Abstract
SARS-CoV-2 and its variants are widely prevalent worldwide. With frequent secondary and breakthrough infections, immune dysfunction in RA patients, and long-term use of immune preparations, SARS-CoV-2 infection poses a significant challenge to patients and rheumatologists. Whether SARS-CoV-2 infection causes RA flares and what factors aggravate RA flares are poorly studied. A questionnaire survey was conducted on RA patients infected with SARS-CoV-2 after December 7, 2022, in China through a multicenter and inter-network platform regarding general personal condition, primary disease, comorbidity, SARS-CoV-2 vaccination, viral infection, and impact on the primary disease. A total of 306 RA patients were included in this study, and the patient data were analyzed, in which the general condition of RA patients, medication use before SARS-CoV-2 infection and post-infection typing and manifestations, and medication adjustment did not affect the Flare of RA patients after SARS-CoV-2 infection. The control of disease before SARS-CoV-2 infection (OR = 2.10), RA involving pulmonary lesions (OR = 2.28), and the recovery time of COVID-19 (OR = 2.50) were risk factors for RA flare. RA involving pulmonary lesions, control status of disease before infection, and recovery time of COVID-19 disease are risk factors for RA flare after SARS-CoV-2 infection.
Similar content being viewed by others
Significance & innovation section
RA patients have immune dysfunction, long-term use of immunosuppressive drugs, and low SARS-CoV-2 vaccination rate, which make them a vulnerable group during the COVID-19 pandemic.
Respiratory involvement, control status of disease before infection, and recovery time of COVID-19 disease are risk factors for RA flare after SARS-CoV-2 infection.
Controlling disease activity in RA patients, especially in the context of the SARS-CoV-2 pandemic, has positive implications.
Background
Rheumatoid arthritis (RA) is an autoimmune disease characterized by chronic erosive arthritis involving multiple organs. The pathological basis is synovitis and pannus formation, which can lead to joint deformity and loss of function1. The disease course of RA is prolonged, multiple organs are involved, and patients are treated with corticosteroids, disease-modifying anti-rheumatic drugs, biological agents, and other immunosuppressive drugs for a long time, which impair immune function and make them more susceptible to viral infection2,3.
More than three years into the global pandemic, COVID-19 has posed a severe threat to human health and life. The frequent secondary infections and breakthrough infections in RA patients have become significant threats to global public health. A highly effective vaccine against SARS-CoV-2 offers hope that people can reduce their infection or ease the symptoms of COVID-195,6. However, RA patients have the problems of poor vaccine response, delayed seroconversion, low conversion rate, and low effective inhibition effect7,8. In addition, the immunogenicity of the vaccine also has the possibility of causing the recurrence and aggravation of the original disease in RA patients. RA patients have not been included in the clinical development and phase III efficacy trials of some SARS-CoV-2 vaccines, and RA patients with active disease are not recommended to be vaccinated9.
RA patients are a vulnerable group during the COVID-19 pneumonia pandemic. Immunodeficiency and immunocompromised state caused by long-term use of corticosteroids or other immunosuppressive drugs are all high-risk factors for severe/critical COVID-1910. COVID-19 can lead to pneumonia, acute respiratory distress syndrome, renal failure, thrombotic complications, multi-organ failure, and death11,12. Multiple population-based or health system-based studies have shown that patients with RA are at increased risk of COVID-19 infection and are more likely to have adverse COVID-19 outcomes (a combined endpoint of hospitalization, mechanical support, or death)13. In addition, Rituximab is associated with an increased risk of COVID-19-related death and intensive care hospitalization or death. At the same time, whether glucocorticoids and Janus kinase (JAK) inhibitors can worsen the adverse outcomes of COVID-19 still needs to be further verified14.
With the emergence of Omicron variants with greater transmissibility, pathogenicity, and immune evasion, patients with RA face more significant crises and challenges. RA patients with immune dysfunction and long-term use of hormones and immunosuppressants may cause severe immune response and cytokine storm once infected with SARS-CoV-210,14. In addition, the use of antiviral drugs may have a further adverse effect on the patient's disease status15. Although patients with rheumatoid arthritis (RA) are considered to be at high risk for COVID-19 and prone to adverse outcomes, there is no consistent consensus on the impact of COVID-19 infection on the disease and how to use hormonal and immunosuppressive therapeutics during infection. To clarify the above issues, this study designed a questionnaire for RA patients to collect data. Through multicenter and network platforms, a questionnaire survey was conducted on the general situation, clinical data, SARS-CoV-2 vaccination, and disease changes after infection of RA patients. To identify factors associated with Flare in RA patients infected with SARS-CoV-2. This research is the first study in China to investigate the changes in the real-world condition of RA patients infected with SARS-CoV-2, which is of great significance for guiding the clinical treatment of RA patients. In particular, managing RA patients in the context of COVID-19 provides a reasonable basis for clinicians and healthcare providers.
