Abstract
The short periods of incubation during egg storage (SPIDES) method enhances the quality of chicks and improves hatching rates. Additionally, embryonic thermal conditioning (TC) is a technique used to enhance thermotolerance in birds. Previous studies have evaluated the effects of SPIDES and embryonic TC separately. Yet, our hypothesis postulated that a synergistic effect could be achieved by integrating TC and SPIDES, thereby enhancing the broilers' resilience to thermal stress. We conducted an experiment involving 800 Ross broiler eggs, divided into two groups. The first group, referred to as S0, was maintained under standard storage room conditions and acted as our control group. The second group, known as S1, underwent a process called SPIDES for 5 h at a temperature of 37.8 ± 0.1 °C, on three occasions: days 5, 10, and 15 following egg collection. Upon reaching the 14th day of incubation (DOI), each of these primary groups was randomly subdivided into two equal subgroups. The control subgroup, designated as TC0, remained in the usual incubation conditions. Meanwhile, the other subgroup, TC1, was subjected to prenatal heat conditioning at a temperature of 39.5 ± 0.1 °C for 6 h per day, commencing on the 14th embryonic day (E) and extending until the 18th embryonic day (E). This experimental setup resulted in four distinct experimental subgroups: S0TC0, S1TC0, S0TC1, and S1TC1. The findings indicated that the combined application of SPIDES and TC had a significant positive effect on chick performance after hatching. Specifically, the (S1TC1) group exhibited the heaviest live body weight (LBW) and body weight gain (BWG) at the marketing age in comparison to the other groups. Furthermore, both SPIDES and TC had a positive influence on the relative weights of breast muscles and their histological measurements. The (S1TC1) group displayed significantly higher values in terms of the relative weight of breast muscles and the number of myocytes. In conclusion, SPIDES and TC have beneficial effects on pre- and post-hatch characteristics of broiler chicks up until the marketing age. Additionally, TC techniques improve chick performance, particularly under conditions of heat stress, and enhance the yield of breast muscle in later stages of life.
Similar content being viewed by others
Introduction
Storing eggs is a widely adopted procedure in farm and hatchery operations around the globe1. However, it has been found that extended periods of egg storage, even under optimal conditions, have negative consequences for egg quality, ultimately impacting the quality and performance of the resulting chicks2,3,4.
A natural method known as SPIDES has been derived from the behavior of chickens, where eggs that have been laid for some time are warmed up for one or more hours and then cooled down for 24 h until the next egg is laid. This process is repeated daily until the entire clutch is complete, and all eggs are incubated together starting on the same day for synchronized hatching5. Recently, the application of SPIDES has been shown to modify the conventional technique used in hatcheries6,7. This modification aims to improve egg quality following extended periods of storage, leading to enhanced chick quality and improved performance in the future8.
Climate change presents a significant global challenge, disrupting weather patterns and posing a threat to poultry production. Consequently, thermal stress has become a crucial limiting factor, directly affecting the well-being of birds9. Particularly, the broiler industry faces escalating challenges caused by heat stress (HS) due to the rising temperatures associated with global climate change10. To counteract the adverse effects of heat challenges, extensive efforts have been devoted to devising various approaches11,12. The effects of rising summer temperatures on poultry production are becoming increasingly evident as global warming escalates. Thus, it becomes essential to explore methods that can alleviate heat stress in chickens13. One such approach involves implementing thermal conditioning during the neonatal period of chickens, which has shown promise in enhancing their ability to tolerate high temperatures and reducing the subsequent rise in body temperature when exposed to elevated surrounding temperatures during their later stages of life14,15
Earlier research has primarily concentrated on studying the separate impacts of SPIDES or embryonic TC (TC). Surprisingly, no prior investigations have explored the joint influence of TC and SPIDES. Based on this gap, we developed a theory that the combination of TC and SPIDES could potentially improve broilers' resilience to heat stress. To test our hypothesis, we conducted a comprehensive assessment of productive performance, certain physiological responses, as well as histological and cytological observations of skeletal muscles in broiler chickens exposed to heat stress challenges.
Material and methods
Ethical statement
The authors confirm that the ethical policies of the journal, as noted on the journal's author guidelines page, have been adhered to and The experimental design and all the research protocols were approved by the Experimental Animal Care and Research Ethics Committee of Ain Shams University, Agriculture Sector Committee with ethical approval code (5-2023-8). The study was conducted following ARRIVE guidelines.
Experimental design
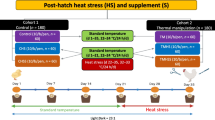
A total of eight hundred Ross breeder eggs, aged 29 weeks, averaging a weight of 63.04 ± 0.58 g, were divided into two groups of equal size based on the storage time. The first group, S0 (control), was kept under specific conditions at a temperature of 15.8°C ± 0.4 and relative humidity (RH) of 75% for a period of up to 20 days. The second group, labeled as S1, experienced the SPIDES method on days 5, 10, and 15 following egg collection, as described by8,16. After the 20-day storage period, all eggs were transferred to an automatic setter provided by a local manufacturer, where they were subjected to an incubation protocol consisting of 37.6 ± 0.02 °C and 60.5% of RH. On the 14th day of the incubation process, we randomly divided both groups (S0 and S1) into two equal subgroups. The control subgroup (TC0) was kept under optimal conditions, while the other subgroup (TC1) was exposed to prenatal thermal conditioning at a temperature of 39.5 ± 0.1 °C and RH of 72%. This thermal conditioning was maintained consistently for 6 h per day, from 10:00 a.m. to 4:00 p.m., over a period of five consecutive embryonic days (E14–E18). As a result, four experimental sub-groups were formed, namely S0TC0, S1TC0, S0TC1, and S1TC1 as reported by8.
Twenty eggs were chosen at random from each of the two main groups for the purpose of measuring egg quality on the 20th day after egg collection. We measured the egg weight with a highly precise electronic balance, which had an accuracy of 0.01g. To determine the egg's shape index (SI), we applied the formulas presented by17,18, which involve dividing the egg's width by its length and then multiplying the result by 100.
To measure the yolk weight, The yolk was separated from the albumen and quantified using electronic scales. The yolk weight percentage was calculated by dividing the yolk weight by the egg weight and then multiplying the outcome by 100.
The yolk index (YI), invented by19, was computed using the measurements of yolk height and yolk diameter with the formula:
We calculated the albumen weight by subtracting the combined weight of the yolk and shell from the total egg weight. Subsequently, the albumen weight percentage was obtained by dividing the albumen weight by the egg weight and multiplying the quotient by 100.
The albumin index was computed as the division of the albumin height by the average albumin width.
The pH of the albumen was promptly assessed using a pH meter that had been calibrated (H12212 pH Meter, HANNA Instruments).
The albumen height was converted into Haugh units (HU) to assess the freshness of the eggs, following the formula established by20 and modified by21
Egg weight loss percentage was calculated during the storage period (from day 3 to day 7, day 7 to day 15, and day 15 to day 20) and during the incubation period, including the first and second weeks of incubation, as well as during the thermal conditioning period (from embryonic day 14 to 18). This was determined by subtracting the initial egg weight from the final egg weight and dividing the difference by the initial egg weight. The percentage value was obtained by multiplying the result by 100, as described by22.
Upon hatching, the chicks were individually weighed and then distributed into four treatment groups. Each treatment group consisted of six replicate pens, with 25 birds housed in each pen.
