Abstract
In the Americas, the fall armyworm (Spodoptera frugiperda) exists in two genetically distinct strains, the corn (C) and rice (R) strains. Despite their names, these strains are not associated with host plant preferences but have been shown to vary in pheromone composition and male responses. Recently, S. frugiperda was detected in Africa as an invasive species, but knowledge about variation in strain types, pheromone composition and inter-strain mating of populations of the pest in the continent has not been fully examined. Therefore, this study aimed to investigate variations, if any in the pheromone composition of female moths, male moth responses, and mating between C and R mitotypes of S. frugiperda populations in Kenya, as well as their geographic distribution. Strains (mitotypes) of S. frugiperda were identified using mitochondrial DNA (mtDNA) markers, and their pheromonal composition determined by coupled gas chromatography–mass spectrometric (GC–MS) analysis. Male moth responses to these compounds were evaluated using GC-electroantennographic detection (EAD), electroantennogram (EAG), and wind tunnel assays. Oviposition assays were used to determine whether R and C mitotype moths could mate and produce eggs. The results showed that both the R and C mitotypes were present, and there were no statistically significant differences in their distribution across all sampled locations. Five pheromone compounds including (Z)-7-dodecenyl acetate (Z7-12:OAc), (Z)-7-tetradecenyl acetate (Z7-14:OAc), (Z)-9-tetradecenyl acetate (Z9-14:OAc), (Z)-11-tetradecenyl acetate (Z11-14:OAc) and (Z)-11-hexadecenyl acetate (Z11-16:OAc), were detected in the pheromone glands of female moths of both mitotypes, with Z9-14:OAc being the most abundant. The relative percentage composition of Z9-14:OAc was similar in both mitotypes. However, the R mitotype had a 2.7 times higher relative percentage composition of Z7-12:OAc compared to the C mitotype moth, while the C mitotype moth had a 2.4 times higher relative percentage composition of Z11-16:OAc than the R mitotype moth. Male moths of both mitotypes exhibited similar responses to the pheromone compounds, showing the strongest responses to Z9-14:OAc and Z7-12:OAc in electrophysiological and behavioural assays. There was mating between R and C mitotypes with egg production comparable to mating within the same mitotype. Our results revealed that differences between the two S. frugiperda mitotypes are characterized by female moth pheromone composition rather than male moth responses to the pheromones, and that this does not prevent hybridisation between the mitotypes, which may have implications for their management.
Similar content being viewed by others
Introduction
Insects use chemical signals, known as semiochemicals, to communicate with members of their own species (intraspecific) as well as other species (interspecific)1,2. Pheromones are common intraspecific semiochemcials used by insects, including many lepidopteran species, where moths rely on sex pheromones produced by female moths to find mates for reproduction3,4,5. Male moths exhibit dramatic behavioural and physiological responses to these pheromones6,7. However, the sexual communication system of several lepidopteran species can vary widely across different geographic regions8,9,10,11,12,13,14,15, which may have consequences for gene flow between populations and eventually speciation.
Previous studies have shown that the S. frugiperda, exhibits geographic variation in sexual communication16,17. The sex pheromone composition of female S. frugiperda includes a blend of compounds, with (Z)-9-tetradecenyl acetate (Z9-14:OAc) identified as the major sex pheromone component and (Z)-7-dodecenyl acetate (Z7-12:OAc) as the critical secondary sex pheromone component. The relevance and importance of other compounds in the pheromone gland, such as (E)-7-dodecenyl acetate (E7-12:OAc), (Z)-9-dodecenyl acetate (Z9-12:OAc), (Z)-10-tetradecenyl acetate (Z10-14:OAc), (Z)-11-tetradecenyl acetate (Z11-14:OAc), and (Z)-11-hexadecenyl acetate (Z11-16:OAc), remain unclear and may vary depending on the population14,16,18,19. For example, while E7-12:OAc has been detected in the female sex pheromone blend of S. frugiperda populations in Brazil16, it has not been reported as an active component in populations from other regions, suggesting possible geographic variation in the sex pheromone composition of this pest. Understanding such geographic variation and its impact on male responses is crucial for the development of efficient and locally adapted pest management strategies, particularly in the context of pheromone-based monitoring, which plays a vital role in sustainable pest management.
The two distinct strains of S. frugiperda, known as the 'C' and 'R' strains, may account for the regional variation in female pheromone production and male responses14. These strains can be clearly distinguished using mitochondrial CO1 and nuclear Tpi gene markers, while the term 'mitotype' specifically pertains to mitochondrial DNA markers20,21,22. It is known that female C strains found in the USA release a higher amount of Z11-16:OAc, whereas female R strains release a higher amount of Z7-12:OAc and Z9-12:OAc23. However, results from Louisiana showed different findings, whereby C-strain females released more Z9-14:OAc and less Z11-16:OAc than R-strain females24. Interestingly, despite variations in pheromone composition, the attraction of males was found to be unaffected by Z11-16:OAc14. However, C-strain males in some regions responded more strongly to Z7-12:OAc than the R-strain males14. This indicates that variations in pheromone composition can be influenced by both differences between the strains and the geographic variation within the strains. Furthermore, several other factors, including host plant volatiles, interspecific olfactory cues, and environmental factors such as temperature, humidity, and photoperiod, may contribute to the observed regional variation in sexual communication25,26,27,28,29,30. Such variation in sexual communication reliant on sex pheromones of S. frugiperda may impact on their management strategies that depend on effective monitoring of the pest population.
