Abstract
Multiple studies have showed negative impact of non-infectious uveitis on quality of life (QoL). Less is understood regarding life experiences in patients with infectious uveitis. We investigated vision-related QoL in individuals who had recovered from ocular syphilis. 32 adults treated for ocular syphilis at a uveitis service in Brazil completed the 25-item National Eye Institute Visual Function Questionnaire (NEI VFQ-25), and a comprehensive ophthalmic examination was performed. Medical records were reviewed to confirm resolution of ocular inflammation for 3 months pre-enrolment, and collect clinical data. The NEI VFQ-25 composite score was low overall (75.5 ± 19.8, mean ± standard deviation), and subscale scores varied from relative lows of 59.1 ± 39.6 (driving) and 60.9 ± 24.5 (mental health), to relative highs of 84.8 ± 21.8 (ocular) and 89.1 ± 21.0 (color vision). Adults aged over 40 years and those with a final visual acuity of 20/50 or worse had significantly lower mean composite and subscale scores. Other clinical characteristics—including gender, HIV co-infection, and type of uveitis—did not significantly influence scores. Our findings, taken in context with previous observations that prompt recognition achieves better vision outcomes, suggest early treatment may improve QoL after recovery from ocular syphilis.
Similar content being viewed by others
Introduction
Ocular syphilis has been re-emerging since approximately 2000, now accounting for more than 5% of patients referred to some tertiary referral inflammatory eye disease clinics1. The condition manifests as uveitis in the majority of patients, and while posterior and pan- uveitis are the most common forms, anterior and intermediate uveitis also occur2,3,4. Indeed, ocular syphilis has been nicknamed ‘the great masquerader’ due to the myriad of presentations that have been described5. Unlike most other forms of uveitis, the treatment of ocular syphilis is straightforward and inexpensive, and it is definitive: per the recommendations of the United States Centers for Disease Control and Prevention recommendations, a 10 to 14 day course of intravenous penicillin G, or ceftriaxone in the case of allergy, is curative6.
Taken as a group, uveitis causes 5% to 20% of blindness in industrialized countries and 25% in developing regions7, and approximately two-thirds of affected individuals experience prolonged vision loss8. Appropriately, there is an increasing interest in quality of life or patient-reported outcomes in uveitis. Most work has focused on non-infectious forms of uveitis, including groups of patients with different diagnoses from individual clinics9,10,11,12 or enrolled in randomized controlled trials13,14, as well as those with specific diagnoses, such as HLA-B27-asociated, Behçet disease and Vogt-Koyanagi-Harada disease15,16,17. These studies have demonstrated clear negative impact of the disease on quality of life, to an extent greater than that seen in diabetic retinopathy18 and age-related macular degeneration19.
A limited number of studies focused on certain subtypes—herpetic anterior uveitis and ocular toxoplasmosis20,21,22,23—suggest infectious uveitis also may influence a patient’s quality of life in some clinical settings. However, the effect of ocular syphilis on vision-related wellbeing and functioning has not been reported. In this study, we investigated quality of life in individuals who had been diagnosed with ocular syphilis and completed treatment at a tertiary referral uveitis clinic in Brazil, using a validated, interview-based Portuguese language version of the widely utilized visual function questionnaire developed by the US National Eye Institute24. Specifically, we aimed to identify clinical characteristics of ocular syphilis that were associated with reduced quality of life.
Methods
Identification of study participants
This cross-sectional study was conducted at the Uveitis Clinic of Ribeirão Preto General Hospital, representing the only tertiary uveitis service in the region of Ribeirão Preto, São Paulo, Brazil, and serving approximately 1.7 million citizens. The study was approved by the Ethics Committee of Human Research at Ribeirão Preto General Hospital (approval number: 57349616.1.0000.5440), and the research was performed in accordance with the Declaration of Helsinki.