Method
Ethical approval
The study was approved by the Ethics Committee of Shanxi Bethune Hospital, and informed consent was obtained from each patient.
Study design and participants
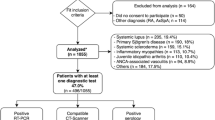
This study was a real-world survey in which information was collected from patients with RA first infected with SARS-CoV-2 in Shanxi Province and 27 other provinces in China between January and March 2023. The primary endpoint of this study was RA flare after infection with SARS-CoV-2. This study cites the consensus reached at OMERACT 10, where "RA flare" is defined as the worsening of signs and symptoms of sufficient intensity and duration to lead to a change in therapy. Meanwhile, the protocol also shows that Questionnaire survey is an effective method for assessing RA flares16. Patients with RA were recruited from rheumatology clinics and patient cohorts established by rheumatologists. All data collected were anonymized, original, valid, and assisted by rheumatologists. Patients who attended the outpatient department were provided with a paper-and-pencil questionnaire, which they completed under the guidance of rheumatologists, a process that typically took about 10 min. On the other hand, patients who took part in the online survey received a follow-up telephone consultation with rheumatologists, during which their condition was reassessed, and the provided information was confirmed. These measures ensure that the information we collect is true and reliable. The questionnaire involved 34 items, including demographic characteristics, clinical characteristics of RA, treatment, disease activity assessment, COVID-19 vaccination, COVID-19 infection, and treatment. For comprehensive details regarding the questionnaire, kindly refer to the appendix.
Inclusion criteria
Patients were aged 18-80 years and diagnosed by rheumatologists according to the 2010 ACR/EULAR criteria.
Patients with the first infected by SARS-Cov-2
Exclusion criteria
Questionaries with un-reasonable data
Questionaries with incomplete completion
The flow diagram is shown in flow Fig. 1.
Outcomes
All outcomes and assignments were assessed by rheumatologists and reported by the patients. RA patients without apparent joint swelling and pain and typical inflammatory markers before COVID-19 infection were defined as the stable disease group, and the rest were the active disease group. According to the organ involvement of RA patients, they were divided into joint involvement, pulmonary lesions, and other system involvement. A total of 430 questionnaires were collected, of which 115 RA patients showed no signs of first SARS-CoV-2 infection, and 9 patients with abnormal data were excluded. This study included 306 patients with RA, including 242 outpatients and 64 online patients.
Statistical Analysis SPSS 22.0 software was used for statistical analysis. Continuous variables with normal distributions are presented as means and standard deviations. Continuous variables with abnormal distributions are continuous variables that were, respectively, presented as medians and interquartile ranges (IQRs). Baseline normal and abnormal quantitative data were analyzed using the student T-test and Wilcoxon rank-sum test. Categorical variables at baseline were analyzed using the chi-square test. Multivariate logistic regression analysis was performed for the outcomes analysis to identify the associations between dependent and independent variables. The results are expressed as adjusted odds ratios (ORs) with 95% confidence intervals (95% CIs). Significance was accepted as a two-sided test with an alpha level of 0.05.
Ethical approval and consent to participate
This study was approved by the Ethics Review Committee of Shanxi Bethune Hospital and was conducted by relevant guidelines/regulations; Confirm that informed consent has been obtained from all participants and their legal guardians. And conducted research in compliance with the Declaration of Helsinki.
Results
Demographic and clinical data
Among the 306 patients, 262 were female, and 44 were male, with an average age of 51.44 ± 13.06 years. The disease duration ranged from 3 months to 32 years, averaging 115.67 ± 104.66 months. RA patients' affected organs and pre-infection medications (antiviral drugs, non-steroidal anti-inflammatory drugs, hormones, JAK inhibitors) were also described in accurate stratification (see Table 1 for details).
The classification of the severity of COVID-19 is determined based on an evaluation of the patient's symptoms, clinical manifestations, and imaging of lung lesions, categorized as asymptomatic, mild, moderate, severe and critical. Due to the small number of moderate, severe, and critical RA patients included in this study, they were combined as moderate and severe. It is worth noting that nearly a quarter of RA patients are unvaccinated against SARS-CoV-2. Time to recovery from COVID-19 in RA patients (symptom resolution/negative nucleic acid test) and medication for COVID-19 treatment were also described (see Table 1 for details).
Univariate analysis
Univariate analysis showed that the general conditions of RA patients (age, gender, BMI, smoking and drinking history, and whether they were complicated with other underlying diseases) and the clinical data of RA patients (course of disease, drug use) did not affect the flare after infection with SARS-CoV-2 (see Table 2 for details). The involvement of different organs in RA patients, especially the RA involving pulmonary lesions (OR = 2.26, 95% CI: 1.13–4.53), could aggravate the risk of Flare in RA patients infected with SARS-CoV-2.RA patients with poor disease control before SARS-CoV-2 infection (OR = 2.16, 95% CI: 1.30–3.61) and RA patients with prolonged recovery time from COVID-19 (OR = 2.35, 95% CI: 1.34–410) were more likely to have RA flare. Some RA patients were treated with oxygen therapy during hospitalization for COVID-19, and these patients generally had flare or aggravation (OR = 2.72, 95% CI: 1.13–6.67).
Multivariate logistic regression model
In the multivariate logistic regression model, RA involving pulmonary lesions (OR = 2.28, 95% CI: 1.09-4.79), disease control before infection (OR = 2.10, 95% CI: 1.23-3.58), and recovery time from COVID-19 (OR = 2.50, 95% CI: 1.09-4.79) were risk factors for flare in RA patients infected with SARS-CoV-2 (see Table 2 for details).
Discussion
Basic information on RA patients
In this Internet-based Questionnaire survey of RA patients, we found that the general conditions of RA patients, such as age, gender, alcohol/alcohol history, BMI, and medication status, were not associated with the disease changes of RA patients after SARS-CoV-2 infection. RA patients are more likely to have flares when their organs are involved, especially when RA involves pulmonary lesions. Similarly, among RA patients hospitalized for COVID-19, oxygen therapy was also a factor for flare in univariate analysis. The excessive activation of the immune system and the involvement of cytokines in destroying an alveolar structure are the key factors for severe lung disease in COVID-19 patients. Coronaviruses use membrane ACE2 receptors to enter human cells. At the same time, ACE2 is also highly expressed in lung cells of patients with RA-related interstitial lung disease (ILD), and patients with RA and ILD are more susceptible to high inflammation and acute lung injury than the general population17,18. In addition, RA-ILD and COVID-19-infected lung lesions have remarkable similarities in epidemiological, clinical, and immunological characteristics. This increases the difficulty in disease judgment, medication, and treatment of RA patients with lung involvement infected with SARS-CoV-2 and the risk of Flare in RA patients19. Some RA patients have multiple system involvement. Model 2 was established according to the system involvement, and RA patients were divided into joint involvement only, one organ involvement, and two or more organ involvement. The involvement of 2 or more organs was found to be a risk factor for RA flare, as detailed in the appendices.
Drugs for the treatment of RA
The study found that the drugs used by RA patients before COVID-19 infection, such as hormones, DMARD, biological agents, and non-steroidal anti-inflammatory drugs, were not associated with flare. This is similar to the results of the current study. As a PRO derived from the real world, it verifies the safety of traditional synthetic DMARDs and biologic DMARDs in RA patients during ACOVID-19, which has important guiding significance for the clinical medication of RA patients20. Similar recommendations from the American College of Rheumatology and the European League Against Rheumatism in 2021 do not recommend abrupt discontinuation of corticosteroids, regardless of SARS-CoV-2 exposure, and patients should be maintained at a lower dose. For patients with asymptomatic or mild COVID-19 and relatively stable autoimmune disease, it is recommended to withhold immunosuppressive agents, biological agents (except tocilizumab), and JAK inhibitors (current evidence is controversial) until 7–14 days after COVID-19 symptom relief or 10–17 days from the date of nucleic acid positive21,22. For patients with severe or even life-threatening autoimmune diseases, hormones can be used based on weighing the pros and cons, and biological agents or JAK inhibitors can be initiated 23.
Baricitinib is an effective treatment for patients with COVID-19 pneumonia. By inhibiting the JAK1 and JAK2 pathways, it can block the immune cascade and reduce viral replication, which is beneficial for reducing respiratory failure and mechanical ventilation. It can also prevent the worsening of symptoms associated with COVID-1924,25. It has also been reported that using Baricitinib can increase the risk of critical illness of COVID-19 in RA patients. As a potent immunosuppressant, Baricitinib cannot distinguish between infected host cells and healthy cells and has an inhibitory effect on the immune response of patients during the treatment 26. In particular, RA patients have autoimmune dysfunction, and the use of JAK inhibitors (compared with TNF-α inhibitors) will increase the risk of infection, major adverse cardiovascular events (MACE), and thrombosis in RA patients27. Although JAK inhibitors have been widely used to treat COVID-19, the risk of preexisting Flare after SARS-CoV-2 infection in RA patients who use JAK inhibitors should also be vigilant. So. Clinicians should carefully consider the appropriate timing of JAK inhibitor administration in RA patients with COVID-19 pneumonia.