Spodoptera frugiperda has recently invaded Africa31, spreading rapidly to most sub-Saharan countries32, and the presence of both strains has been confirmed on the continent33,34,35,36,37. A recent study indicates geographic variation in male moth responses between West African populations (Benin and Nigeria) and populations of S. frugiperda in America38. However, the extent of this variation remains unclear, and there is no available information regarding East African S. frugiperda populations. Furthermore, the introduction of S. frugiperda to new regions, including Africa, increased the likelihood of inter-strain mating and gene flow between different populations39, which may lead to new combinations of traits. Despite this, the possibility of inter-strain mating between the R and C mitotypes of S. frugiperda has not been examined in Africa.
In this study, we hypothesized that there would be variations in the pheromone composition and response of S. frugiperda populations in Africa. Additionally, mating would occur between the two mitotypes of S. frugiperda. To test these hypotheses, firstly, we identified S. frugiperda mitotypes using mtDNA markers from field-collected specimens across populations in Kenya. Secondly, we identified the pheromone compounds present in female glands using GC–MS. Thirdly, we evaluated male moth responses to the pheromone compounds using GC-EAD, EAG, and wind tunnel assays. Finally, we conducted mating and oviposition assays to determine whether R and C mitotype moths would mate and produce eggs. Overall, this study provides valuable insights into sex pheromone communication and mating potential, contributing to the development of effective pheromone-based monitoring and management strategies for S. frugiperda in Africa.
Results
Genetic identification and distribution of C and R mitotypes of S. frugiperda
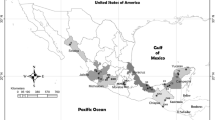
Using Lep, LCO 1490, and HCO 2198 markers, a clear separation was found between the C and R mitotypes of sampled S. frugiperda. Of all moths collected, 73 belonged to the C mitotype, while the remaining 58 were identified as the R mitotype (Fig. 1). Both the R and C mitotypes of S. frugiperda were found in all the sampled locations (Fig. 2a). However, there were no significant differences in the number of R (df = 6, χ2 = 3.3, P > 0.05) and C (df = 6, χ2 = 1.7, P > 0.05) mitotypes of S. frugiperda found across locations (Fig. 2b).
Phylogenetic analysis of S. frugiperda based on mtCOI sequences from publicly available GenBank and representative haplotypes from this study. The USA R (U72977) and C (U72974) mitotypes are added for reference. V is the representative samples obtained from the study area, while n is the number of individuals classified as belonging to R and C mitotypes. A sequence from Spodoptera exigua (KX580621) is included as an out-group. The number of moths belonging to R and C mitotypes, as well as their identity with other samples from publicly available GenBank, are listed in Supplementary Table S1.
Map showing the S. frugiperda collection sites in Kenya (a). The figure was generated using QGIS software (version 3.30.2; http://qgis.org). Number of individuals from the R and C (n = 58 and 73, respectively) mitotypes of S. frugiperda found in different locations in Kenya (b). A Chi-squared test indicated no significant difference (ns) in the frequencies of S. frugiperda mitotypes among locations.
Differences in female S. frugiperda pheromone production between C and R mitotypes
Female moths of both the C and R mitotypes were found to have five pheromone compounds in their glands. In both mitotypes, the major sex pheromone component Z9-14:OAc exhibited a higher relative percentage than other compounds (Fig. 3). The relative percentage composition of Z9-14:OAc was comparable between the mitotypes, and Tukey’s test did not reveal significant differences in its amounts (df = 4, P > 0.05). However, the rice mitotype female moths had a 2.7 times higher relative percentage composition of the critical secondary sex pheromone component Z7-12:OAc compared to the corn mitotype female moths, while the corn mitotype female moths had a 2.4 times higher relative percentage composition of the compound Z11-16:OAc than the rice mitotype female moths. The relative percentage composition of these compounds was significantly different between the two mitotypes (df = 4, P < 0.05). The two minor compounds, Z7-14:OAc and Z11-14:OAc, were found in very low relative percentages and Tukey’s test did not detect significant differences in the amounts of these compounds between the two mitotypes (df = 4, P > 0.05) (Fig. 3).
Comparison of relative percentage pheromone composition between the R and C mitotypes of female S. frugiperda moths. ns not significant, *P < 0.05 using Tukey’s range test. A heatmap showing the relative abundance of compounds in the two mitotypes and their corresponding chromatograms are shown in supplementary Figs. 1 and 2.
GC-EAD analysis
Both the laboratory-established C and R mitotypes of male S. frugiperda exhibited significantly higher EAD responses to Z7-12:OAc and Z9-14:OAc (df = 6, P < 0.001) compared to other tested compounds (Fig. 4). There were no significant differences in EAD responses to these two compounds between the two mitotypes (df = 6, P > 0.05). Moth responses to E7-12:OAc, Z11-14:OAc, and Z11-16:OAc were the lowest in both mitotypes, and there were no significant differences (df = 6, P > 0.05) in EAD responses to these three compounds between the two mitotypes (Fig. 4).
EAG responses
Male S. frugiperda moths from both the laboratory-established C and R mitotypes exhibited comparable EAG responses to pheromone compounds (Fig. 5). The moth response increased relatively with the doses of Z7-12:OAc and Z9-14:OAc. The moth response relatively increased as the doses of Z7-12:OAc and Z9-14: OAc increased (Fig. 5). However, there were no significant differences in the moth responses to these two compounds between the two mitotypes of male moths, indicating similar responsiveness (P > 0.05) (Fig. 5).