Adults aged 18 years or older, who had been diagnosed with and treated for ocular syphilis between January 2000 and April 2019, were identified by a search of hospital medical records, and contacted by telephone call or approached during a clinic attendance with an invitation to participate in the study. Subjects were enrolled in the study, and interviewed and examined, between August 2016 and April 2019. Individuals who could not be contacted or did not agree to participate were not included, and written informed consent was obtained for all participants.
The diagnosis of ocular syphilis was based on a documented history of ocular inflammation that resolved following intravenous treatment with aqueous penicillin G or ceftriaxone, with serological confirmation of syphilis by a reactive treponemal (fluorescent treponemal antibody absorption, FTA-abs) test and a non-treponemal (venereal disease research laboratory, VDRL]) test with a titer 1:2 or higher.
Collection of clinical data
The medical record was used to verify the diagnosis of ocular syphilis, and to confirm that the ocular inflammation had been inactive for at least 3 months prior to enrolment in the study. Additional data that were collected from the medical record included: self-identified gender, age at presentation with uveitis, human immunodeficiency virus (HIV) serology, type of ocular inflammation, type of uveitis, laterality of uveitis, presenting best-corrected visual acuity (BCVA), and treatment regimen. The type of uveitis was defined anatomically, using the Standardization of Uveitis Nomenclature (SUN) classification25. Isolated papillitis was classified as posterior uveitis for the purposes of the statistical analysis.
A comprehensive ophthalmic evaluation was performed, including measurement of BCVA, slit-lamp examination and indirect ophthalmoscopy with pupillary dilation. The BCVA of the better seeing eye was used for evaluating associations between clinical characteristics and vision-related quality of life, since this is strongly correlated with self-reported visual disability26. A BCVA of 20/50 or worse defined vision loss.
Vision-related quality of life assessment
The 25-item National Eye Institute Visual Function Questionnaire (NEI VFQ-25) was used to assess the patient-perceived vision-related quality of life24. A validated Portuguese language interview format version was employed; this had 39 total items, including 14 additional items from the original 51-item NEI-VFQ27.
The NEI VFQ-25 generates 12 subscales: general health, general vision, ocular pain, near activities, distance activities, vision-specific social functioning, vision-specific mental health, vision-specific role difficulties, vision-specific dependency, driving, color vision, and peripheral vision. Higher scores indicate a better quality of life; lower scores indicate a worse quality of life. Following the standard instructions for analysis of the NEI VFQ-25, scores for each item were converted to a 0 to 100 scale, with the lowest and the highest possible scores set at 0 and 100 points, respectively. Items within each subscale were averaged to give 12 subscale scores. A composite score was calculated by averaging all subscale scores with the exception of the general health score.
Statistical analysis
Statistical analysis was performed using Microsoft Excel 16.64 for Mac (Microsoft Corporation, Redmond, WA) and SPSS 16.0 for Windows (SPSS Incorporated, Chicago, IL). Continuous variables were expressed as the mean ± standard deviation (SD). Categorical variables were expressed as absolute and relative frequencies. Student’s t-test, Fisher’s exact test, Fisher-Freeman-Halton’s exact test, and Mann–Whitney U test were used to compare groups. A p-value of less than 0.05 was taken to indicate a statistically significant difference between groups.
Results
Of a total of 50 eligible individuals, 32 patients (8 women including one transgender person, and 24 men, ranging in age from 23 to 80 years) participated in this study. Five patients (15.6%) were infected with HIV; CD4 counts were 317 to 994 cells/mm3 and viral loads were undetectable to 58.685 copies/ml in three subjects, and not available for two subjects. The most common diagnosis was posterior uveitis (n = 20 patients, 62.5%), followed by panuveitis (n = 11 patients, 34.4%) and intermediate uveitis (n = 1 patient, 3.1%). No patients had isolated anterior uveitis, or an ocular surface inflammatory disease including scleritis. Nineteen patients (59.4%) had bilateral uveitis: of the 51 affected eyes, there were 34 (66.%) with posterior uveitis (66.7%), 15 with panuveitis (29.4%) and 2 with intermediate uveitis (3.9%). More patients were treated with intravenous penicillin G than intravenous ceftriaxone (n = 19, 59.4% versus n = 13, 40.6%), and adjunctive corticosteroid treatment was commonly prescribed (n = 21, 65.6%).