Vaccination of patients with RA
Unvaccinated RA patients had a similar risk for RA flare after SARS-CoV-2 infection compared with patients with RA who were vaccinated against SARS-CoV-2. With existing studies showing that vaccination reduces symptoms of COVID-19, Kawano Y et al. reviewed outcomes in patients with Autoimmune rheumatic disease (ARD) who were infected with SARS-CoV-2 over the past years and found that the proportion of patients with ARD who had severe COVID-19 has decreased since the early days of the pandemic. However, unvaccinated ARD patients have more severe cases28. There is also evidence that the neutralizing titer of antibodies against the standard variant of S protein (D614G) in RA patients after vaccination is lower than that in the general population, and the effectiveness of vaccine prevention and the reduction of hospitalization is also lower29,30,31. Despite breakthrough infections in RA patients and the greater escape transmissibility of Micron and its variants, recent evidence suggests that immunity from vaccination and infection can reduce Omicron infectivity. Infectivity decreased markedly after breakthrough infection to 28% in vaccinated versus 36% in unvaccinated participants. The booster dose also reduces infectivity further, with an 11% reduction in the likelihood of transmission for each additional amount32.
Another concern, however, is that more than a quarter of RA patients have never been vaccinated because of disease activity or concerns about the safety of the SARS-CoV-2 vaccine, posing a potential risk for changes in patient condition. Existing studies have shown that vaccination of RA patients in remission or with low disease activity has no significant effect on disease activity or exacerbation. However, the COVID-19 vaccination rate in RA patients is still low33,34,35. The ACR has continuously issued updated guidelines for vaccinating RA patients, advocating that patients in stable condition receive the SARS-CoV-2 vaccine (optimal choice) and a booster dose. Vaccination is recommended for people at high risk as soon as possible, regardless of disease activity and severity (except in critically ill patients). For RA patients, the safety of inactivated vaccines (the safest), recombinant subunit vaccines, or mRNA vaccines is relatively high36. Therefore, clinicians should actively recommend SARS-CoV-2 vaccination in stable RA patients, and pre-exposure prophylaxis is an essential treatment for RA patients.
Patients with RA and regular visits
RA patients have irregular return visits and self-withdrawal of drugs during COVID-19. The non-standard diagnosis and treatment methods aggravate the risk of Flare in RA patients to a certain extent. During the period of SARS-CoV-2 infection, one-third of RA patients reduced or discontinued immunosuppressive agents, and one-sixth of RA patients suspended or reduced the use of biological agents and small molecule targeted drugs. More than half of the patients did not have regular follow-up visits, and some patients adjusted their treatment drugs. Similarly, Adrian Ciurea et al. investigated the course management of RA patients in Switzerland during the COVID-19 pandemic. They found that the number of patient consultations decreased by 52%, and the proportion of drug non-adherence also increased by37. Regular medical visits and outpatient treatment (with antiviral drugs or monoclonal antibodies to treat SARS-CoV-2) can reduce the incidence of severe COVID-19 outcomes in ARD patients38. Clinicians should pay attention to the impact of COVID-19 on the medical treatment of RA patients, and more convenient medical methods such as online diagnosis and treatment and telephone follow-up can be carried out to ensure the medical needs of RA patients.
Medical insurance has a long-term significance for the disease control of RA patients. Zara Izadi et al. used the COVID-19 Global Alliance for Rheumatology (GRA) registry to extract data to observe global differences in COVID-19 pneumonia outcomes; It was found that social policies and resources (environmental pollution, national economy, government policies, health needs) had a large impact on COVID-19 outcomes of ARD patients globally39. Similarly, data from populations have found that the COVID-19 pandemic has disproportionately affected racial and ethnic minority groups, with higher mortality rates in African-American, Native American, and Latino communities40. Adequate medical insurance can reduce the risk of adverse outcomes in RA patients with COVID-19, and RA patients should actively seek medical care after being infected with SARS-CoV-2. Active management of SARS-CoV-2 exposure in RA patients should be based on known risk factors for poor prognoses, such as age > 65 years, comorbidities, and the degree of immunosuppression41.
RA and COVID-19
Studies have found that RA patients with a long recovery time from COVID-19 are more likely to have RA disease exacerbation. The type and treatment of COVID-19 did not, which may also be related to the small number of critically ill cases of COVID-19 that were included in the study. COVID-19 and RA are highly similar in clinical manifestations (fever, myalgia, arthralgia, fatigue, etc.), pathogenesis, and treatment drugs. The complications, severity, and mortality of COVID-19 patients are caused by excessive activation of cytokines (IL-1, IL-6, TNF-a, CCL2, and CXCL10, etc.), namely cytokine storm, which is remarkably similar to the pathogenesis of immune disorders in RA patients42,43. In addition, cases of musculoskeletal manifestations due to COVID-19 immune disorders cannot be clearly distinguished from joint and/or muscle dysmenorrhea in RA patients.