Wind tunnel bioassay
The responses of male S. frugiperda to pheromone compounds varied significantly in a wind tunnel (Fig. 6). Moths of both the laboratory-established C and R mitotypes significantly increased their approaching and landing responses to Z9-14:OAc and Z7-12:OAc (Fig. 6a, b) (df = 6, P < 0.001). Lower approaches by both moth mitotypes were observed when they were exposed to Z7-14:OAc, E7-12:OAc, Z11-14:OAc, and Z11-16:OAc when compared with the other tested compounds. The landing response of male moths from both mitotypes to Z11-16:OAc was significantly higher than to Z7-14:OAc, E7-12:OAc, and Z11-14:OAc (df = 6, P < 0.05). On the other hand, male moths of both mitotypes exhibited similarly low landing responses to Z7-14:OAc, E7-12:OAc, and Z11-14:OAc, with no significant differences between them (df = 6, P > 0.05) (Fig. 6a, b).
Behavioural responses of R and C mitotypes of S. frugiperda moths towards different pheromone compounds in a wind tunnel. The data represent the number of approaches (a) and landing responses (b) per male tested (n = 10). Bars with different letters are significantly different at p ≤ 0.05 using chi-squared tests.
Inter-mitotype mating
There was egg production in all laboratory reared S. frugiperda mitotype combinations, with no statistical differences (P > 0.05) between any of the mating combinations (Fig. 7a). DNA analysis of field collections of S. frugiperda samples revealed that female moths from the C mitotype mated with male moths from the R mitotype, and likewise, female moths from the R mitotype mated with male moths from the C mitotype (Fig. 7b). The number of eggs produced by these pairs were not significantly different (P > 0.05) (Fig. 7b).
Mean number (± SE) of eggs laid by different combinations of S. frugiperda moths from mitotype-specific laboratory colonies (n = 20) (a). Mean number (± SE) of eggs laid by different combinations of S. frugiperda moths from randomly paired adult moths collected from the field (n = 21) (b). Bars with different letters are significantly different at P ≤ 0.05. Phylogenetic analysis of S. frugiperda adult pairs that produced eggs is shown in supplementary Fig. 3.
Discussion
In this study, we investigated the potential variation in female moth pheromone composition and male moth response between R and C mitotype S. frugiperda populations in Africa, specifically in Kenya. Among the five pheromone compounds identified in the female glands, the major sex pheromone component Z9-14:OAc was the most abundant, with a relative percentage composition similar in both mitotypes. On the other hand, the R mitotype females of S. frugiperda had a higher relative percentage composition of Z7-12:OAc, while the C mitotype female moths had a higher relative percentage composition of Z11-16:OAc. This study also confirmed that the S. frugiperda populations in Kenya have the same pheromone profiles as the S. frugiperda populations in America, which also exhibited higher relative percentage compositions of Z7-12:OAc in R strains and Z11-16-OAc in C strains14,19,23. However, previous studies have reported variation in pheromone composition among different S. frugiperda populations. For instance, R strain of S. frugiperda from Louisiana R strain had a higher amount of Z11-16:OAc, while the C strain had higher amount of Z9-14:OAc24. Similarly, C strain and R–C hybrid descendant females from Benin and Nigeria produced relatively higher amounts of Z9-14:OAc, Z7-12:OAc, and Z11-16:OAc than Z9-12:OAc and E7-12:OAc38. Other minor compounds were also detected in S. frugiperda populations in different regions, such as Z9-12:OAc in China, Florida, Brazil, Nigeria, and Benin, E7-12:OAc and (Z)-10-tetradecenyl acetate in Brazilian S. frugiperda populations16,19,38,40. Recently, Tabata et al. (2023)41 also reported the detection of another key minor compound, (Z,E)-9,12-tetradecadienyl acetate, in female extracts in Japan. We checked for the presence of E7-12:OAc, and results indicated no detectable amounts present in our samples. However, we identified a minor compound, Z7-14:OAc, in the female glands, which had not previously been reported in S. frugiperda moths but had been detected in several other lepidopteran insects42,43,44. Further research is required to determine whether this compound is a pheromone component because some compounds can be detected in the female gland but may not constitute the pheromone blend emitted by females, and hence male moths may not respond to it.
Our results from the GC-EAD and EAG experiments provide important insights into the pheromonal compounds responsible for male attraction in S. frugiperda moths. The fact that both C and R mitotypes of male moths had higher antennal responses to Z9-14:OAc and Z7-12:OAc suggests that these two compounds are important bioactive components of the female pheromone blend. Additionally, the dose–response study using EAG revealed that male moth responses increased as the doses of Z9-14:OAc and Z7-12:OAc increased. This underscores the significance of identifying key pheromone compounds and determining the detectable amounts by target insects to develop effective pest management strategies. Similar to our findings, higher antennal responses to Z9-14:OAc were observed in S. frugiperda moths from Florida38, and weak antennal responses to Z11-16OAc were observed in S. frugiperda populations from China, Florida and Brazil38,45. However, it is important to note that there are variation in the response of different S. frugiperda populations to these compounds. For example, E7-12:OAc evoked weak antennal responses in Kenya S. frugiperda populations in our study, although it elicited higher antennal responses in the S. frugiperda populations from Mexico and Brazil16,38. Additionally, C-strain males in Florida and Puerto Rico showed a higher response to Z7-12:OAc than R-strain males14.