Table 1 presents clinical characteristics of the study participants, and shows results of the analysis of vision outcomes. Across the 32 patients, initial BCVA in the better seeing eye was better than 20/50 in 19 (59.4%), 20/50–20/150 in 6 (18.7%), and 20/200 or worse in 7 (21.9%). Following treatment and after complete resolution of the ocular inflammation, BCVA in that eye of the patient was better than 20/50 in 26 (81.3%), 20/50–20/150 in 5 (15.6%), and 20/200 or worse in 1 (3.1%). None of the clinical characteristics that were recorded (including demographics, type of uveitis, presenting BCVA, and treatment) were significantly associated with the final BCVA in the better-seeing eye (p > 0.05).
Mean composite score and subscale scores of the NEI VFQ-25 completed by the 32 patients are presented in Table 2. Taking the patients as a total, mean composite score was 75.5 ± 19.8, and mean scores for the 12 subscales varied from lows of 59.1 ± 39.6 (driving) and 60.9 ± 24.5 (mental health), to highs of 84.8 ± 21.8 (ocular) and 89.1 ± 21.0 (color vision).
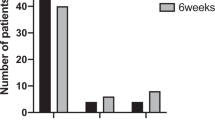
In comparison to patients with good final BCVA, those with a final BCVA of 20/50 or worse had significantly lower mean scores for general vision, near activities, distance activities, social functioning, mental health, role difficulties, and dependency subscales, as well as a significantly lower mean composite score (p < 0.05). For a subset of these subscales (distance activities, social functioning, mental health, and role difficulties), lower mean scores were also significantly associated with an initial BCVA of 20/50 or worse. In comparison to younger adults (40 years and less), middle and older aged adults (over 40 years) had significantly reduced scores for near activities, distance activities, social functioning, mental health, role difficulties, dependency, driving, and peripheral vision subscales, plus a significantly lower mean composite score (p < 0.05). Other clinical characteristics (including demographics, type of uveitis, and treatment) did not significantly influence mean composite score or subscale scores (p > 0.05). The differences in scores overall and by age, and initial and final BCVA are illustrated in Fig. 1.
Mean subscale and composite scores on the National Eye Institute Visual Function Questionnaire-25 for patients with ocular syphilis (n = 32 individuals) overall and by age, and initial (presenting) and final (post-resolution) best-corrected visual acuities. Plots were created in Microsoft Excel 16.64 for Mac.
Discussion
Non-infectious uveitis has been shown to reduce an individual’s quality of life substantially9,10,11,12,13,14,15,16,17. There is less clarity around infectious uveitis: three studies of ocular toxoplasmosis drew different conclusions21,22,23, and a study of herpetic anterior uveitis showed a modest effect20. We assessed quality of life in a group of 32 patients who had recovered from ocular syphilis—manifested as intermediate, posterior or pan-uveitis—following standard treatment with antibiotics, supplemented with corticosteroid in approximately two-thirds. We found that middle and older aged adults, over 40 years when diagnosed and treated, experienced a lower overall quality of life than younger adults. Persons with reduced BCVA, reading 20/50 or worse after resolution of the uveitis, also reported reduced composite quality of life in comparison to those without vision loss.
Ocular syphilis has a readily available and inexpensive, safe and effective treatment that cures the condition5. On that basis, one might expect no impact on quality of life following treatment. However, our patients had a low mean composite scores (75.5 ± 19.8) when compared with the reference group in the original description of the NEI VFQ-25 (93.0)24, as well as groups of middle-aged and older Latinos without visual impairment (86.3 for English-speaking, 85.1 for Spanish-speaking)28. However, there may be a delay in reaching the diagnosis of ocular syphilis, and this has been cited as a reasons for poor outcome29. Indeed, visual recovery is incomplete in up to one-third of eyes, and a range of complications—cataract, glaucoma, macular pucker, optic atrophy and rhegmatogenous retinal detachment—may result in long-term visual disability3,4,30.