RA disease and COVID-19 are mutual risk factors. SARS-CoV-2 induces a high inflammatory response of "cytokine storm" in RA patients through pleocytosis. Immune disorders and long-term use of hormones and immunosuppressants in RA patients are independent risk factors for SARS-CoV-2 infection and acute hospitalization of COVID-1944,45. In an earlier study from Wuhan, China, patients with ARD had an unusually high relative risk for SARS-CoV-2 infection compared with the general population (OR 10.90; 95% CI 5.43–21.89)46. Similarly, in the whole family, the infection rate was much higher in RA patients than in other family members (OR 2.68; 95% CI 1.14–6.27). ARD patients who also took hydroxychloroquine had a lower risk of COVID-19 than those who took other disease-modifying anti-rheumatic drugs (OR 01.0; 94% CI 0.044–19.1; p = 04.95)47. In addition to a higher risk of infection, RA patients have also been found to have a higher rate of hospitalization and risk of critical illness. National cohort studies in several countries have shown that ARD patients hospitalized within 30 days of COVID-19 diagnosis have a higher risk of ICU admission, acute renal failure requiring renal replacement therapy, and venous thromboembolism, as well as higher mortality compared with the general population48,49.
We also found that RA patients generally had poor sleep and psychological anxiety during COVID-19 pneumonia. Similarly, Dominic L. Sykes studied the lasting effects of COVID-19 on patients with ARD and found that long-term symptoms, including anxiety (p = 0.001), fatigue (p = 0.004), myalgia (p = 0.022), were common in patients with RA. In particular, women are more likely to have long-term residual symptoms50.
The symptoms of COVID-19 and RA are highly similar. SARS-CoV-2 infection with cytokine storm and immune system disorders increases RA patients' risk of disease activity. This study shows that involving pulmonary lesions, preinfectional disease control, and time to recovery from COVID-19 are risk factors for Flare in RA patients with SARS-CoV-2 infection. At present, hormones and anti-rheumatic drugs such as hydroxychloroquine, tocilizumab, and Baricitinib are used as the main treatment for COVID-19 infection. Clinicians should make the optimal choice by comprehensive evaluation when using drugs to treat COVID-19 and control RA disease activity.
However, there are still some limitations of our study, and recall bias is difficult to avoid by using the questionnaire to collect information. Due to the limitation of the people who filled in the questionnaires, the number of critical RA patients with COVID-19 was relatively small, and the death of RA patients due to COVID-19 was not included in this questionnaire. In the next step, we will use the national medical insurance system to collect data, taking the activity of the primary disease, organ involvement, and the use of hormones, immunosuppressants, and anticoagulants in RA patients in the year before and after December 2022 as the node (December 2022 in China can be regarded as not infected with SARS-CoV-2). Clinical and laboratory data were collected and analyzed. Various treatment methods' serological characteristics, efficacy, and safety were studied. The critical condition, mortality, and hospitalization expenses of RA patients were compared horizontally, and the short and long-term conditions of RA patients after COVID-19 infection were evaluated to provide more comprehensive guidance for the diagnosis and treatment of RA patients.
Conclusion
Patients with RA are involved in lung lesions, disease control status before infection, and recovery time from COVID-19 disease, which are considered risk factors for RA outbreaks following SARS-CoV-2 infection.
Consent for publication
The consent of the owner has been obtained and published.
Data availability
All raw data can be obtained by contacting the corresponding author.
References
Khasnis, A. A., Schoen, R. T. & Calabrese, L. H. Emerging viral infections in rheumatic diseases. Semin. Arthritis Rheum. 41(2), 236–246. https://doi.org/10.1016/j.semarthrit.2011.01.008 (2011).
Yousefghahari, B. et al. Risk of COVID-19 infection in patients with rheumatic disease taking disease-modifying anti-rheumatic drugs. Clin. Rheumatol. 40(10), 4309–4315. https://doi.org/10.1007/s10067-021-05779-4 (2021).
Louthrenoo, W. Treatment considerations in patients with concomitant viral infection and autoimmune rheumatic diseases. Best Pr. Res. Clin. Rheumatol. 29(2), 319–342. https://doi.org/10.1016/j.berh.2015.05.010 (2015).
Carabelli, A. M., Peacock, T. P., Thorne, L. G., et al. SARS-CoV-2 variant biology: immune escape, transmission and fitness. Nat. Rev. Microbiol. Published online January 18 (2023). https://doi.org/10.1038/s41579-022-00841-7
Yıldırım, R., Üsküdar Cansu, D., Dinler, M., Korkmaz, C. Reactivation in major organ involvement following SARS-CoV-2 mRNA vaccination in Behçet's syndrome patient receiving immunosuppressive therapy. Rheumatology. 2022;61(SI2):SI197-SI199. https://doi.org/10.1093/rheumatology/keac246
Chalkias, S. et al. A bivalent omicron-containing booster vaccine against Covid-19. N. Engl. J. Med. 387(14), 1279–1291. https://doi.org/10.1056/NEJMoa2208343 (2022).
Deepak, P., Kim, W., Paley, M.A., et al. Glucocorticoids and B cell depleting agents substantially impair immunogenicity of MRNA vaccines to SARS-CoV-2. Infect. Dis. (except HIV/AIDS) (2021). https://doi.org/10.1101/2021.04.05.21254656
Sattui, S. E. et al. Early experience of COVID-19 vaccination in adults with systemic rheumatic diseases: results from the COVID-19 global rheumatology alliance vaccine survey. RMD Open. 7(3), e001814. https://doi.org/10.1136/rmdopen-2021-001814 (2021).
Voysey, M. et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomized controlled trials in Brazil, South Africa, and the UK. The Lancet. 397(10269), 99–111. https://doi.org/10.1016/S0140-6736(20)32661-1 (2021).
Brodin, P. Immune determinants of COVID-19 disease presentation and severity. Nat. Med. 27(1), 28–33. https://doi.org/10.1038/s41591-020-01202-8 (2021).
Chen, R. et al. Longitudinal hematologic and immunologic variations associated with the progression of COVID-19 patients in China. J. Allergy Clin. Immunol. 146(1), 89–100. https://doi.org/10.1016/j.jaci.2020.05.003 (2020).
Lucas, C. et al. Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature. 584(7821), 463–469. https://doi.org/10.1038/s41586-020-2588-y (2020).
Grainger, R., Kim, A. H. J., Conway, R., Yazdany, J. & Robinson, P. C. COVID-19 in people with rheumatic diseases: risks, outcomes, treatment considerations. Nat. Rev. Rheumatol. 18(4), 191–204. https://doi.org/10.1038/s41584-022-00755-x (2022).
MacKenna, B. et al. Risk of severe COVID-19 outcomes associated with immune-mediated inflammatory diseases and immune-modifying therapies: a nationwide cohort study in the OpenSAFELY platform. The Lancet Rheumatol. 4(7), e490–e506. https://doi.org/10.1016/S2665-9913(22)00098-4 (2022).
Zur, M., Peselev, T., Yanko, S. et al. Efficacy and safety of antiviral treatments for symptomatic COVID-19 outpatients: Systematic review and network meta-analysis. Antiviral Res. 221: 105768 (2023).
Rieke, A., Christof, P., Choy Ernest H., et al. Developing a construct to evaluate flares in rheumatoid arthritis: a conceptual report of the OMERACT RA Flare Definition Working Group. J. Rheumatol. 38: 1745–50 (2011). https://doi.org/10.3899/jrheum.110400
Williamson, J., Black, L., Black, A., Koduri, G. & Kelly, C. There are similarities between rheumatic disease with lung involvement and COVID-19 pneumonia. Ir. J. Med. Sci. 191(1), 1–5. https://doi.org/10.1007/s11845-021-02545-y (2022).
Zaccardelli, A., Wallace, Z. S. & Sparks, J. A. Acute and post-acute COVID-19 outcomes for patients with rheumatoid arthritis: lessons learned and emerging directions 3 years into the pandemic. Curr. Op. Rheumatol. 35(3), 175–184. https://doi.org/10.1097/BOR.0000000000000930 (2023).
Raghu, G. & Wilson, K. C. COVID-19 interstitial pneumonia: monitoring the clinical course in survivors. The Lancet Respirat. Med. 8(9), 839–842. https://doi.org/10.1016/S2213-2600(20)30349-0 (2020).
Costantino, F. et al. COVID-19 in French patients with chronic inflammatory rheumatic diseases: Clinical features, risk factors and treatment adherence. Joint Bone Spine. 88(1), 105095. https://doi.org/10.1016/j.jbspin.2020.105095 (2021).
Mikuls, T.R., Johnson, S.R., Fraenkel, L., et al. American college of rheumatology guidance for the management of rheumatic disease in adult patients during the COVID‐19 pandemic: Version 3. Arthr. Rheumatol. 73(2). https://doi.org/10.1002/art.41596 (2021).
Landewé, R.B.M., Kroon, F.P.B., Alunno, A., et al. EULAR recommendations for the management and vaccination of people with rheumatic and musculoskeletal diseases in the context of SARS-CoV-2: the November 2021 update. Ann Rheum Dis. Published online February 23 annrheumdis-2021–222006 (2022). https://doi.org/10.1136/annrheumdis-2021-222006
Alunno, A. et al. 2021 update of the EULAR points to consider on the use of immunomodulatory therapies in COVID-19. Ann. Rheum. Dis. 81(1), 34–40. https://doi.org/10.1136/annrheumdis-2021-221366 (2022).
Kalil, A. C. et al. Baricitinib plus remdesivir for hospitalized adults with Covid-19. N. Engl. J. Med. 384(9), 795–807. https://doi.org/10.1056/NEJMoa2031994 (2021).
Goletti, D. & Cantini, F. Baricitinib therapy in Covid-19 pneumonia—an unmet need fulfilled. N. Engl. J. Med. 384(9), 867–869. https://doi.org/10.1056/NEJMe2034982 (2021).
Bahari, Z., Jangravi, Z., Ghoshooni, H., Afarinesh, M. R. & Meftahi, G. H. Pharmacological mechanism of immunomodulatory agents for the treatment of severe cases of COVID-19 infection. Inflamm. Res. 70(4), 389–405. https://doi.org/10.1007/s00011-021-01445-2 (2021).
Benucci, M. et al. Cardiovascular safety, cancer and Jak-inhibitors: Differences to be highlighted. Pharmacol. Res. 183, 106359. https://doi.org/10.1016/j.phrs.2022.106359 (2022).
Kawano, Y., Patel, N.J., Wang, X., et al. Temporal trends in COVID-19 outcomes among patients with systemic autoimmune rheumatic diseases: from the first wave through the initial Omicron wave. Ann. Rheum. Dis. Published online August 9 (2022). ard-2022–222954. https://doi.org/10.1136/ard-2022-222954
Bakasis, A. D. et al. COVID-19 infection among autoimmune rheumatic disease patients: Data from an observational study and literature review. J. Autoimmun. 123, 102687. https://doi.org/10.1016/j.jaut.2021.102687 (2021).
Galmiche, S. et al. Immunological and clinical efficacy of COVID-19 vaccines in immunocompromised populations: a systematic review. Clin. Microbiol. Infect. 28(2), 163–177. https://doi.org/10.1016/j.cmi.2021.09.036 (2022).
Furer, V. et al. Immunogenicity and safety of the BNT162b2 mRNA COVID-19 vaccine in adult patients with autoimmune inflammatory rheumatic diseases and in the general population: a multicentre study. Ann. Rheum. Dis. 80(10), 1330–1338. https://doi.org/10.1136/annrheumdis-2021-220647 (2021).
Tan, S.T., Kwan, A.T., Rodríguez-Barraquer, I., et al. Infectiousness of SARS-CoV-2 breakthrough infections and reinfections during the Omicron wave. Nat. Med. Published online January 2, 2023. https://doi.org/10.1038/s41591-022-02138-x
Tedeschi, S. K. et al. Rheumatoid arthritis disease activity assessed by patient-reported outcomes and flow cytometry before and after an additional dose of COVID-19 vaccine. Ann. Rheum. Dis. 81(7), 1045–1048. https://doi.org/10.1136/annrheumdis-2022-222232 (2022).
Geng, Y., Fan, Y., Wang, Y., et al. Flare and change in disease activity among patients with stable rheumatoid arthritis following coronavirus disease 2019 vaccination: a prospective Chinese cohort study. Chin. Med. J. 2023; Publish Ahead of Print. https://doi.org/10.1097/CM9.0000000000002562
Chen, J. et al. The COVID-19 vaccine: Attitudes and vaccination in patients with autoimmune inflammatory rheumatic diseases. Rheumatol. Autoimmun. 2(2), 82–91. https://doi.org/10.1002/rai2.12028 (2022).
Prontskus, V. et al. COVID -19 vaccination and the incidence of de novo or recurrent rheumatoid arthritis: A French and international (VIGIBASE) signal detection study. Clin. Pharma. Therapeutics. 113(5), 1107–1116. https://doi.org/10.1002/cpt.2866 (2023).
Curtis, J.R., Johnson, S.R., Anthony, D.D., et al. American college of rheumatology guidance for COVID ‐19 vaccination in patients with rheumatic and musculoskeletal diseases: Version 4. Arthr. Rheumatol. 74(5) (2022). https://doi.org/10.1002/art.42109
Ciurea, A. et al. Impact of the COVID-19 pandemic on the disease course of patients with inflammatory rheumatic diseases: results from the Swiss Clinical Quality Management cohort. Ann. Rheum. Dis. 80(2), 238–241. https://doi.org/10.1136/annrheumdis-2020-218705 (2021).