Our wind tunnel bioassay produced results that were consistent with those obtained using GC-EAD and EAG techniques. We observed that males from both moth mitotypes exhibited a greater level of attraction towards Z9-14:OAc and Z7-12:OAc compared to the other compounds tested in the wind tunnel. Both moth mitotypes showed a higher frequency of approach and landing responses towards Z9-14:OAc and Z7-12:OAc relative to the other compounds tested. According to Wang et al. (2022)45, Z9-14:OAc alone induces male sexual behaviours in a wind tunnel, including flying, upwind anemotaxis, approach, and landing. Similarly, Z9-14:OAc showed strong attraction to both strains of S. frugiperda in Puerto Rico14. In Costa Rica, Z7-12:OAc and Z9-12:OAc were highly attractive to S. frugiperda when presented alone46. On the other hand, Cruz Esteban et al. (2020)47 observed higher moth attraction to blends of Z7-12:OAc + Z9-14:OAc than to single compounds in Mexico. We found the attraction of male moths of both mitotypes to Z11-16:OAc was low and not clear in the wind tunnel, which has also been observed by Cruz Esteban et al. (2020)47 in Mexico. Moreover, E7-12:OAc, Z7-14:OAc, and Z11-14:OAc did not attract male moths of both mitotypes, and E7-12:OAc was not an essential component for S. frugiperda sexual communication in China40. However, these compounds may not attract males by themselves but can significantly enhance attraction when added to the two critical sex pheromone components, Z7-12:OAc and Z9-14:OAc. It is crucial to consider the potential synergistic effects constituent components rather than testing single compounds. Furthermore, olfactory responses of S. frugiperda to pheromone blends may be influenced by various factors, including background host plant volatiles, interspecific olfactory cues, and environmental factors such as temperature, humidity, and photoperiod, that may vary geographically. Future work should consider examining the effect of these factors on the pheromone composition and behavioral responses of S. frugiperda.
Our study showed that mating between the two identified mitotypes of S. frugiperda results in comparable egg production to that observed within the same mitotype. Even though the sample size was small, this results imply that hybrid offsprings with intermediate characteristics between the two mitotypes or expressing the dominant gene of one of the mitotypes can be produced. Our findings are consistent with recent studies that have reported evidence of hybridization between C and R strain populations in Africa, which has led to the existence of genetically hybrid S. frugiperda populations48,49,50,51. These results have important implications for the genetic diversity and potential spread of S. frugiperda populations in Africa. It highlights the need for further investigation into the effects of hybridization on the biology and ecology of this pest species. However, we recommend further research using Tpi genes to enhance the accuracy of strain identification, as mitochondrial markers alone may not reliably distinguish the two strains.
Conclusion
In conclusion, our study provides important insights into the pheromone communication system of S. frugiperda in Africa, and our findings have practical implications for developing effective pest monitoring and management strategies. The high attraction of both mitotypes to Z9-14:OAc and Z7-12:OAc highlights the potential of these compounds to develop pheromone-based management techniques. Moreover, our study demonstrated that the S. frugiperda population in Kenya has the same pheromone profile as the American S. frugiperda, which suggests that pheromone-based management strategies developed for S. frugiperda in America could be applicable to African populations. However, mating between C and R mitotypes of S. frugiperda populations in Africa raises questions about the potential impact of hybridization on pest management strategies. Therefore, further research is needed to fully understand the complex interactions between different pheromone compounds and male moth response, as well as the potential differences in pheromone production and male response between hybrid and pure mitotypes (R and C mitotypes). Overall, our findings not only provide a basis for further research but also offer valuable insights for the development of more effective and sustainable pheromone-based management strategies for S. frugiperda populations in Africa.
Materials and methods
Spodoptera frugiperda sampling and colony establishment
Third and fourth instar larvae were collected from maize plants in various locations within S. frugiperda affected areas in Kenya (Fig. 2a) and reared under controlled laboratory conditions (24 ± 4 °C, 60 ± 5% RH, and a 12L:12D photoperiod). The larvae were placed in vials (30 ml) and fed an artificial diet as described in Prasanna et al. (2018)52. The sex of the pupae was identified by observing the identifying features of the terminal segments using a 10 × Leica EZ4 HD stereo microscope (Leica Microsystems, Wetzlar, Germany). Female pupae have a wider gap between the genital and anal openings than male pupae53. A single pair of identified sexed pupae was put inside a Petri dish in an oviposition cage (20 cm × 20 cm 20 cm). Newly emerged adults were fed using a cotton ball soaked in a 10% honey-water solution, and wax paper was placed inside the oviposition cage as an egg laying substrate. Adult pairs that produced fertile offspring were genetically analysed to identify their mitotype, and the offspring were labelled and reared on artificial diet to establish a mitotype-specific laboratory colony. Only the offspring resulting from the mating of male and female parents belonging to the same mitotype were used to establish a mitotype-specific laboratory colony. The second to fourth laboratory generations were used for the pheromone extraction, electrophysiological and wind tunnel assays.
Mitotypes identification
The Isolate II Genomic DNA Kit (Bioline, London, United Kingdom) was used to extract genomic DNA from individual insects, following the method described by Gichuhi et al. (2020)33. The DNA was eluted in a final volume of 50 μl and quality and quantity checks were performed with the Nanodrop 2000/2000c Spectrophotometer (Thermo Fisher Scientific, Wilmington, USA). Polymerase chain reaction (PCR) was done to amplify the mitochondrial COI gene using LepF1 5′ ATTCAACCAATCATAAAGATATTGG 3′ and LepR1 5′ TAAACTTCTGGATGTCCAAAAAATCA 3′ 54 markers in addition to the traditional barcode region markers LCO 1490 5′ GGTCAACAAATCATAAAGATATTGG 3′ and HCO 2198 5′ TAAACTTCAGGGTGACCAAAAAATCA 3′ 55. The PCRs were performed in a 20 µL volume containing 5X My Taq Reaction Buffer (5 mM dNTPs, 15 mM MgCl2, stabilizers and enhancers), 0.5 pmol µl-1 of each primer, 0.5 mM MgCl2, 0.0625 U µl−1 My Taq DNA polymerase (Bioline, London, UK) and 15 ng µl−1 of DNA template. The reactions were set up in the Nexus Mastercycler gradient (Eppendorf, Germany) with the following cycling conditions: initial denaturation for 2 min at 95 °C, followed by 40 cycles of 30 s at 95 °C, 30 s annealing (52 °C for LepF1/R1 and 54.1 °C for LCO/HCO), extension for 1 min at 72 °C, then a final elongation step of 10 min at 72 °C. The PCR-amplified products were separated on a 1.2% agarose gel. KETA GL imaging system trans-illuminator was utilized to analyse and record DNA bands on the gel (Wealtec Corp, Meadowvale Way Sparks, Nevada, USA). Following the manufacturer's instructions, successively amplified products were excised and purified using the Isolate II PCR and Gel Kit (Bioline, London, UK). Purified samples were sent to Macrogen Europe BV (Meibergreef, Amsterdam, the Netherlands) for bi-directional sequencing.