We found associations between quality of life scores across seven subscales (including general vision, near activities, and distance activities) in addition to the composite score for BCVA after resolution, and across four subscales (also including general vision, near activities, and distance activities), but not the composite for presenting BCVA. Patients with initial and final BCVA of 20/50 or worse had lower scores regardless of uveitis type and treatment. The same association between BCVA and scores on the NEI VFQ-25 has been reported in studies of other types of uveitis13,19,31. It was also noted during development of the questionnaire, with similar correlation for better and worse seeing eyes, especially in subscales related to general vision, near vision, and distance vision24.
In patients with ocular syphilis, being aged over 40 years was statistically correlated with lower quality of life. A large cross-sectional study from Germany that included 619 working adults showed a clear age dependency of NEI VFQ-25 scores: the composite score decreased by approximately one point for every decade of life, for adults with and without eye diseases32. Lower near and distance activity subscales of visual functioning were observed in patients with Behçet uveitis aged 30 years or older16; and general vision, near activities, and role difficulties subscales were negatively correlated an age of 45 years or more in herpetic anterior uveitis20. However, the opposite result of no correlation between age and NEI VFQ-25-derived quality of life scores have also been reported for groups of patients with non-infectious uveitis11,12,18 and ocular toxoplasmosis22.
Limitations of our study include the collection of data at a tertiary referral center, and the inability to recruit all eligible patients, potentially introducing selection bias. Also, discrepancies in sample size may have influenced some results: although we did not observe associations between quality of life, and gender, HIV infection status and type of uveitis, less than 20% of our patients were HIV-positive, 25% were women, and the majority had posterior uveitis. We did not attempt to correlate quality of life scores with ocular complications given the small size of the patient group. Despite these limitations, our study provides unique data on the quality of life in patients who have suffered from ocular syphilis. Given the association we observed between BCVA and NEI VFQ-25 scores, plus previous observations that prompt recognition achieves better visual outcomes3,33,34, we speculate that early diagnosis and treatment may result in higher quality of life for individuals with ocular syphilis.
Data availability
All data generated or analyzed during this study are included in this published article (and its Supplementary Information files).
References
Gonzalez Fernandez, D., Nascimento, H., Nascimento, C., Muccioli, C. & Belfort, R. Jr. Uveitis in São Paulo, Brazil: 1053 new patients in 15 months. Ocul. Immunol. Inflamm. 25, 382–387 (2017).
Amaratunge, B. C., Camuglia, J. E. & Hall, A. J. Syphilitic uveitis: A review of clinical manifestations and treatment outcomes of syphilitic uveitis in human immunodeficiency virus-positive and negative patients. Clin. Exp. Ophthalmol. 38, 68–74 (2010).
Hoogewoud, F. et al. Prognostic factors in syphilitic uveitis. Ophthalmology 124, 1808–1816 (2017).
Furtado, J. M. et al. Clinical manifestations and ophthalmic outcomes of ocular syphilis at a time of re-emergence of the systemic infection. Sci. Rep. 8, 12071 (2018).
Furtado, J. M. et al. Ocular syphilis. Surv. Ophthalmol. 67, 440–462 (2022).
Centers for Disease Control and Prevention. Sexually Transmitted Infections Treatment Guidelines, 2021: Neurosyphilis, Ocular Syphilis, and Otosyphilis. https://www.cdc.gov/std/treatment-guidelines/neurosyphilis.htm (2021).
de Smet, M. D. et al. Understanding uveitis: The impact of research on visual outcomes. Prog. Retin. Eye Res. 30, 452–470 (2011).
Durrani, O. M. et al. Degree, duration, and causes of visual loss in uveitis. Br. J. Ophthalmol. 88, 1159–1162 (2004).
Miserocchi, E., Modorati, G., Mosconi, P., Colucci, A. & Bandello, F. Quality of life in patients with uveitis on chronic systemic immunosuppressive treatment. Ocul. Immunol. Inflamm. 18, 297–304 (2010).