The floor pens had dimensions of approximately 1.22 m × 2.44 m and were equipped with fresh pine shavings as the litter material. and their live body weights (LBW) were measured weekly till five weeks of age. An electronic balance with a precision of 0.1 g was used for accurate measurements. The average body weight gain (BWG), feed consumption (FC), and feed conversion ratio (FCR) were calculated, FCR was calculated as per gram feed consumed per gram weight produced (g feed/g weight gain).. The environmental temperature, relative humidity, and THI throughout the entire experimental duration as reported in the first part of this study in8. THI in the present study ranged from 29.5 to 33.5. This suggests that the broilers were raised in conditions characterized by significant heat stress. The weekly calculated values of THI have been mentioned8 (it will useful to show data in a table to see this conditions of stress after 24 days of raising, period when 33.5 THI should be stressing to the chicken broilers).
Slaughter traits
When the chicks reached 35 days of age, we randomly chose six chicks from each of the four sub-groups. These chicks were weighed and then slaughtered using cervical dislocation. During the autopsy, the abdomen was opened, and the liver and heart were removed and weighed. The weights were recorded in grams, rounded to the nearest 0.001 g. The carcasses without internal organs were weighed individually, and the percentage of dressed carcass yield was recorded. The different breast muscles, namely the pectoralis major and pectoralis minor, were dissected and weighed. All recorded weights were proportional to the live body weight at the corresponding age.
Blood parameters
In total, 24 blood samples were gathered, with six samples taken from each of the four sub-groups. These samples were collected to determine various plasma parameters. The concentrations of total proteins (g/dl) and albumin (g/dl) were assessed utilizing commercial kits acquired from Spectrum Diagnostics. The globulin levels were derived by deducting the plasma albumin content from the overall plasma protein concentration. Cholesterol (mg/dl), high-density lipoprotein (mg/dl), and triglycerides (mg/dl) were determined using commercial kits from Spectrum Diagnostics. The enzymatic activities of AST and ALT (U/L) were quantified through calorimetric assays using commercial kits obtained from Spectrum Diagnostics.
Plasma hormonal assay
Triiodothyronine (T3) level was assessed using the Eliza technique and commercial Eliza kits obtained from Precheck Bio. Company, following the methodology described by23
Histology and cytology of skeletal muscles
Samples representative of the left major pectoralis muscle in chicks were carefully dissected during the slaughtering process, with dimensions measuring 0.5 × 0.5 cm. The provided samples were promptly preserved by immersing them in an appropriate amount of a 10% formalin solution. To prepare these samples for examination, we followed the paraffin technique outlined in a study by24. Subsequently, we prepared thin sections, each measuring 4–5 microns in thickness, and affixed two sections from each sample onto glass slides. We then proceeded to stain these sections using the standard hematoxylin and eosin (H & E) staining procedure. These histological procedures were carried out at the Pathology Laboratory situated within Cairo University's National Cancer Institute in Egypt.
We examined the histological features of the pectoral muscle tissues by utilizing a low-magnification light microscope (Optica) at 4× magnification. To enhance the clarity of our findings, we captured images of specific samples using a digital camera (Samsung ES75). For precise quantification of various histological components within the pectoral muscles, we conducted histomorphometric measurements employing specialized image analysis software, OPTIKA PROVIEW © 2003–2020.
In each of the three pectoral muscle sections, we quantified the myocyte count within nine distinct fields (three fields per section) for all treatments, employing a magnification level of 10×. Additionally, we measured both the large (LD) and small (SD) diameters of 100 myocytes present on each slide for each treatment. These measurements allowed us to calculate cytometric and histometric indices, following the methodology outlined in25, using the formula provided.
where: LD is the Myocytes' large diameter; SD is the Myocytes' small diameter; \(\pi\) = 3.1416.
Statistical analysis
The data were divided into two distinct experimental periods, each characterized by a unique experimental design. The initial experimental period extended from the storage phase up to the TC procedure. During this phase, the data were subjected to one-way analysis of variance, treating SPIDES as a fixed effect. This analysis was conducted using SAS's GLM procedure, as outlined in the following model:
where: Yij is a trait of interest; µ is an effect of the overall mean; Si is a fixed effect of ith SPIDES period; \(\varepsilon_{ij}\) is a random experimental error assumed NID (0, σe2).
During the second experimental period, which encompassed the duration from E14 until the conclusion of the study, the data underwent a two-way analysis of variance. This analysis involved treating SPIDES and TC as fixed factors and considering their interactions. The analysis was performed using SAS’s GLM procedure, and the model used is described as follows:
where: Yijk is trait of interest; µ is an effect of overall mean; Si is a fixed effect of ith SPIDES period; Ti is a fixed effect of jth thermal conditioning; S|Tj is the interaction effect between Si and Tj. \(\varepsilon_{ijk}\) is a random experimental error assumed NID (0, σe2).
To differentiate between means when applicable, we applied Duncan's multiple range test, as introduced by Duncan in 1955. For the analysis, we divided the percentages of slaughter traits by 100 and subjected them to an arcsin transformation of the square root. However, the actual percentage means are presented for clarity. In terms of statistical significance, we considered a probability level of 0.05 (p ≤ 0.05) as the threshold for acceptance.
To differentiate between means when applicable, we applied Duncan's multiple range test, as introduced by26. For the analysis, we divided the percentages of slaughter traits by 100 and subjected them to an arcsin transformation of the square root. However, the actual percentage means are presented for clarity. In terms of statistical significance, we considered a probability level of 0.05 (p ≤ 0.05) as the threshold for acceptance.
Results
Egg quality
Table 1 shows the data of the influence of SPIDES during a prolonged storage period of 20 days on measurements of both external and internal egg quality.
According to Table 1, the extended preincubation period of 20 days and the frequency of SPIDES during this period could not exert a significant influence on parameters such as egg weight, egg shape index, shell weight percentage, and egg surface area, and yolk color at the 20th day of storage (DOS). Conversely, internal characteristics related to egg quality exhibited significant changes due to SPIDES. Specifically, the ratio of albumen to yolk weights, along with albumen and yolk indices, displayed marked enhancements in the S1 group when contrasted with the control group (S0). The S1 group, characterized by reduced albumen weight ratio, albumen index, and Haugh unit, showcased these improvements.
Egg weight loss%
Egg weight loss% during storage
Table 2 displays the findings for egg weight on the 3rd day of storage (3rd-DOS) and illustrates the effect of SPIDES on egg weight loss percentage during the storage period.
Table 2 reveals that there was no statistically significant differences in the mean egg weights between the two tested groups (S0 and S1) on the 3rd day of storage. However, it is evident that the eggs' weight loss percentage increased significantly in the SPIDES-treated group, with the S1 group exhibiting the highest total egg weight loss percentage throughout the entire storage period compared to the control group (S0).
Egg weight loss during incubation
Table 3 displays the influence of SPIDES and/or TC on the percentage of egg weight loss over the entire incubation duration.
The data clearly indicate that SPIDES did not yield a significant effect on egg weight loss percentage during the entire incubation period. Nevertheless, it's noteworthy that the values for egg weight loss percentage exhibited a significant increase during the TC period, specifically from E14 to E18, in the thermal-conditioned groups (TC1S0, TC1S1) when compared to the control groups (TC0S0, TC0S1).
The data presented in Table 3 reveal a direct correlation between egg weight (g) throughout the incubation time and the changes in egg weight loss. Specifically, the results indicate that SPIDES had no significant impact on egg weight (g) at E18. In contrast, TC led to a significant decrease (p < 0.05) in the average egg weight (g) at the end of TC at E18, primarily due to an increase in egg weight loss percentage.
Productive performance data
Table 4 displays the impact of prenatal heating treatment (SPIDES and TC) on body weight (BW) at hatch and at the marketing age of 35 day of age (DOA).
The clear observation is that the prolonged storage duration led to a significant reduction in body weight in the control group in contrast to the SPIDES group.. Nevertheless, no significant differences were observed in body weight at hatch regarding the thermal condition treatment. Furthermore, the average body weight (BW) at 35 days of age (DOA) was significantly increased due to the influence of both SPIDES, TC, and their interaction. SPIDES positively enhanced the productive performance of the broiler chicks. The TC treatment improved the thermal tolerance of the thermally conditioned groups and Led to increased body weights in comparison to the control groups. Remarkably, the synergistic effect of SPIDES and TC had the most prominent impact, as evidenced by the heaviest body weight at hatch (45.40 g) and at 35 days (2.09 kg) achieved for the S1TC1 group compared to 40.3 g and 1.74 kg, respectively, for the control group (S0TC0). Hence, S1TC1 recorded 2.09 kg, S1TC0 recorded 2.04 kg, S0TC1 recorded 1.9 kg, while S0TC0 recorded 1.54 kg (the difference on final weight is very important to mention).
Table 5 displays the impact of prenatal heating interventions (SPIDES and TC) on parameters such as body weight gain (BWG), feed consumption (FC), and feed conversion ratio (FCR) throughout the entire rearing period.
The trends in average body weight gain (BWG) results followed the same pattern as described earlier for LBW (as shown in Table 4). This similarity can be attributed to the influence of extended storage duration, as well as the impact of SPIDES and TC during the prenatal stage.
As shown in Table 5, a notably significant reduction in feed consumption (FC) was observed in the untreated control group (S0TC0) in comparison to the other heated treatments (SPIDES and TC). Conversely, the S1TC1 group exhibited the highest feed consumption value over the entire rearing period, suggesting an improved thermotolerance mechanism aimed at achieving enhanced growth performance under such stressful conditions. Regarding the findings related to the feed conversion ratio (FCR), it was observed that the groups subjected to thermal conditioning (TC) exhibited more favorable FCR values compared to the non-TC groups. However, less favorable FCR values were computed for the SPIDES group (S1) in comparison to the S0 group, this could be ascribed to the elevated feed intake observed in the S1 group when contrasted with the S0 group. (You can use the FCR corrected to gain weight, this will show to you the better FCR for group S1TC1 for sure. This group had the highest final weight and gain weight and this influence remarkly the FC and FCR). Done.
Blood constituents
Lipid profile
The lipid profile data collected on the 35th day of age after prolonged exposure to heat stress (as depicted in Table 2) have been presented in Table 6. It is noteworthy to mention that, while the overall plasma cholesterol level did not show a significant impact due to TC, it was significantly elevated in the SPIDES groups compared to the control groups. TC led to a significant reduction in plasma triglyceride levels, whereas the TC groups exhibited higher HDL values compared to the non-thermally influenced groups during conditions of heat stress.
Blood proteins profile
Plasma levels of total protein, albumin, and globulin on the 35th day of age are displayed in Table 7. Notably, the TC group (TC1) exhibited significantly elevated levels of total protein, albumin, and globulin compared to the control group (TC0). Regarding the impact of SPIDES treatments on plasma protein levels, it was evident that there were varying patterns observed among the different treatment groups. Notably, the S1 group exhibited the highest values for total proteins and globulin levels in comparison to the control group.
Aspartate aminotransferase (AST) and alanine aminotransferase (ALT) enzymes
The results displayed in Table 8 depicted how SPIDES and TC influenced the plasma levels of liver function enzymes, specifically aspartate aminotransferase (AST) and alanine aminotransferase (ALT). Surprisingly, in the present study, the liver enzymes, both aspartate aminotransferase (AST) and alanine aminotransferase (ALT), did not show significant alterations due to TC when exposed to heat stress conditions. The findings demonstrated that irrespective of the TC's influence, SPIDES treatment effectively lowered the activity of ALT and AST enzymes. Notably, the S1 group displayed a pronounced decrease, indicating enhanced liver function in comparison to the control group.
Breast muscles and some internal organ’s relative weights
Table 9 presents the relative weight of breast muscles on the 35th day of age (DOA). It is evident that embryonic thermal conditioning resulted in a significant increase in the relative weight of the entire breast muscles and the percentage of major pectoralis muscles. However, SPIDES did not have any noticeable impact. Furthermore, the relative weight of minor pectoralis muscles remained unchanged due to both TC and SPIDES.
Metabolic hormone plasma level
The data displayed in Table 10 depicts the plasma T3 levels influenced by SPIDES, both with and without TC, at both hatch and the marketing age (35 DOA). The findings indicated a significant (P ≤ 0.05) reduction in plasma T3 levels at the hatch for the TC groups in comparison to those without TC. The application of SPIDES treatment led to a significant reduction (P ≤ 0.05) in plasma T3 levels at hatching when compared to the remaining groups.. Plasma T3 levels were assessed at the marketing age of 35 days post-hatch, during conditions of chronic heat stress. The results revealed a significant reduction in T3 levels among the TC groups when compared to the control group.
Histological and cytological study
Histological and cytological observations of breast muscle
Figure 1 illustrates the microscopic arrangement of major pectoralis muscles across distinct treatment groups. Through histological examination, it becomes apparent that sections obtained from various treatments exhibit noteworthy distinctions. As a result, the examination and measurement of cytological variances emerge as a superior method for distinguishing between different experimental groups, given their display of characteristic skeletal muscular structures.
Transverse section through pectoralis breast muscle from birds of different treatments at 35 DOA. F fascicles, E endomysium, M myocytes, P perimysium (H & E ×100). S0TC0 = control no SPIDES & incubation under normal condition, S0TC1 control no SPIDES & prenatal thermal conditioning, S1TC0 SPIDES & incubation under normal condition, S1TC1 SPIDES & prenatal thermal conditioning, TC prenatal thermal conditioning, SPIDES short period of incubation during egg storage period.
The count of myocytes per field witnessed a significant increase due to both SPIDES and embryonic thermal conditioning. This rise in myocyte count signifies enhanced lateral growth performance. Furthermore, during the storage period, SPIDES exhibited a positive impact on augmenting the number of myocytes per field.
Regarding the dimensions of myocytes (both in terms of large and small diameters), their thickness (D\(\overline{x}\)), and cross-sectional area (SA), the present findings indicate that the elevated myocyte count observed in the thermally conditioned and SPIDES-exposed groups is correlated with reductions in myocyte dimensions, mean thickness, and cross-sectional area. These reductions were found to be statistically significant when compared to the control groups (Table 11).
Discussion
Extended storage periods of eggs, even when kept in ideal storage conditions, can significantly impair egg quality. This deterioration is evident in the subsequent quality and performance of the chicks that hatch from these eggs4. This effect might arise from the potential loss of albumen solvent from the albumen to the yolk due to the degradation of the ovomucin-lysozyme complex after prolonged storage periods, as observed by27. This breakdown of the ovomucin-lysozyme complex is manifested in the reduction of albumen height, which, in turn, influences the Haugh unit, leading to a significant decrease during extended storage periods. Furthermore28, conducted a study revealing that subjecting eggs to SPIDES at 37.5 °C for 4 h during an extended storage period of 10 days resulted in significantly elevated percentages of albumen weight and Haugh Unit values in comparison to the control group. In contrast, the research conducted by7 found that implementing SPIDES for a duration of 2 h at 36 °C, until the eggshell temperature (EST) reached 32–34 ℃ during extended storage periods ranging from 6 to over 21 days of refrigerated storage, did not yield any discernible impact on internal egg quality. In relation to yolk color, Our results are consistent with those reported in the study by27, who indicated that yolk color remained unaffected by prolonged storage. They attributed any potential alteration in yolk color to the antioxidant content in the hens' diet and other environmental factors.
It has been hypothesized that27 the significant increase in yolk ratio, along with the concurrent decline in yolk index value—a trend consistent with our current results, particularly in the control group relative to the SPIDES group (S1)—may be ascribed to the transfer of water from the albumen to the yolk, which could be linked to a weakening of the vitelline membrane strength (VMS). This weaker membrane could render the yolk more vulnerable to breakage, facilitating the gradual infiltration of water from the albumen. Consequently, the yolk percentage increased compared to the albumen, resulting in the characteristic mottled flattened appearance of the yolk, as manifested in the reduced yolk index of the control group compared to the S1 group.
Conversely, the decline in albumen pH might be due to the evaporation and carbon dioxide exchange from the eggs, shifting the bicarbonate buffer system towards CO2 production, consequently raising the albumen pH6.
The ratio of weight loss in chicken eggs exhibited a noteworthy increase due to extended storage, as evidenced by the studies conducted by29. Furthermore, as indicated by30, the egg weight loss experienced a substantial rise with longer storage periods, with an estimated increase exceeding 10% for eggs stored for 35 days. This escalating trend in egg weight loss percentages over extended storage durations could be attributed to the evaporation-driven loss of solvents from the egg contents through the eggshell27. Concerning with SPIDES effect,22 reported that subjecting the Cobb 500 broiler breeder flock to preheating for durations of 6 or 12 h during extended storage periods of 9 to 12 days led to a significant increase in egg weight loss percentages in comparison to non-heated groups (control). In opposition to the outcomes observed in the current investigation31 reported that pre-incubation treatments applied to 37-week-old broiler breeder eggs had no significant impact on egg weight losses throughout the entire storage period.
Observations made by32 unveiled that the prolonged storage of Guinea fowl eggs for a period of 10 days led to a significant rise in egg weight loss during storage, in contrast to eggs stored for just 4 days. This underscores a direct correlation between the duration of egg storage and the rate of egg weight loss.
our findings align with the results of33, As indicated by their research, which involved raising the eggshell temperature to 38.9 °C for the Cobb 500 commercial breeder flocks aged 37 to 45 weeks, starting from E 8 onwards, led to significant percentages of egg weight loss at E18. However, this effect did not extend to create substantial differences in the overall egg weight. The current observation of non-significance in egg weight loss percentage during incubation due to SPIDES corresponds with the conclusions drawn by34,35, who determined that a prolonged storage period for chicken eggs did not have a noteworthy impact on the percentages of egg weight loss during the incubation process. Moreover, when examining the incubation-related egg weight loss of Cobb 500 broiler breeder flocks that were preheated for 6 or 12 h after being stored for 9 to 12 days, there was no statistically significant distinction from the non-heated (control) groups, as indicated by22.
It is evident that an extended storage duration resulted in a decrease in the body weight of the control group compared to the SPIDES groups. In this context,33 observed that elevating the eggshell temperature to 38.9 °C for 6 h during the incubation of Cobb 500 commercial breeder flocks aged 37 to 45 weeks, starting from E8 onwards, led to extended chick length after hatching but reduced body weight compared to the control group. In contrast to our findings,36 demonstrated a significant improvement in chick weight at hatch following thermal conditioning at 40 °C for 4 h per day from E14 to E17, in comparison to the control group.
Furthermore, average body weights (BW) experienced a significant increase at 35 days of age (DOA) due to the effect of both SPIDES and thermal conditioning (TC), as well as their interaction. SPIDES positively impacted the productive performance of broiler chicks, with its effectiveness being linked to the timing of SPIDES. The TC treatment displayed the capability to enhance the heat tolerance of the thermally conditioned groups, leading to higher body weights when contrasted with the control groups. Notably, the combined effect of SPIDES and TC was even more pronounced, evident in the heaviest body weight at hatch (45.40 g) achieved by the S1TC2 group when contrasted with the control group(S0TC0) with a weight of 40.3 g. In alignment with the present results, El-Garhy37 identified a significant augmentation in live body weight (LBW) and the rate of body weight gain (BWG) both at hatch and at the 5th week of age when compared with untreated groups. Partially concurring with the current findings, Tona et al.38 found that extended storage of eggs from young breeders (35 weeks of age) significantly decreased the relative weight gain of 7-day-old chicks.
Regarding the feed conversion ratio (FCR) results, it was observed that the groups subjected to thermal conditioning (TC) demonstrated better FCR than the non-TC groups. However, the SPIDES group (S1) exhibited poorer FCR compared to the S0 group. This observation was attributable to the higher feed consumption (FC) observed in the S1 treatment group in contrast to the S0 group. (review this affirmation with correted FCR). Done.
In accordance with our findings, previous research conducted by39 revealed that chicks originating from thermally conditioned eggs displayed significantly higher live body weight (LBW), body weight gain (BWG), and improved feed conversion ratio (FCR) both at hatch and throughout subsequent ages, in comparison with the control group. Similarly, Meteyake et al.40 showed that exposing Ross eggs to thermal conditioning at 39.5 °C for 12 h daily, starting from embryonic day 7 (E7) and continuing until embryonic day 16 (E16), led to improved final body weight, reduced feed conversion, and lowered feed intake.
In contrast, studies by41,42,43, demonstrated that a decrease in body weight from hatching until 6 weeks of age for chicks hatched from eggs exposed to different thermal conditioning regimes. Sengor et al.41 exposed eggs to TC at 39 °C for 2 h per day at E14 and E15, while42 subjected eggs to 39.6 °C or 40.6 °C. Similarly, Zaboli et al.43 found that thermal conditioning of Ross 308 eggs at 39.5°C and 65% relative humidity for 12 h per day from E7 to E16 led to a decrease in body weight when contrasted with the control group.
Contrastingly, In the later phases of the incubation process, studies by44,45 had no significant variances in body weight, feed consumption, and feed conversion ratio between control chicks and those subjected to embryonic thermal conditioning. Furthermore, neither exposing Hubbard eggs to 38.8°C for 6 or 18 h per day from E10 to E1846 nor subjecting them to 39°C and 65% relative humidity for 18 h per day from E0 to E1847 had a significant impact on body weight at hatch and up to 4 weeks of age.
It was proposed that stress might lead to an elevation in overall cholesterol levels by boosting LDL while reducing HDL, which promotes the onset of cardiovascular disorders48. The same finding was obtained under conditions of prolonged exposure to heat stress at 34 °C for 8 h daily from the 22nd to the 42nd h49, as well as in instances where a broiler was subjected to extended periods of heat stress at 36 °C50. Similarly, under conditions of cyclic stress, there was a significant rise in plasma cholesterol levels51.
Triglyceride plasma levels were notably lowered by TC, in contrast to the higher HDL values observed in TC groups compared to non-thermally influenced groups under heat stress conditions. This indicates that the TC groups adapted metabolically to the heat stress52. Furthermore, they noted a significant elevation in triglyceride levels, total cholesterol levels, and low-density lipoprotein cholesterol levels in broiler chicks at 42 days old as a result of prolonged exposure to chronic heat stress at 32 °C from the 22nd to the 42nd day of age. On the other hand, cyclic heat stress at 36 °C from the 16th to the 42nd day of age had no significant effect on plasma triglycerides, HDL, or LDL levels51.
The plasma levels of lipid parameters due to the influence of SPIDES displayed inconsistent value trends, suggesting that the variations in lipid parameters were primarily influenced by chronic heat stress, as well as the interaction between SPIDES and TC. The S1TC1 group exhibited significantly higher plasma HDL levels and lower TG levels when contrasted with the control group S0TC0, indicating the enhancing effect of SPIDES on plasma lipid profile and lipid metabolism under heat stress conditions. These findings align with those of32 who found that storing Guinea fowl eggs for 15 days increased total cholesterol and triglyceride concentrations compared to chicks hatched from eggs stored for three days.
The significant elevated levels of total protein and albumin observed in the (S1TC1) group, as opposed to the control (S0TC0) group, potentially indicate the heightened metabolic protein activity within the S1TC1 group due to its increased feed consumption (FC) under heat stress circumstances. This observation underscores the synergistic collaboration between TC and SPIDES in enhancing liver protein synthesis, metabolic efficiency, and overall chick performance when faced with heat stress conditions36,53.
In the present investigation, it was unexpectedly observed that the levels of both liver enzymes remained largely unchanged by TC when subjected to conditions of heat stress. This lack of statistically significant alteration might be attributed to the interplay between the varied performance of chicks hatched from SPIDES groups and their adaptive response to heat stress, wherein they seem to have developed a viable metabolic mechanism to enhance their resilience against heat-induced challenges. Notably, it is worth highlighting that irrespective of TC's impact, the administration of SPIDES treatment was able to considerably diminish the activity of ALT and AST enzymes. This effect was particularly pronounced in the S1 group, indicating improved liver functionality when contrasted with the control group. In this context, it is worth noting that54 documented that subjecting broiler chicks to thermal stress at 34 °C for a duration of 8 h resulted in heightened plasma levels of AST and ALT. Furthermore,55 noted that elevated ambient temperatures had the potential to trigger liver impairment, leading to an escalation in serum AST and ALT concentrations.
Our results are consistent with the discoveries made in the study by56, who illustrated that subjecting embryos to temperature conditioning (TC) at 39.5 °C for either 3 or 6 h per day during the later stages of incubation (E16 to E18) resulted in enhanced development of the major pectoralis muscle in chicks by the 35th day of age, when contrasted with the control group. Additionally, applying TC at 39.5 °C for 12 h per day, spanning from E7 to E16, significantly improved the yield of breast muscle and concurrently reduced the relative percentage of abdominal fat at the age of market readiness. This was corroborated by the studies of57,58,59.
In a similar vein, a study by39 unveiled that subjecting broiler eggs to temperature conditioning at 38.6 °C and 65% relative humidity for 6 h per day, starting from E16 to E8, resulted in a rise in the weight of breast muscles upon reaching the market age.
The thyroid hormone holds a pivotal function in avian thermogenesis, facilitating thermoregulation by regulating the generation of metabolic heat. This regulation is essential for sustaining a consistent body temperature across varying thermal environments, as emphasized by60. The results indicated a significant (P ≤ 0.05) reduction in plasma T3 levels at hatch for the TC groups in comparison to those without TC.
This reduction in T3 levels in the TC chicks corresponded with a lowered metabolic rate, leading to decreased heat production in response to the heat stress conditions. The findings indicated a significant (P ≤ 0.05) reduction in plasma T3 levels at hatch within the TC groups compared to the non-TC groups. This decrease suggests a higher metabolic rate in TC1 chicks in contrast to the control group (TC0), which in turn promoted improved growth performance. This observation has been attributed to embryonic Thermal manipulation between developmental days E6 to E16, influencing the establishment of the brain's thermoregulatory center. This alteration of the 'setpoint' for thermotolerance systems subsequently influences the metabolic rate under similar environmental conditions, as elucidated by60. Energy usage, both in a general sense and specifically within domestic fowl, can be categorized into two main aspects: maintenance and production. In warm-blooded organisms (homeotherms), maintaining a stable body temperature (Tb) requires a significant portion of energy for basic upkeep. Consequently, if we can reduce the energy needed for maintenance while maintaining a consistent overall energy consumption, it's probable that more energy would be accessible for productive purposes. Alternatively, another approach could involve a combination of lowering the energy required for maintenance along with an overall reduction in energy consumption61. Numerous researchers have employed embryonic thermal manipulation at around 39.5 °C for 6 to 12 h per day to enhance thermotolerance in the face of heat stress. This manipulation has been associated with changes in circulating T3 and T4 plasma levels, indicating an enhancement in thermotolerance under thermal stress conditions46,62,63.
Conversely, applying the SPIDES treatment during the 20 days of incubation showed a significant reduction (P ≤ 0.05) in T3 plasma levels at hatch compared to the other groups. This effect might be attributed to the earlier hatching time of the S1 group. The accelerated embryonic metabolic rate before hatch could be responsible for aiding the embryo in transitioning to lung ventilation and engaging its respiratory and leg muscles to hatch. Given that the S1 group hatched earlier, their T3 plasma levels likely returned to normal more swiftly compared to the chicks that hatched later.
Assessment of estimated plasma T3 levels at the marketing age of 35 days, under chronic heat stress conditions, revealed a significant reduction in TC groups when contrasted with the control group. This reduction in T3 levels among TC chicks indicates a lowered metabolic rate and subsequently reduced heat production under heat stress. This aligns with the observation that broiler chickens tend to decrease both the size and activity of their thyroid gland under high ambient temperatures64. Additionally, conducting chronic incubation at a temperature of 39.0 °C for 3 h from embryonic days E6 to E8 Led to a reduction in the plasma levels of T357,65 noted a significant reduction in both T3 levels and the T3/T4 ratio, alongside an elevation in plasma T4 levels, as an outcome of subjecting embryos to temperature conditioning (TC) at 39.5 °C for 12 h daily, encompassing the period from E7 to E16. Similarly, Al-Zghoul et al.46 noted that elevating the surrounding temperature to 41.0 °C for 6 h per day at 3, 7, or 42 days of age led to a significant reduction in plasma T3 levels at 42 days of age in contrast to normal incubation or control conditions.
However, it should be noted that embryonic thermal manipulation at 39 °C for 3 h per day from E16 to E18 of embryonic development increased the concentration of free T3 hormone on E19 and decreased the concentration of free T4 hormone in the embryo's blood, as opposed to the control group66. In contrast, embryonic TC at 40 °C for 4 h per day from E14 to E17 had no significant effect on T3 serum levels in Golden Montazah chickens36.
The count of myocytes per field witnessed a significant rise due to both SPIDES and embryonic thermal conditioning. This surge in myocyte numbers signifies enhanced lateral growth performance. Additionally, during the storage period, SPIDES exhibited a beneficial impact on augmenting myocyte count per field.
In terms of myocyte dimensions, encompassing both large and small diameters, thickness, and cross-sectional area, the present findings suggest that the escalated count of myocytes within thermally conditioned and SPIDES-exposed groups is mirrored in their myocyte dimensions, mean thickness, and cross-sectional area. Notably, these measurements were significantly lesser when compared to the control groups.
It is widely recognized that muscle fiber count, dimensions, and fiber-type distribution are intricately interlinked67. Muscle function in mature animals is significantly influenced by factors such as type and number of muscle fiber, which subsequently impact fiber size. The total muscle mass is determined by the aggregate of the myofiber count, their cross-sectional area, and their length68.
Through histological examination and cytological measurements of pectoralis muscles in this current investigation, it is implied that subjecting broiler eggs to temperature conditioning throughout the latter stages of embryonic development leads to an increase in the count of myocytes within the pectoralis muscles upon reaching the marketing age.
In previous investigations, it was reported that subjecting embryos to thermal conditioning (TC) between developmental stages E16 and E18 led to the stimulation of myonuclei proliferation. As a result, the number of myonuclei in the breast muscles of newly hatched broiler chicks increased. This phenomenon of myonuclei hyperplasia subsequently brought about an enlargement of the pectoralis muscles by augmenting the count of muscle fibers, or myocytes. This, in turn, contributed to an increased yield of breast muscles at the time of market age. This concept aligns with the outcomes of69.
Similarly,70 demonstrated that deviating from the standard incubation temperature range could directly impact the molecular processes essential for myoblast proliferation and differentiation. Alternatively, it might indirectly influence myogenesis by promoting metabolic activity and encouraging embryonic movement within the egg. In a similar vein, exposing late-term embryos to elevated temperatures (38.5 °C or 39.5 °C for 3 or 6 h per day) was found to enhance both myofiber diameter and myoblast proliferation, thereby promoting overall muscle growth at the market stage, as documented by56,71.
Additionally, these findings align with69, who demonstrated that subjecting embryos to heat conditioning at 39.5 °C for 5 h per day during the E16 to E18 timeframe significantly amplified the count of myocytes. This increase was observed to influence myocyte dimensions and cross-sectional area, leading to substantial post-hatch growth among the thermally conditioned groups compared to the control groups.
The improved histological structure of the thermally conditioned and S1 groups is further substantiated by the higher relative weights of breast muscles observed at market age in the thermally conditioned and S1 groups, particularly in the S1TC1 treatment when compared to the other groups.
Conclusion
SPIDES treatment group displayed marked enhancements in internal characteristics related to egg quality, SPIDES positively enhanced the productive performance of the broiler chicks. Moreover, The TC treatment improved the thermal tolerance of the thermally conditioned groups and led to increased body weights. Remarkably, the synergistic effect of SPIDES and TC had the most prominent impact. The count of myocytes had a significant increase due to both SPIDES and embryonic thermal conditioning, it could be concluded that SPIDES and TC have beneficial effects on pre- and post-hatch characteristics of broiler chicks up until the marketing age. Additionally, TC techniques improve chick performance, particularly under conditions of heat stress, and enhance the yield of breast meat in later stages of life.
Data availability
The datasets employed and/or examined in the present study can be obtained from the corresponding author upon a reasonable request.
Abbreviations
- AST:
-
Aspartate aminotransferase
- ALT:
-
Alanine aminotransferase
- BW:
-
Body weights
- BWG:
-
Body weight gain
- FC:
-
Feed consumption
- FCR:
-
Feed conversion ratio
- SPIDES:
-
Short periods of incubation during egg storage
- TC:
-
Thermal conditioning
- T3:
-
Triiodothyronine
- TG:
-
Triglyceride
- HDL:
-
High density lipoprotein
References
Damaziak, K., Paweska, M., Gozdowski, D. & Niemiec, J. Short periods of incubation, egg turning during storage and broiler breeder hens age for early development of embryos, hatching results, chicks quality and juvenile growth. Poultry Sci. 97, 3264–3276. https://doi.org/10.3382/ps/pey163 (2018).
Abioja, M. O., Abiona, J. A., Akinjute, O. F. & Ojoawo, H. T. Effect of storage duration on egg quality, embryo mortality and hatchability in FUNAAB-ɑ chickens. J. Anim. Physiol. Anim. Nutr. 105, 715–724 (2021).
Martínez, Y., Soliz, N.D., Bejarano, M.A., Paz, P. & Valdivie, M. Effect of storage duration and temperature on daily changes in external and internal egg quality of eggs from Dekalb White® laying hens. J. Eur. Poultry Sci./Archiv Geflügelkunde (2021).
Melo, E. F. et al. Effect of egg storage duration and egg turning during storage on egg quality and hatching of broiler hatching eggs. Anim. Int. J. Anim. Biosci. 15, 100111. https://doi.org/10.1016/j.animal.2020.100111 (2021).
Özlü, S. Research note: Storage period and prewarming temperature effects on synchronous egg hatching from broiler breeder flocks during the early laying period. Poultry Sci. 100, 100918. https://doi.org/10.1016/j.psj.2020.12.016 (2021).
Özlü, S., Uçar, A., Erkuş, T., Nicholson, A. D. & Elibol, O. Research note: Effects of turning and short period of incubation during long-term egg storage on embryonic development and hatchability of eggs from young and old broiler grandparent flocks. Poultry Sci. 100, 101026. https://doi.org/10.1016/j.psj.2021.101026 (2021).
Maman, A.H.; & Yildirim, I. The effect of short periods of incubation during egg storage (SPIDES) on internal egg quality, hatchability and chick quality of long stored old layer breeder eggs. Eur. Poultry Sci./Arch. Geflügelkunde (2022).
Hemida, M. A. et al. Hepatic heat shock proteins, antioxidant-related genes, and immunocompetence of heat-stressed broilers in response to short periods of incubation during egg storage and thermal conditioning. J. Therm. Biol. 116, 103640. https://doi.org/10.1016/j.jtherbio.2023.103640 (2023).
Batool, F. et al. An updated review on behavior of domestic quail with reference to the negative effect of heat stress. Anim. Biotechnol. 34, 424–437. https://doi.org/10.1080/10495398.2021.1951281 (2023).
Vandana, G. D. et al. Heat stress and poultry production: Impact and amelioration. Int. J. Biometeorol. 65, 163–179. https://doi.org/10.1007/s00484-020-02023-7 (2021).
Madkour, M. et al. Mitigating the detrimental effects of heat stress in poultry through thermal conditioning and nutritional manipulation. J. Therm. Biol. 103, 103169. https://doi.org/10.1016/j.jtherbio.2021.103169 (2022).
Madkour, M., Aboelazab, O., Abd El-Azeem, N., Younis, E. & Shourrap, M. Growth performance and hepatic antioxidants responses to early thermal conditioning in broiler chickens. J. Anim. Physiol. Anim. Nutr. (Berl) 107, 182–191. https://doi.org/10.1111/jpn.13679 (2023).
Madkour, M. et al. Early life thermal stress modulates hepatic expression of thermotolerance related genes and physiological responses in two rabbit breeds. Ital. J. Anim. Sci. 20, 736–748. https://doi.org/10.1080/1828051X.2021.1914207 (2021).
Madkour, M. et al. Antioxidants status and physiological responses to early and late heat stress in two rabbit breeds. J. Anim. Physiol. Anim. Nutr. (Berl) 107, 298–307. https://doi.org/10.1111/jpn.13781 (2023).
Ouchi, Y., Chowdhury, V. S., Cockrem, J. F. & Bungo, T. J. Effects of thermal conditioning on changes in hepatic and muscular tissue associated with reduced heat production and body temperature in young chickens. Front. Vet. Sci. 7, 610319 (2021).
Bakst, M. R., Welch, G. R. & Camp, M. J. Observations of turkey eggs stored up to 27 days and incubated for 8 days: Embryo developmental stage and weight differences and the differentiation of fertilized from unfertilized germinal discs. Poultry Sci. 95, 1165–1172. https://doi.org/10.3382/ps/pew010 (2016).
Reddy, P. M., Reddy, V. R., Reddy, C. V. & Rao, P. S. P. Egg weight, shape index and hatchability in Khaki Campbell duck eggs. Indian J. Poultry Sci. 14, 26–31 (1979).
Anderson, K. E., Tharrington, J. B., Curtis, P. A. & Jones, F. T. Shell characteristics of eggs from historic strains of single comb white leghorn chickens and the relationship of egg shape to shell strength. Int. J. Poultry Sci. 3, 17–19 (2004).
Wells, R.G. The measurement of certain egg quality characteristics: A review. In Egg Quality. A Study of the Hen’s Egg. 207–250 (Oliver and Boyd, 1968).
Haugh, R. R. The Haugh unit for measuring egg quality. U. S. Egg Poultry Mag. 43, 552–555 (1937).
Monira, K. N., Salahuddin, M. & Miah, G. J. Effect of breed and holding period on egg quality characteristics of chicken. Int. J. Poultry Sci. 2, 261–263 (2003).
Silva, F. H. A. et al. Influence of egg pre-storage heating period and storage length on incubation results. Braz. J. Poultry Sci. 10, 17–22 (2008).
Britton, K. E. et al. Is “T4 toxicosis” a normal biochemical finding in elderly women?. Lancet (London, England) 2, 141–142. https://doi.org/10.1016/s0140-6736(75)90053-7 (1975).
Abd El-Hamid, Z. Histology: Part I (Dar El Shaab for Press, 1981).
Radu-Rusu, R. M., Teuşan, V. & Vacaru-Opriş, I. Aspects concerning the histological structure of the biceps brachialis muscles in chicken broilers. Lucrări Ştiinţifice Ser. Zooteh. 52, 266–270 (2009).
Duncan, D. B. M. Multiple range and multiple F tests. Biometrics 11, 1–42 (1955).
Akter, Y., Kasim, A., Omar, H. B., Sazili, A. Q. & Danesh, H. M. Effect of storage time and temperature on the quality characteristics of chicken eggs. J. Food Agric. Environ. 12, 87–92 (2014).
Kalaba, Z. M., Abo Egla, E. H. A. & Taman, M. A. If pre-storage heating improve the hatchability and decreased embryonic mortality of broiler breeder eggs stored for long period. J. Anim. Poultry Prod. 8, 149–153 (2017).
Goliomytis, M., Tsipouzian, T. & Hager-Theodorides, A. L. Effects of egg storage on hatchability, chick quality, performance and immunocompetence parameters of broiler chickens. Poultry Sci. 94, 2257–2265. https://doi.org/10.3382/ps/pev200 (2015).
González-Redondo, P. Effect of long-term storage on the hatchability of red-legged partridge (Alectoris rufa) eggs. Poultry Sci. 89, 379–383. https://doi.org/10.3382/ps.2009-00408 (2010).
Petek, M. & Dikmen, S. The effects of prestorage incubation and length of storage of broiler breeder eggs on hatchability and subsequent growth performance of progeny. Czech J. Anim. Sci. 51, 73–77 (2006).
Kouame, Y. A. E., Voemesse, K., Lin, H., Onagbesan, O. M. & Tona, K. Effects of egg storage duration on egg quality, metabolic rate, hematological parameters during embryonic and post-hatch development of guinea fowl broilers. Poultry Sci. 100, 101428. https://doi.org/10.1016/j.psj.2021.101428 (2021).
van den Brand, H. et al. Interaction between eggshell temperature and carbon dioxide concentration after day 8 of incubation on broiler chicken embryo development. Anim. Int. J. Anim. Biosci. 15, 100223. https://doi.org/10.1016/j.animal.2021.100223 (2021).
Fasenko, G. M., Robinson, F. E., Whelan, A. I., Kremeniuk, K. M. & Walker, J. A. Prestorage incubation of long-term stored broiler breeder eggs: 1. Effects on hatchability. Poultry Sci. 80, 1406–1411. https://doi.org/10.1093/ps/80.10.1406 (2001).
Reijrink, I. A. M., Meijerhof, R., Kemp, B. & Van Den Brand, H. The chicken embryo and its micro environment during egg storage and early incubation. World’s Poultry Sci. J. 64, 581–598. https://doi.org/10.1017/S0043933908000214 (2008).
Badran, A. M., Desoky, A. A., Abou-Eita, E. M. & Stino, A. F. K. Epigenetic thermal adaptation of chickens during late embryonic development. Egypt. Poultry Sci. 32, 675–689 (2012).
El-Garhy, O. H. Effect of short period of incubation during egg storage (spides), storage period and flock age on productivity of post-hatching broiler chickens. Ann. Agric. Sci. Moshtohor 59, 743–754 (2021).
Tona, K., Onagbesan, O., De Ketelaere, B., Decuypere, E. & Bruggeman, V. Effects of age of broiler breeders and egg storage on egg quality, hatchability, chick quality, chick weight, and chick posthatch growth to forty-two days. J. Appl. Poultry Res. 13, 10–18. https://doi.org/10.1093/japr/13.1.10 (2004).
Abdel-Fattah, S. & Shourrap, M. Growth muscular proliferation and metabolic hormones expression in broiler chicks as affected by folic acid administration and embryonic thermal conditioning. Egypt. J. Nutr. Feeds 16, 195–202 (2013).
Meteyake, H.T., Bilalissi, A., Oke, O.E., Voemesse, K. & Tona, K. Effect of thermal manipulation during incubation and heat challenge during the early juvenile stage on production parameters of broilers reared under a tropical climate. Eur. Poultry Sci./Arch. Geflügelkunde (2020).
Sengor, E., Yardimci, M., Okur, N. & Can, U. Effect of short-term pre-hatch heat shock of incubating eggs on subsequent broiler performance. S. Afr. J. Anim. Sci. 38, 58–64 (2008).
Willemsen, H. Effect of Temperature Manipulation During Late Incubation on Metabolism and Performance of Broiler Embryos and Chickens. (Katholieke University Leuven, GroepWetenschap & Technologie, 2011).
Zaboli, G. R. et al. Thermal manipulation during pre and post-hatch on thermotolerance of male broiler chickens exposed to chronic heat stress. Poultry Sci. 96, 478–485. https://doi.org/10.3382/ps/pew344 (2017).
Collin, A., Picard, M. & Yahav, S. The effect of duration of thermal manipulation during broiler chick embryogenesis on body weight and body temperature of post-hatched chicks. J. Anim. Res. 54, 105–111 (2005).
Ipek, A., Sahan, U., Baycan, S. C. & Sozcu, A. The effects of different eggshell temperatures on embryonic development, hatchability, chick quality, and first-week broiler performance. Poultry Sci. 93, 464–472. https://doi.org/10.3382/ps.2013-03336 (2014).
Al-Zghoul, M. B. et al. Thermal manipulation during chicken embryogenesis results in enhanced Hsp70 gene expression and the acquisition of thermotolerance. Res. Vet. Sci. 95, 502–507. https://doi.org/10.1016/j.rvsc.2013.05.012 (2013).
Saleh, K. M. M., Tarkhan, A. H. & Al-Zghoul, M. B. Embryonic thermal manipulation affects the antioxidant response to post-hatch thermal exposure in broiler chickens. Anim. Open Access J. MDPI https://doi.org/10.3390/ani10010126 (2020).