Sequence analyses
Geneious Version 8 (http://www.geneious.com) was used to assemble and edit the successful sequences56. The primer sequences were removed from the consensus sequences derived from both the forward and reverse reads, resulting in consensus sequences with a length of approximately 608 base pairs. Similarity searches were performed by querying the consensus sequences via Basic Local Alignment Search Tool (BLAST) at the GenBank database hosted by the National Centre for Biotechnology Information (NCBI) to confirm the species' identity. The BLAST algorithm searches for regions of local similarity between sequences by comparing consensus sequences to reference sequences in the GenBank database. In addition, the query was conducted in BOLD (Barcode of Life Database). MEGA version X was used for phylogenetic and molecular evolutionary analyses57 using the Maximum Likelihood method based on the Tamura-Nei model58. The tree's reliability was assessed using 1000 bootstrap replications.
Pheromone extraction and analysis
Virgin female moths (2 to 3 days old) exhibiting various behaviours to call their mates, such as wing fanning and extruding the tip of the abdomen. Female exhibiting such behaviours were selected for pheromone extraction. During the first two to four hours of the dark period, the abdominal tip (between the 8th and 9th abdominal segments) of moths was gently excised. The excised glands were placed individually into a glass vial with 50 μl hexane. After 30 min, the gland was removed from the solvent, and the extracts were kept at −20 °C until used for GC–MS analysis. The pheromone extract samples were analyzed using an Agilent 5975/6880 GC–MS, with both DB-Wax (60 m × 0.25 mm × 0.25 μm; Agilent) and an HP-5 MS (30 m × 0.25 mm × 0.25 μm, Agilent) columns. Helium was used as the carrier gas at 1.2 ml/min. The oven temperature was kept at 35 °C for 5 min and then increased to 280 °C at a rate of 10 °C per minute. The pheromone samples (2 μl) from the original 50 μl hexane extracts were directly injected into the GC using an autosampler. Retention times were converted to retention indices relative to the retention times of n-alkanes standards according to Adams (1996)59 and Babushok (2015)60. The compounds were tentatively identified by comparing mass spectra and the Kovats retention index to the Adams library and NIST databases. For confirmation, a comparison of the retention times, retention indices and mass spectra of authentic standards run under the same conditions was carried out. All compounds found in the control were regarded as contaminants and thus ignored during identification.
GC-EAD analysis
This experiment examined the moth antennal responses to pheromone compounds to identify the compounds that elicited the greatest antennal response. The male moth responses to the five pheromone compounds identified from the female gland of S. frugiperda (Z7-12:OAc, Z7-14:OAc, Z9-14:OAc, Z11-14:OAc, and Z11-16:OAc) were studied. E7-12:OAc, which has been discovered in Brazilian S. frugiperda populations, was included to determine whether it elicited antennal responses in S. frugiperda populations from Africa. The synthetic standard of these sex pheromone compounds (90% purity) were purchased from Alfa Chemistry (New York, USA). Electroantennogram detection responses to pheromones were recorded using GC-EAD 2011 (V.1.2.3, Syntech, Kirchzarten, Germany) and an Agilent Technologies 6890 GC (Santa Clara, CA, USA) equipped with a DB-Wax capillary column (60 m × 0.25 mm × 0.25 μm; Agilent). The GC oven temperature programmed from 40 °C for 3 min, 10°Cmin−1 to 240 °C (held for 5 min). Helium was the carrier gas at 1.2 ml min−1. The antennae of 3–5-day-old unmated males were clipped at the base of flagella segments with micro scissors, and the tips were also clipped. The basal part of the antenna was inserted into a glass electrode filled with Beadle-Ephrussi Ringer solution as the reference electrode. The last one to two distal ends of antennal segments were inserted into the tip of the recording glass capillary electrode. The signals generated by the antenna were passed through a high-impedance amplifier (NL1200; Syntech, Hilversum, Netherlands). An antenna was stimulated with 10 μg of each synthetic pheromone compound that was dissolved in 2 μl of hexane and injected into the GC. Five moth antennae from each mitotype were tested for each compound.
Pheromone dose response study
EAG recordings of synthetic blends containing Z7-12:OAc and Z9-14:OAc (compounds that elicited the greatest antennal response in GC-EAD analysis) were evaluated at doses of 0.01, 0.1, 1, and 10 μg, using the antennae of 3 to 5-day-old unmated male S. frugiperda. Adult moths were monitored as described above. Serial dilutions of the synthetic compounds were prepared in hexane to create 0.01, 0.1, 1, and 10 μg solutions.
Wind tunnel bioassay
The attraction of male S. frugiperda moths to female sex pheromone was studied using an aluminium framed wind tunnel (120 × 32 × 32 cm). The experiments were conducted at 25 ± 2 °C and 60 ± 10% relative humidity, while air speed inside the wind tunnel was 20 cm/s. The responses of male moths to single doses (10 μg) of pheromone compounds were examined. Each compound dissolved in 10 μl of hexane was pipetted onto a small piece of filter paper (1 cm2) and placed outside the wind tunnel in a glass cylinder with quick-fit connection on both ends using a portable volatile collection kit (B.J. Pye, Hertfordshire, UK). Volatiles were drawn from the container using charcoal-filtered air at a rate of 500 ml min−1 a vacuum pump. Moths were acclimatized to the wind tunnel room conditions 1 h before testing. Unmated male moths (3–5 days old) were carefully introduced individually through a hole located at the top of the wind tunnel, 120 cm away from the odour source during 2 to 4 h within the scotophase. A total of ten moths of each mitotype were tested for individual compounds. A light source provided by a bulb (40 Watt, fitted with red filter) positioned at 50 cm above the tunnel was used to observe moth movement. The moth responses, such as the number of approaches (20 cm from the pheromone source) and flight distance (the distance from the release point to where the moth ultimately settled after approaching the odour source), were recorded visually. Moths that did not respond within 5 min were discarded. Filter paper impregnated with 10 μl hexane was included as controls for all experiments.
Mating between C and R mitotypes
Two experiments were conducted to determine whether R and C mitotype moths’ could mate and produce eggs. In the first experiment, an oviposition test was conducted using a known S. frugiperda (mitotype-specific) colony. Pupae were sexed and monitored as described above. A single pair of sexed adult moths from mitotype-specfic colonies was allowed to lay eggs on wax paper in an oviposition cage. The moths were combined as described below (Table 1). Five pairs of S. frugiperda moths were tested from each combination. Leica EZ4 HD stereo microscope (Leica Microsystems, Wetzlar, Germany) with a 16× magnification was used to count the egg masses and number of eggs per mass.
In the second experiment, unknown mitotypes of S. frugiperda moths were used due to the possibility of greater genetic diversity in moths collected from the field as compared to the controlled, known mitotypes used in the first experiment. The larvae were collected from different locations and monitored as described above. Pupae from field collections and unknown mitotypes were randomly selected and subsequently sexed using the method described above. A single pair of sexed moths was allowed to lay eggs on wax paper placed inside the oviposition cage. After oviposition, all adult pairs that produced eggs were genetically analysed to identify their mitotype. Eggs and egg mass produced from randomly sexed pupae and molecularly identified mating pairs of C male (Cm) vs. R female (Rf), C female (Cf) vs. R male (Rm), and C male (Cm) vs. C female (Cf) were counted to determine whether mating pairs of R–C mitotypes differed in their ability to produce eggs. By chance, during the random selection and sexing of pupae from field collections, no pupae of R mitotype males (Rm) and R mitotype females (Rf) were selected. Consequently, no eggs produced from this specific combination. A total of seven pairs of moths were used for this experiment.
Data analysis
The normality and homogeneity of all data were checked using the Shapiro–Wilk and Bartlett tests, respectively. The Chi-squared test was used to analyze the number of R and C mitotypes of S. frugiperda found across different locations, as well as the moth approaches and landing responses in the wind tunnel. All the pheromone compounds found in 50 female moths of each mitotype were converted into relative percentages, and a heatmap clustering analysis (supplementary material) was used to identify the most abundant compounds in the female gland. Further analysis was conducted to examine whether there were differences in the relative percentages of pheromone composition between the R and C mitotypes using analysis of variance (ANOVA). The spike amplitude (mV) of GC-EAD and EAG responses were transformed using log (x + 1) to meet assumptions of normality and homogeneity and subjected to a one-way analysis of variance. Tukey's range test (p ≤ 0.05) was used to compare the mean values of the data. Due to data overdispersion, egg counts were analysed using a generalized linear model (GLM) with a negative binomial distribution. The number of S. frugiperda eggs laid by each mitotype combination was used as the response variable, and mean values of the data were compared using the 'emmeans' package. R statistical software version 4.0.4 was used for all statistical analyses.
Data availability
The raw data generated during the current study are available from the corresponding author upon reasonable request.
References
Montagné, N., Gévar, J. & Lucas, P. Semiochemicals and Communication in Insects. Extended Biocontrol 3, 173–181 (2022).
Regnier, F. E. & Law, J. H. Insect pheromones. J. Lipid Res. 9, 541–551 (1968).
Abd El-Ghany, N.M. Pheromones and chemical communication in insects. In Pests, Weeds and Diseases in Agricultural Crop and Animal Husbandry Production. IntechOpen. Vol. 3. 16–30 (2020).
Landolt, P. J. Sex attractant and aggregation pheromones of male phytophagous insects. Am. Entomol. 43, 12–22 (1997).
Cardé, R. T. & Haynes, K. F. Structure of the pheromone communication channel in moths. Adv. Insect Chem. Ecol. 4, 283–332 (2004).
Lemmen, J. & Evenden, M. Peripheral and behavioral plasticity of pheromone response and its hormonal control in a long-lived moth. J. Exp. Biol. 212, 2000–2006 (2009).
Ganai, M. A. et al. Pheromones in lepidopteran insects: Types, production, reception and its application. J. Pharmacogn. Phytochem. 6, 2552–2558 (2017).
McElfresh, J. S. & Millar, J. G. Geographic variation in sex pheromone blend of Hemileuca electra from southern California. J. Chem. Ecol. 25, 2505–2525 (1999).
Wu, W. et al. Comparative study of pheromone production and response in Swedish and Zimbabwean populations of turnip moth, Agrotis segetum. J. Chem. Ecol. 25, 177–196 (1999).
Gemeno, C., Lutfallah, A. F. & Haynes, K. F. Pheromone blend variation and cross-attraction among populations of the black cutworm moth (Lepidoptera: Noctuidae). Ann. Entomol. Soc. Am. 93, 1322–1328 (2000).
Kawazu, K. et al. Attraction of Cnaphalocrocis medinalis (Lepidoptera: Crambidae) males in Southeast Asia to female sex pheromone traps: Field tests in southernmost China, northern Vietnam and southern Philippines with three synthetic pheromone blends regarding geographic. Appl. Entomol. Zool. 40, 483–488 (2005).
Groot, A. T. et al. Geographic and temporal variation in moth chemical communication. Evolution 63, 1987–2003 (2009).
Zhu, J. et al. Reidentification of pheromone composition of Sparganothis sulfureana (Clemens) and evidence of geographic variation in male responses from two US states. J. Asia. Pac. Entomol. 12, 247–252 (2009).
Unbehend, M. et al. Geographic variation in sexual attraction of Spodoptera frugiperda corn- and rice-strain males to pheromone lures. PLoS One 9, 44–47 (2014).
Gao, K. et al. Geographic variation in sexual communication in the cotton bollworm, Helicoverpa armigera. Pest Manag. Sci. 76, 3596–3605 (2020).
Batista-Pereira, L. G. et al. Isolation, identification, synthesis, and field evaluation of the sex pheromone of the Brazilian population of Spodoptera frugiperda. J. Chem. Ecol. 32, 1085–1099 (2006).
Cruz-Esteban, S. et al. Geographic variation in pheromone component ratio and antennal responses, but not in attraction, to sex pheromones among fall armyworm populations infesting corn in Mexico. J. Pest Sci. 91, 973–983 (2018).
Tumlinson, J. H., Mitchell, E. R., Teal, P. E. A., Heath, R. R. & Mengelkoch, L. J. Sex pheromone of fall armyworm, Spodoptera frugiperda (J.E. Smith)—Identification of components critical to attraction in the field. J. Chem. Ecol. 12, 1909–1926 (1986).
Marr, M. Differences in pheromone composition between the two strains of the fall armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae). In Friedrich Schiller University Jena, Diploma Thesis (2009).
Nagoshi, R. N. et al. Genetic characterization of fall armyworm infesting South Africa and India indicate recent introduction from a common source population. PLoS One 14, e0217755 (2019).
Nagoshi, R. N. et al. Genetic comparisons of fall armyworm populations from 11 countries spanning sub-Saharan Africa provide insights into strain composition and migratory behaviors. Sci. Rep. 9, 8311 (2019).
Groot, A. T. et al. The roles and interactions of reproductive isolation mechanisms in fall armyworm (Lepidoptera: Noctuidae) host strains. Ecol. Entomol. 35, 105–118 (2010).
Groot, A. T. et al. Host strain specific sex pheromone variation in Spodoptera frugiperda. Front. Zool. 5, 1–13 (2008).
Lima, E. R. & McNeil, J. N. Female sex pheromones in the host races and hybrids of the fall armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae). Chemoecology 19, 29–36 (2009).
Delisle, J., Mcneil, J. N. & Biologie, D. D. The combined effect of photoperiod and temperature on the calling behaviour of the true armyworm, Pseudaletia unipuncta. Physiol. Entomol. 12, 157–164 (1987).
Royer, L. & McNeil, J. N. Effect of relative humidity conditions on responsiveness of European corn borer (Ostrinia nubilalis) males to female sex pheromone in a wind tunnel. J. Chem. Ecol. 19, 61–69 (1993).
Delisle, J. & Royer, L. Changes in pheromone titer of oblique- banded leafroller, Choristoneura rosaceana, virgin females as a function of time of day, age, and temperature. J. Chem. Ecol. 20, 45–69 (1994).
Landolt, P. J. & Phillips, T. W. Host plant influences on sex pheromone behavior of phytophagous insects. Annu. Rev. Entomol. 42, 371–391 (1997).
Reddy, G. V. P. & Guerrero, A. Interactions of insect pheromones and plant semiochemicals. Trends Plant Sci. 9, 253–261 (2004).
Groot, A. T., Claßen, A., Staudacher, H., Schal, C. & Heckel, D. G. Phenotypic plasticity in sexual communication signal of a noctuid moth. J. Evol. Biol. 23, 2731–2738 (2010).
Goergen, G., Kumar, P. L., Sankung, S. B., Togola, A. & Tamò, M. First report of outbreaks of the fall armyworm spodoptera frugiperda (J E Smith) (Lepidoptera, Noctuidae), a new alien invasive pest in West and Central Africa. PLoS One 11, 1–10 (2016).
CABI. Spodoptera frugiperda. In Invasive Species Compendium. https://www.cabi.org/isc (CAB International, 2022).
Gichuhi, J. et al. Diversity of fall armyworm, Spodoptera frugiperda and their gut bacterial community in Kenya. PeerJ 2020, 1–20 (2020).
Nagoshi, R. N. et al. Comparative molecular analyses of invasive fall armyworm in Togo reveal strong similarities to populations from the eastern United States and the Greater Antilles. PLoS One 12, 1–15 (2017).
Nagoshi, R. N. et al. Analysis of strain distribution, migratory potential, and invasion history of fall armyworm populations in northern Sub-Saharan Africa. Sci. Rep. 8, 1–10 (2018).
Nagoshi, R. N. et al. Genetic characterization of fall armyworm infesting South Africa and India indicate recent introduction from a common source population. PLoS One 14, 1–16 (2019).
Sarr, O. M. et al. Strain composition and genetic diversity of the fall armyworm Spodoptera frugiperda (Lepidoptera, Noctuidae): new insights from seven countries in West Africa. Int. J. Trop. Insect Sci. 41, 2695–2711 (2021).
Haenniger, S. et al. Sexual communication of Spodoptera frugiperda from West Africa: Adaptation of an invasive species and implications for pest management. Sci. Rep. 10, 3–11 (2020).
Kondidie, D. B. Genetic variability and gene flow of the fall armyworm Spodoptera frugiperda (J.E. Smith) in the Western hemisphere and susceptibility to insecticides. In University of Nebraska-Lincoln, PhD Dissertation (2011).
Jiang, N. J. et al. Revisiting the sex pheromone of the fall armyworm Spodoptera frugiperda, a new invasive pest in South China. Insect Sci. 29, 865–878 (2022).
Tabata, J. et al. Sex pheromone of the fall armyworm, Spodoptera frugiperda: Identification of a trace component that enhances attractiveness and specificity. Entomol. Exp. Appl. 171, 535–541 (2023).
Frérot, B. & Foster, S. P. Sex pheromone evidence for two distinct taxa within Graphania mutans (Walker). J. Chem. Ecol. 17, 2077–2093 (1991).
Wu, W. Q., Hansson, B. S. & Lofstedt, C. Electrophysiological and behavioural evidence for a fourth sex pheromone component in the turnip moth, Agrotis segetum. Physiol. Entomol. 20, 81–92 (1995).
Jing, X. et al. Identification and field evaluation of the female sex pheromone of the sand Salix carpenterworm, Holcocerus arenicola Staudinger (Lepidoptera: Cossidae). Zeitschrift fur Naturforsch. - Sect. C J. Biosci. 65, 403–411 (2010).
Wang, C. et al. Optimization of a pheromone lure by analyzing the peripheral coding of sex pheromones of Spodoptera frugiperda in China. Pest Manag. Sci. 78, 2995–3004 (2022).
Andrade, R., Rodriguez, C. & Oehlschlager, A. C. Optimization of a pheromone lure for Spodoptera frugiperda (Smith) in Central America. J. Braz. Chem. Soc. 11, 609–613 (2000).
Cruz-Esteban, S., Rojas, J. & Malo, E. Una feromona para la captura de machos del gusano cogollero (Lepidoptera: Noctuidae) en México. Acta Zool. Mex. 8445, 1–11 (2020).
Nagoshi, R. N., Meagher, R. L., Nuessly, G. & Hall, D. G. Effects of fall armyworm (Lepidoptera: Noctuidae) interstrain mating in wild populations. Environ. Entomol. 35, 561–568 (2006).
Otim, M. H. et al. Detection of sister-species in invasive populations of the fall armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae) from Uganda. PLoS One 13, 1–19 (2018).
Nagoshi, R. N. Evidence that a major subpopulation of fall armyworm found in the Western Hemisphere is rare or absent in Africa, which may limit the range of crops at risk of infestation. PLoS One 14, 1–19 (2019).
Zhang, L. et al. High-depth resequencing reveals hybrid population and insecticide resistance characteristics of fall armyworm (Spodoptera frugiperda) invading China. BioRxiv: 813154 (2019).
Prasanna, B. M. et al. Host plant resistance to fall armyworm. In Fall Armyworm in Africa: A Guide for Integrated Pest Management (Prasanna, B.M. et al. eds.). 1st ed. 45–62 (CIMMYT, 2018)
Sisay, B. et al. Responses of the fall armyworm (Spodoptera frugiperda) to different host plants: Implications for its management strategy. Pest Manag. Sci. 79, 845–856 (2023).
Hajibabaei, M., Janzen, D. H., Burns, J. M., Hallwachs, W. & Hebert, P. D. N. DNA barcodes distinguish species of tropical Lepidoptera. Proc. Natl. Acad. Sci. USA 103, 968–971 (2006).
Folmer, O. M., Black, W., Hoeh, R. & Lutz, R. V. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotechnol. 3, 294–299 (1994).
Kearse, M. et al. Geneious basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28, 1647–1649 (2012).
Kumar, S., Stecher, G., Li, M., Knyaz, C. & Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 35, 1547–1549 (2018).
Tamura, K. & Nei, M. Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol. Biol. Evol. 10, 512–526 (1993).
Adams, R. P. Identification of essential oil components by gas chromatography/mass spectroscopy. Biochem. Syst. Ecol. 24, 13 (1996).
Babushok, V. I. Chromatographic retention indices in identification of chemical compounds. TrAC Trends Anal. Chem. 69, 98–104 (2015).
Acknowledgements
The authors gratefully acknowledge the financial support for this research by the following organizations and agencies: European Union grant number DCI-FOOD/2018/402-634; Swedish Research Council grant registration number 2019-04267; the Swedish International Development Cooperation Agency (Sida); the Swiss Agency for Development and Cooperation (SDC); the Australian Centre for International Agricultural Research (ACIAR); the Norwegian Agency for Development Cooperation (Norad); the Federal Democratic Republic of Ethiopia; and the Government of the Republic of Kenya. The views expressed herein do not necessarily reflect the official opinion of the donors. We thank Levi Ombura, Basilio Njiru, Maureen Adhiambo, Peter Malusi and Faith Ndunge for technical assistance during the experiments.
Author information
Authors and Affiliations
Contributions
Conceptualisation, B.S., A.T. and S.S.; Methodology, B.S.; Data collection and Analysis, B.S.; Original draft preparation, B.S.; Review and Editing, A.T., S.S., B.T., C.W., K.K.G., F.K. and P.A.; Supervision, A.T., S.S., B.T. and C.W.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sisay, B., Tamiru, A., Subramanian, S. et al. Pheromonal variation and mating between two mitotypes of fall armyworm (Spodoptera frugiperda) in Africa. Sci Rep 14, 3848 (2024). https://doi.org/10.1038/s41598-024-53053-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-53053-9
- Springer Nature Limited