Jalil, A., Yin, K., Coyle, L., Harper, R. & Jones, N. P. Vision-related quality of life and employment status in patients with uveitis of working age: A prospective study. Ocul. Immunol. Inflamm. 20, 262–265 (2012).
Gui, W. et al. Quality of life in patients with noninfectious uveitis treated with or without systemic anti-inflammatory therapy. Ocul. Immunol. Inflamm. 23, 135–143 (2015).
Arriola-Villalobos, P. et al. Vision-related quality of life in patients with non-infectious uveitis: A cross-sectional study. Ocul. Immunol. Inflamm. 26, 717–725 (2018).
Frick, K. D. et al. Associations among visual acuity and vision- and health-related quality of life among patients in the multicenter uveitis steroid treatment trial. Invest. Ophthalmol. Vis. Sci. 53, 1169–1176 (2012).
Naik, R. K. et al. Normative comparison of patient-reported outcomes in patients with noninfectious uveitis. JAMA Ophthalmol. 131, 219–225 (2013).
Missaka, R. F. B. G. et al. Self-reported quality of life in patients with long-standing Vogt–Koyanagi–Harada disease. Ocul. Immunol. Inflamm. 28, 409–420 (2020).
Onal, S., Savar, F., Akman, M. & Kazokoglu, H. Vision- and health-related quality of life in patients with Behçet uveitis. Arch. Ophthalmol. 128, 1265–1271 (2010).
Hoeksema, L. & Los, L. I. Vision-related quality of life in patients with inactive HLA-B27-associated-spectrum anterior uveitis. PLoS ONE 11, e0146956 (2016).
Hui, M. M. et al. Visual functioning and health-related quality-of-life are compromised in patients with uveitis. Ocul. Immunol. Inflamm. 25, 486–491 (2017).
Schiffman, R. M., Jacobsen, G. & Whitcup, S. M. Visual functioning and general health status in patients with uveitis. Arch. Ophthalmol. 119, 841–849 (2001).
Hoeksema, L. & Los, L. I. Vision-related quality of life in herpetic anterior uveitis patients. PLoS ONE 9, e85224 (2014).
de-la-Torre, A., González-López, G., Montoya-Gutiérrez, J. M., Marín-Arango, V. & Gómez-Marín, J. E. Quality of life assessment in ocular toxoplasmosis in a Colombian population. Ocul. Immunol. Inflamm. 19, 262–266 (2011).
Canamary, A. M. J. et al. Quality-of-life and psychosocial aspects in patients with ocular toxoplasmosis: A clinical study in a tertiary care hospital in Brazil. Ocul. Immunol. Inflamm. 28, 679–687 (2020).
Peyron, F. et al. Long-term impact of treated congenital toxoplasmosis on quality of life and visual performance. Pediatr. Infect. Dis. J. 30, 597–600 (2011).
Mangione, C. M. et al. Development of the 25-item National Eye Institute Visual Function Questionnaire. Arch. Ophthalmol. 119, 1050–1058 (2001).
Jabs, D. A., Nussenblatt, R. B., Rosenbaum, J. T. & Standardization of Uveitis Nomenclature (SUN) Working Group. Standardization of uveitis nomenclature for reporting clinical data. Results of the First International Workshop. Am. J. Ophthalmol. 140, 509–516 (2005).
Rubin, G. S., Muñoz, B., Bandeen-Roche, K. & West, S. K. Monocular versus binocular visual acuity as measures of vision impairment and predictors of visual disability. Invest. Ophthalmol. Vis. Sci. 41, 3327–3334 (2000).
Simão, L. M., Lana-Peixoto, M. A., Araújo, C. R., Moreira, M. A. & Teixeira, A. L. The Brazilian version of the 25-Item National Eye Institute Visual Function Questionnaire: Translation, reliability and validity. Arq. Bras. Oftalmol. 71, 540–546 (2008).
Globe, D. et al. