence interval. We first performed bivariate analyses. Differences between DKD status were assessed using Pearson chi-square tests for categorical variables, Student’s t-tests for means, and Mann–Whitney tests for medians. Then, multivariate binary logistic regression was carried out for independent variables with P values < 0.25 and clinically essential variables to determine factors associated with DKD. A forward stepwise likelihood ratio was used. The classification table, coefficient of determination, Omnibus test of model coefficients, Hosmer–Lemeshow test, and area under receiving operating characteristics (ROC) curve were reported. We also assessed multicollinearity and the interaction between variables. P values, adjusted odd ratios, and 95% confidence intervals were presented. The statistical significance threshold was pre-set at P < 0.05. All missing data were list-wise deleted in the multiple logistic regression analysis as we intend to analyse the real-world clinical data as it is. Demographic differences between patients selected in the study and those included in the multiple logistic regression analyses were compared using Chi-square tests.
Ethical approval
This research was approved by the Medical Review and Ethics Committee of the Ministry of Health Malaysia (NMRR ID-23-01030-S6L). The Medical Research and Ethics Committee waived the requirement for informed consent because this study used secondary data without personal identifiers. All methods followed the Declaration of Helsinki and the Malaysian Good Clinical Practice Guidelines.
Results
Characteristics of patients
Among the 80,360 T2D patients, around 59.7% were older adults aged 60 years or older (Table 1). There were more females (62.2%), more Malay patients (68.4%), and more non-current smokers (94.0%). About 37.4% of the patients were diagnosed with T2D for five to ten years. The proportion of patients with overweight/obesity, hypertension, and dyslipidaemia was 84.5%, 89.4%, and 92.4%, respectively. Diabetic retinopathy (10.4%) was the most common complication, followed by ischemic heart disease (5.1%), stroke (1.8%), diabetic foot ulcers (0.9%), and nontraumatic lower-extremity amputation (0.5%). Oral glucose-lowering drugs (OGLD) were used in 91.7% of the patients, and 30.0% were on insulin therapy. About 86.7% and 87.9% of T2D patients were given antihypertensive and lipid-lowering agents, respectively. Meanwhile, 18.6% of patients were given antiplatelet agents.
The mean HbA1c was 7.8 ± 2.1%, and 35.7% of patients had poor glycosylated haemoglobin A1c (HbA1c) > 8.0%. The mean blood pressure (BP) was 136/77 mmHg, and 59.2% and 75.1% of the patients had BP ≥ 140/80 mmHg and ≥ 130/80 mmHg, respectively. The respective mean LDL-cholesterol, triglycerides, and HDL-cholesterol levels were 2.8, 1.7, and 1.3 mmol/L. The respective achievement of triglyceride and HDL-cholesterol targets were 62.8% and 58.7%. About 45.8% and 5.5% of patients attained LDL-cholesterol goals < 2.6 and < 1.4 mmol/L, respectively.
Prevalence of diabetic kidney disease
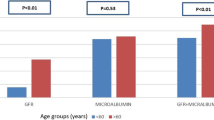
A total of 56.7% of patients were found to have DKD (Fig. 2). The Venn diagram shows that 22.4% of patients had eGFR < 60 mL/min/1.73 m2, while 48.1% had positive albuminuria. Around 13.8% of patients had both eGFR < 60 mL/min/1.73 m2 and positive albuminuria. About 8.6% of the patients had non-proteinuric kidney disease, while 34.3% had albuminuria with a normal eGFR. Regarding DKD stages, 12.4%, 6.9%, 2.4%, and 0.7% of patients were at stages G3a, G3b, G4, and G5, respectively.
Characteristics of patients with diabetic kidney diseases
Patients with DKD were significantly older, male, of Malay ethnicity, current smokers, and had been diagnosed with diabetes for a longer duration (Supplementary Table S2). Patients with DKD generally had a higher mean body mass index (BMI), and a higher proportion of them were underweight and obese. DKD was associated with all the comorbidities and complications studied. More patients had hypertension, dyslipidaemia, diabetic retinopathy, diabetic foot ulcers, nontraumatic lower-extremity amputation, ischemic heart disease, and stroke. Correspondingly, a higher proportion of patients with DKD were treated with insulin, antihypertensive, lipid-lowering agents, and antiplatelet agents.
Patients with DKD had higher mean HbA1c levels. Despite having a less stringent HbA1c target, 40.5% had uncontrolled HbA1c levels > 8.0%. Meanwhile, a higher proportion of patients with DKD had high systolic and diastolic BP. Thus, lower proportions achieved BP < 140/80 mmHg and < 130/80 mmHg. Among those with DKD, only 10,807 (23.9%) patients achieved a more stringent treatment goal of < 130/80 mmHg.
A higher proportion of patients with DKD had LDL-cholesterol < 2.6 and < 1.4 mmol/L. Among the patients with CKD, only 6.0% achieved a more stringent LDL-cholesterol target of < 1.4 mmol/L. Patients with DKD had a higher mean triglyceride level, with a correspondingly lower proportion attaining the target triglyceride level. Meanwhile, patients with DKD had a lower mean HDL-cholesterol level with a lower proportion achieving the HDL-cholesterol goal.
Factors associated with diabetic kidney disease
Eighteen independent factors were associated with DKD, as shown in Table 2. Increasing age, longer duration since diabetes diagnosis, overweight, obesity, hypertension, diabetic retinopathy, diabetic foot ulcer, nontraumatic lower-extremity amputation, ischaemic heart disease, stroke, insulin (alone and in combination with oral glucose-lowering drugs), higher numbers of antihypertensive agents, antiplatelet agents, poorer HbA1c control, and higher systolic BP categories were associated with higher odds ratios for DKD. In contrast, females and patients attaining triglyceride and HDL targets were less likely to have DKD. There were ethnic variations; Chinese, Indian, Bumiputera Sarawak, and other ethnicities had lower adjusted odds ratios than the Malay ethnic group.
Supplementary Table S3 compares the demographic characteristics of included and non-included patients in the multiple logistic regression analysis. A higher proportion of those included were in the younger age category, females, and of Malay ethnicity.
Discussion
More than half of our patients with T2D had DKD, and this prevalence falls between the 27.1% to 83.7% range reported worldwide5. This wide range of prevalence could be due to differences in DKD definition, patient profiles, healthcare settings, and health systems5,25. Our result closely approximates the global prevalence (56%) reported in the DEMAND (Developing Education on Microalbuminuria for Awareness of renal and cardiovascular risk in Diabetes) study1,26. Moreover, the composition of DKD by reduced eGFR and positive albuminuria among our patients is also similar to the global average; most DKD diagnoses are due to albuminuria with normal eGFR, followed by reduced eGFR and positive albuminuria, and finally, non-proteinuric kidney disease1,26. Indeed, a review article reported that non-proteinuric rather than proteinuric kidney diseases are the leading cause of ESKD, and non-proteinuric DKD prevails over the proteinuric form among T2D diabetes27.
Our prevalence of DKD is also comparable with 53% reported in a Singaporean study among a multi-ethnic group of primary care patients with T2D25. Again, the composition of DKD is similar to ours: 21% of their patients had reduced eGFR, while 48% had albuminuria25. The breakdown by GFR categories from stage G3a to G5 was also comparable with our study25. Besides that, a similar prevalence of reduced GFR and prevalence by GFR stages were reported in northern Thailand among T2D patients in the primary care setting28.
Overall, DKD is a common complication in patients with T2D in Malaysia. These results have important clinical and public health implications. Clinically, it implies the need to manage patients more aggressively to prevent the progression to ESKD, especially since diabetes was already the main contributor to dialysis in Malaysia10,20. DKD screening activities must also be intensified urgently, particularly because many patients in early DKD are asymptomatic and called the ‘silent majority’6. The clinical practice guidelines recommend screening for DKD during the initial visit and annually thereafter20. The lack of adherence to these recommendations constitutes clinical inertia that must be managed appropriately29. The WHO has recently recommended monitoring the proportion of diabetes patients with DKD in healthcare facilities, which should be considered in our health clinic settings to ensure optimal patient and programme monitoring14.
From a public health standpoint, the high DKD burden should alert health policymakers and programme managers about the substantial financial costs and adverse health outcomes associated with the disease, such as frailty, ESKD, reduced quality of life, and premature deaths1. The problem will only be magnified if more Malaysians develop diabetes, and if diabetes control remains unattainable among existing patients, more of them will end up having ESKD20. Around 106,000 dialysis patients are projected in Malaysia by the year 204030, and the relative high share of ESKD health expenditure in the public sector will stress the financing mechanism of the disease31. Therefore, primary, secondary, and tertiary prevention of diabetes must be optimised to tackle the diabetes epidemic in Malaysia. This effort is consistent with the National Action Plan for Healthy Kidneys 2018–2025, a strategic plan to decrease CKD burdens in the country32.
Established risk factors for DKD can be divided into non-modifiable and modifiable risk factors1,5. Our study findings are consistent with increasing age, male sex, ethnicity, and long duration of diabetes as non-modifiable factors associated with DKD1,5. Previous studies reported ethnic differences with Asian, Hispanic, and indigenous Australians tend to have a higher prevalence of DKD than Caucasians1. Further, Asian participants were found to have the highest proteinuria compared to Hispanic, African, Caucasian, and other ethnic groups26. The reasons for ethnic variations in DKD are complex and multifactorial1. The factors include genetic factors and developmental programming, age of T2D onset, lifestyle factors, socioeconomic disadvantages, access to and uptake of care, inadequate screening rates, and poorer attainment of treatment targets1.
Our multi-ethnic populations in Malaysia confer an advantage in observing ethnic variations in the prevalence of DKD. However, it is unclear why the Malay ethnicity is more likely to be associated with DKD. Further research is recommended to investigate the underlying reasons for the observed ethnic differences in Malaysia.
Obesity, hypertension, poor glycaemic control, poor blood pressure control, and lipid abnormalities are known modifiable risk factors for DKD, and our study again showed consistent findings1,5. These results further emphasise the crucial need to optimise body weight and control metabolic targets among T2D patients. However, most of our patients with DKD did not achieve HbA1c, intensified BP, or intensified LDL-cholesterol goals. This is alarming because inadequate metabolic control can lead to the progression of DKD to ESKD1. Moreover, poor control of these risk factors will increase the competing risk of premature mortality, mainly due to cardiovascular diseases1.
We also found that diabetic retinopathy, diabetic foot ulcer, nontraumatic lower-extremity amputation, ischaemic heart disease, stroke, insulin, antiplatelet agents, and higher numbers of antihypertensive agents were associated with DKD. All these factors are proxies for more severe diabetes conditions and are clinically logical to be related to DKD. Similar associations have been reported in other epidemiological studies among patients with T2D in Singapore, Thailand, Hong Kong, and Italy25,28,33,34. Interestingly, we found that underweight patients were independently associated with DKD; the adjusted odds ratio was even higher than that for obesity. A Korean nationwide cohort study reported that the underweight BMI category was an independent risk factor for ESKD among patients with diabetes35. Moreover, those with weight loss > 10% had the fastest decline in kidney function35. Some plausible mechanisms include sarcopenia and oxidative DNA damage associated with weight loss35.
We acknowledge missing data as a limitation in this registry-based study. We noted that a higher proportion of patients selected for this study and those included in the regression analysis were females, in the younger age category, and of Malay ethnicity, and this could cause selection bias and distort the actual prevalence of DKD in this study. As increasing age and male sex are established non-modifiable risk factors for DKD1,5, our study sample could have pulled the DKD prevalence downwards. In other words, the actual prevalence of DKD among T2D patients in Malaysia could be higher if more males and older patients were included. In contrast, since we found that the Malay ethnicity was more likely to have DKD, a higher proportion of Malay patients could have pushed the prevalence upwards. Nevertheless, our current prevalence estimate is still valid based on available real-world clinical data, with the prevalence falling between the range reported worldwide and similar to our neighbouring countries in Singapore and Thailand5,25,28.
This study has other limitations. The cross-sectional design disallows any causal inference between DKD and the associated factors. Measurement errors can occur because of the lack of standardisation mechanisms between health clinics across Malaysia. Nevertheless, the data reflect real-world clinical scenarios. Besides that, the analysis was limited to the data available in the National Diabetes Registry. Uncaptured information, such as socioeconomic disadvantages, family history of DKD, previous episodes of acute kidney injury, and inflammatory markers, all associated with DKD, could not be investigated in this study5. We were also unable to quantify the level of albuminuria as the details were not captured in the database. Our study definition of DKD was based on a single positive reading of albuminuria, a decreased GFR, or both with no repeated measurement typically taken 3 months apart to confirm the chronic nature of CKD18. Nevertheless, our results are still valid with similar definitions employed in other cross-sectional studies with no repeated measures to determine CKD or DKD prevalence25,28,36,37. Finally, the study population was confined to T2D patients in public health clinics; hence, the results cannot be generalised to patients treated in hospitals and those with type 1 diabetes.
To our best knowledge, this nationwide study is among the first to report the prevalence of DKD and its associated factors among patients with T2D in Malaysia. Real-world clinical information offers the added advantage of depicting the actual situations in the field. Our results have established a baseline prevalence of DKD among T2D patients in Malaysia, which may aid in monitoring the WHO indicator for DKD14. This study uncovers a high prevalence of DKD with important clinical and public health implications, as discussed above. We hope the results will help inform policymaking and the development of clinical practice guidelines in the country. This study also benefitted from the multi-ethnic population in Malaysia. The observed ethnic differences in the prevalence of DKD may provide an impetus for new studies on kidney complications among multi-ethnic diabetes patients to improve clinical outcomes.
In summary, DKD is highly prevalent among T2D patients in Malaysia. Increasing age, male sex, Malay ethnic group, longer duration of diabetes, overweight, obesity, hypertension, diabetic retinopathy, diabetic foot ulcer, nontraumatic lower-extremity amputation, ischaemic heart disease, stroke, insulin, higher numbers of antihypertensive agents, antiplatelet agents, poorer HbA1c control, higher systolic BP, non-achievement of triglyceride target, and non-attainment of HDL-cholesterol goal are independent factors associated with DKD. Clinicians, program managers, and health policymakers should target these modifiable factors to manage DKD and prevent its progression to ESKD.
Data availability
The National Diabetes Registry dataset retrieved and analysed in this study is not available publicly due to local ethics regulation and could be obtained via written permissions to the Director General of Health, Malaysia.
References
Thomas, M. C. et al. Diabetic kidney disease. Nat. Rev. Dis. Primers. 1(1), 15018 (2015).
Kovesdy, C. P. Epidemiology of chronic kidney disease: An update 2022. Kidney Int. Suppl. 12(1), 7–11 (2022).
Ghaderian, S. B., Hayati, F., Shayanpour, S. & Beladi Mousavi, S. S. Diabetes and end-stage renal disease; a review article on new concepts. J. Renal Inj. Prev. 4(2), 28–33 (2015).
Afkarian, M. et al. Clinical manifestations of kidney disease among US adults with diabetes, 1988–2014. JAMA. 316(6), 602–610 (2016).
Koye, D. N., Magliano, D. J., Nelson, R. G. & Pavkov, M. E. The global epidemiology of diabetes and kidney disease. Adv. Chronic Kidney Dis. 25(2), 121–132 (2018).
Yau, A., Parikh, S. V. & Almaani, S. Diabetic kidney disease: The “silent” majority?. Kidney Int. Rep. 6(12), 2939 (2021).
Guo, K. et al. Prevalence of chronic kidney disease and associated factors in Chinese individuals with type 2 diabetes: Cross-sectional study. J. Diabetes Complicat. 30(5), 803–810 (2016).
Janmohamed, M. N. et al. Prevalence of chronic kidney disease in diabetic adult out-patients in Tanzania. BMC Nephrol. 14(1), 183 (2013).
World Bank. Diabetes prevalence (% of population ages 20 to 79). https://data.worldbank.org/indicator/SH.STA.DIAB.ZS. Published 2021. (Accessed 22 Feb 2023).
Ong, L. M., Bavanandan, S., Hooi, L. S. & Choo, C. L. 29th Report of the Malaysian Dialysis and Transplant Registry 2021 (2022).
Ministry of Health Malaysia. National Diabetes Registry Report 2013–2019 (2020).
Wan, K. S., Moy, F. M., Mustapha, F. I., Ismail, M. & Hairi, N. N. Changes in body mass index, glycosylated hemoglobin A1C, blood pressure, and LDL-cholesterol among type 2 diabetes patients in Malaysia: A population-based longitudinal study. J. Diabetes. 13(11), 915–929 (2021).
Saminathan, T. A. et al. Prevalence of chronic kidney disease and its associated factors in Malaysia; findings from a nationwide population-based cross-sectional study. BMC Nephrol. 21(1), 344 (2020).
World Health Organization. Noncommunicable Disease Facility-Based Monitoring Guidance: Framework, Indicators and Application (World Health Organization, 2022).
Mahmud, S. H., Mohamed, N. E. & Bakar, A. A. Implementing quality assurance in public health facilities: The Malaysian experience. J US-China Public Adm. 12(10), 752–758 (2015).
Institute for Public Health. National Health and Morbidity Survey (NHMS) 2019: Vol 1: NCDs - Non-communicable Diseases: Risk Factors and other Health Problems (2020).
Cheo, S. W. et al. A practical approach to chronic kidney disease in primary care. Malays. Fam. Phys. 17(1), 10 (2022).
de Boer, I. H. et al. KDIGO 2020 clinical practice guideline for diabetes management in chronic kidney disease. Kidney Int. 98(4), S1–S115 (2020).
Levey, A. S. et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 150(9), 604–612 (2009).
Ministry of Health Malaysia. Clinical Practice Guidelines. Management of Type 2 Diabetes Mellitus. (6th edition). (2020).
Jalalonmuhali, M., Lim, S. K., Md Shah, M. N. & Ng, K. P. MDRD vs. CKD-EPI in comparison to 51Chromium EDTA: A cross sectional study of Malaysian CKD cohort. BMC Nephrol. 18(1), 1–6 (2017).
Ministry of Health Malaysia. Clinical Practice Guidelines on Management of Obesity. 2004.
World Health Organization Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 363(9403), 157–163 (2004).
Ministry of Health Malaysia. Clinical Practice Guidelines. Management of Hypertension 5th edition (2018) (2018).
Low, S. et al. Prevalence of chronic kidney disease in adults with type 2 diabetes mellitus. Ann. Acad. Med. Singap. 44(5), 164–171 (2015).
Parving, H.-H., Lewis, J., Ravid, M., Remuzzi, G. & Hunsicker, L. Prevalence and risk factors for microalbuminuria in a referred cohort of type II diabetic patients: A global perspective. Kidney Int. 69(11), 2057–2063 (2006).
Bolignano, D. & Zoccali, C. Non-proteinuric rather than proteinuric renal diseases are the leading cause of end-stage kidney disease. Nephrol. Dial. Transplant. 32(suppl_2), ii194–ii199 (2017).
Jitraknatee, J., Ruengorn, C. & Nochaiwong, S. Prevalence and risk factors of chronic kidney disease among type 2 diabetes patients: A cross-sectional study in primary care practice. Sci. Rep. 10(1), 6205 (2020).
Khunti, K. & Davies, M. J. Clinical inertia—time to reappraise the terminology?. Prim. Care Diabetes. 11(2), 105–106 (2017).
Bujang, M. A. et al. Forecasting the incidence and prevalence of patients with end-stage renal disease in Malaysia up to the year 2040. Int. J. Nephrol. 2017, 2735296 (2017).
Ismail, H., Abdul Manaf, M. R., Abdul Gafor, A. H., Mohamad Zaher, Z. M. & Ibrahim, A. I. N. Economic burden of ESRD to the Malaysian health care system. Kidney Int. Rep. 4(9), 1261–1270 (2019).
Ministry of Health Malaysia. National Action Plan for Healthy Kidneys (ACT-KID) 2018–2025. Putrajaya (2018).
Yang, L. et al. Risk factors of chronic kidney diseases in Chinese adults with type 2 diabetes. Sci. Rep. 8(1), 14686 (2018).
De Cosmo, S. et al. Kidney dysfunction and related cardiovascular risk factors among patients with type 2 diabetes. Nephrol. Dial. Transpl. 29(3), 657–662 (2014).
Bae, E. H. et al. Underweight and weight change increases end-stage renal disease risk in patients with diabetes: A nationwide population-based cohort study. Nutrients. 14(1), 154 (2021).
Duan, J. et al. Prevalence and risk factors of chronic kidney disease and diabetic kidney disease in Chinese rural residents: A cross-sectional survey. Sci. Rep. 9(1), 10408 (2019).
Duan, J.-Y. et al. Prevalence and risk factors of chronic kidney disease and diabetic kidney disease in a central Chinese urban population: A cross-sectional survey. BMC Nephrol. 21, 1–13 (2020).
Acknowledgements
We thank the Director-General of Health Malaysia for the permission to publish the research findings.
Author information
Authors and Affiliations
Contributions
K.S.W conceived the study, curated the data, did statistical analysis, interpreted the results, drafted, finalised, and approved the manuscript. N.N.H, F.M, M.F.M.Y, H.M.R, M.I, F.M.M and N.A.A conceived the study, interpreted the results, finalised, and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
target these modifiable factors to manage DKD and prevent its progression to ESKD.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wan, K.S., Hairi, N.N., Mustapha, F. et al. Prevalence of diabetic kidney disease and the associated factors among patients with type 2 diabetes in a multi-ethnic Asian country. Sci Rep 14, 7074 (2024). https://doi.org/10.1038/s41598-024-57723-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-57723-6
- Springer Nature Limited