Qian, G., Wang, X., Patel, N.J., et al. Outcomes with and without outpatient SARS-CoV-2 treatment for patients with COVID-19 and systemic autoimmune rheumatic diseases: a retrospective cohort study. The Lancet Rheumatol. Published online January 2023: S2665991323000061. https://doi.org/10.1016/S2665-9913(23)00006-1
Izadi, Z., Gianfrancesco, M.A., Schmajuk, G. et al. Environmental and societal factors associated with COVID-19-related death in people with rheumatic disease: an observational study. Lancet Rheumatol. 4: e603-e613 (2022).
Morales, D. R. & Ali, S. N. COVID-19 and disparities affecting ethnic minorities. The Lancet. 397(10286), 1684–1685. https://doi.org/10.1016/S0140-6736(21)00949-1 (2021).
Rodriguez-Miguelez, P., Heefner, A. & Carbone, S. Recognizing risk factors associated with poor outcomes among patients with COVID-19. Progr. Cardiovas. Dis. 76, 3–11. https://doi.org/10.1016/j.pcad.2023.01.006 (2023).
Kim, J. S. et al. Immunopathogenesis and treatment of cytokine storm in COVID-19. Theranostics. 11(1), 316–329. https://doi.org/10.7150/thno.49713 (2021).
Dey, A., Vaishak, K., Deka, D., et al. Epigenetic perspectives associated with COVID-19 infection and related cytokine storm: an updated review. Infection. Published online March 12, 2023. https://doi.org/10.1007/s15010-023-02017-8
Hasseli, R. et al. Older age, comorbidity, glucocorticoid use and disease activity are risk factors for COVID-19 hospitalisation in patients with inflammatory rheumatic and musculoskeletal diseases. RMD Open. 7(1), e001464. https://doi.org/10.1136/rmdopen-2020-001464 (2021).
Chen, M., Wei, Y., Zhang, Q., Wan, Q., Chen, X. Epidemiology and clinical characteristics of COVID-19 in rheumatic diseases at a tertiary care hospital in Wuhan, China. Clin. Exp. Rheumatol. 39(2):442–443 (2021). https://doi.org/10.55563/clinexprheumatol/13xbku
Zhong, J. et al. COVID-19 in patients with rheumatic disease in Hubei province, China: a multicentre retrospective observational study. The Lancet Rheumatol. 2(9), e557–e564. https://doi.org/10.1016/S2665-9913(20)30227-7 (2020).
D’Silva, K. M. et al. COVID-19 outcomes in patients with systemic autoimmune rheumatic diseases compared to the general population: A US multicenter. Compar. Cohort Study. Arthr. Rheumatol. 73(6), 914–920. https://doi.org/10.1002/art.41619 (2021).
Cordtz, R., Lindhardsen, J., Soussi, B.G., et al. Incidence and severeness of COVID-19 hospitalization in patients with inflammatory rheumatic disease: a nationwide cohort study from Denmark. Rheumatology. 60(SI):SI59-SI67 (2021). https://doi.org/10.1093/rheumatology/keaa897
Gica, Ş, Akkubak, Y., Aksoy, Z. K., Küçük, A. & Cüre, E. Effects of the COVID-19 pandemic on psychology and disease activity in patients with ankylosing spondylitis and rheumatoid arthritis. Turk. J. Med. Sci. 51(4), 1631–1639. https://doi.org/10.3906/sag-2011-188 (2021).
Acknowledgements
We thank the RA patients for their willingness to participate in our survey.
Funding
This work was supported by the 2023 COVID-19 Emergency Project of Shanxi Bethune Hospital (No. 2023xg03), the 2023 COVID-19 Emergency Project of Shanxi Health Commission (No. 2023XG002), and the Shanxi graduate education innovation project(No. 2023SJ137).
Author information
Authors and Affiliations
Contributions
L.R. designed the questionnaire, collected the information, and wrote the manuscript, Z.J.K. was responsible for the statistical analysis of the data; L.Q. and Z.L. drew the table. S.Y.Z. revised some content of the article; Z.J.Y. made important suggestions on the design of the questionnaire and the writing of the article. L.Y.Z. proposed the overall study concept and reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, R., Zhao, JK., Li, Q. et al. Analysis of related factors for RA flares after SARS-CoV-2 infection: a retrospective study from patient survey. Sci Rep 14, 4243 (2024). https://doi.org/10.1038/s41598-024-52748-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-52748-3
- Springer Nature Limited