Bergmann, N., Gyntelberg, F. & Faber, J. The appraisal of chronic stress and the development of the metabolic syndrome: A systematic review of prospective cohort studies. Endocr. Connect. 3, R55-80. https://doi.org/10.1530/ec-14-0031 (2014).
Ahmed-Farid, O. A., Salah, A. S., Nassan, M. A. & El-Tarabany, M. S. Effects of chronic thermal stress on performance, energy metabolism, antioxidant activity, brain serotonin, and blood biochemical indices of broiler chickens. Anim. Open Access J. MDPI https://doi.org/10.3390/ani11092554 (2021).
Attia, Y.A. & Hassan, S.S. Broiler tolerance to heat stress at various dietary protein/energy levels. Eur. Poultry Sci./Arch. Geflügelkunde 81 (2017).
Bueno, J. P. R. et al. Effect of cyclic heat stress on thyroidal hormones, thyroid histology, and performance of two broiler strains. Int. J. Biometeorol. 64, 1125–1132. https://doi.org/10.1007/s00484-020-01886-0 (2020).
Ding, J. et al. Effects of dietary supplementation of fumaric acid on growth performance, blood hematological and biochemical profile of broiler chickens exposed to chronic heat stress. Braz. J. Poultry Sci. 22, 17 (2020).
Akbarian, A. et al. Association between heat stress and oxidative stress in poultry; mitochondrial dysfunction and dietary interventions with phytochemicals. J. Anim. Sci. Biotechnol. 7, 37. https://doi.org/10.1186/s40104-016-0097-5 (2016).
Zhang, J. et al. Curcumin attenuates hepatic mitochondrial dysfunction through the maintenance of thiol pool, inhibition of mtDNA damage, and stimulation of the mitochondrial thioredoxin system in heat-stressed broilers. J. Anim. Sci. 96, 867–879. https://doi.org/10.1093/jas/sky009 (2018).
Lan, R., Wei, L., Chang, Q., Wu, S. & Zhihui, Z. Effects of dietary chitosan oligosaccharides on oxidative stress and inflammation response in liver and spleen of yellow-feather broilers exposed to high ambient temperature. Ital. J. Anim. Sci. 19, 1508–1517. https://doi.org/10.1080/1828051X.2020.1850215 (2020).
Piestun, Y., Harel, M., Barak, M., Yahav, S. & Halevy, O. Thermal manipulations in late-term chick embryos have immediate and longer term effects on myoblast proliferation and skeletal muscle hypertrophy. J. Appl. Physiol. (Bethesda, Md.) 106, 233–240. https://doi.org/10.1152/japplphysiol.91090.2008 (2009).
Loyau, T. et al. Thermal manipulation of the embryo modifies the physiology and body composition of broiler chickens reared in floor pens without affecting breast meat processing quality. J. Anim. Sci. 91, 3674–3685. https://doi.org/10.2527/jas.2013-6445 (2013).
Piestun, Y., Druyan, S., Brake, J. & Yahav, S. Thermal treatments prior to and during the beginning of incubation affect phenotypic characteristics of broiler chickens posthatching. Poultry Sci. 92, 882–889. https://doi.org/10.3382/ps.2012-02568 (2013).
Piestun, Y., Druyan, S., Brake, J. & Yahav, S. Thermal manipulations during broiler incubation alter performance of broilers to 70 days of age. Poultry Sci. 92, 1155–1163. https://doi.org/10.3382/ps.2012-02609 (2013).
Piestun, Y. et al. Thermal manipulations during broiler embryogenesis: effect on the acquisition of thermotolerance. Poultry Sci. 87, 1516–1525. https://doi.org/10.3382/ps.2008-00030 (2008).
Piestun, Y. et al. Thermal manipulations during broiler embryogenesis improves post-hatch performance under hot conditions. J. Therm. Biol. 36, 469–474. https://doi.org/10.1016/j.jtherbio.2011.08.003 (2011).
Al-Zghoul, M. B. et al. Hsp90, Hsp60 and HSF-1 genes expression in muscle, heart and brain of thermally manipulated broiler chicken. Res. Vet. Sci. 99, 105–111. https://doi.org/10.1016/j.rvsc.2014.12.014 (2015).
Piestun, Y., Zimmerman, I. & Yahav, S. Thermal manipulations of turkey embryos: The effect on thermoregulation and development during embryogenesis. Poultry Sci. 94, 273–280. https://doi.org/10.3382/ps/peu047 (2015).
Gonzalez-Rivas, P. A. et al. Effects of heat stress on animal physiology, metabolism, and meat quality: A review. Meat Sci. 162, 108025. https://doi.org/10.1016/j.meatsci.2019.108025 (2020).
Abuoghaba, A. A., Ezzat, W., Rizk, A. M., Qurtam, A. A. & El-Sayed, O. A. Egg production, fertility, hatchability and immune responses of some chicken strains under high ambient temperatures. J. Anim. Physiol. Anim. Nutr. (Berl) 105, 725–730. https://doi.org/10.1111/jpn.13482 (2021).
Al-Rukibat, R. K., Al-Zghoul, M. B., Hananeh, W. M., Al-Natour, M. Q. & Abu-Basha, E. A. Thermal manipulation during late embryogenesis: Effect on body weight and temperature, thyroid hormones, and differential white blood cell counts in broiler chickens. Poultry Sci. 96, 234–240. https://doi.org/10.3382/ps/pew298 (2017).
Ryu, Y. C., Rhee, M. S. & Kim, B. C. Estimation of correlation coefficients between histological parameters and carcass traits of pig longissimus dorsi muscle. Asian-Aust. J. Anim. Sci. 17, 428–433. https://doi.org/10.5713/ajas.2004.428 (2004).
Tůmová, E. & Teimouri, A. Chicken muscle fibres characteristics and meat quality: A review. Sci. Agric. Bohem. 40, 253–258 (2009).
Abdel-Fattah, S., Shourrap, M. & Hemida, M. A. Research article effect of pre-and post-hatch thermal conditioning on productive performance, some metabolic hormones and breast muscles growth of broilers chicks. Asian J. Anim. Vet. Adv. 13, 369–376 (2018).
Werner, C. & Wicke, M. Increasing the temperature at a certain period during the incubation of broiler eggs improves the carcass and breast yields without a negative impact on the meat quality. Arch. Anim. Breed. 51, 11–16 (2008).
Halevy, O., Yahav, S. & Rozenboim, I. Enhancement of meat production by environmental manipulations in embryo and young broilers. World’s Poultry Sci. J. 62, 485–497 (2006).
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
Conceptualization, Methodology, Formal analysis M.M., M.A.H., S.A.A., and M.S., Supervision, Data curation M.M., M.A.H., S.A.A., and M.S. Conceptualization, Resources, Software. M.M., M.A.H., S.A.A., and M.S. Investigation, M.A.H., S.A.A., and M.S. Writing an original draft, Writing—review & editing, Investigation M.M., M.A.H., and S.A.A. All authors have drafted, reviewed, revised, and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abdel-Fattah, S.A., Madkour, M., Hemida, M.A. et al. Growth performance, histological and physiological responses of heat-stressed broilers in response to short periods of incubation during egg storage and thermal conditioning. Sci Rep 14, 94 (2024). https://doi.org/10.1038/s41598-023-50295-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-50295-x
- Springer Nature Limited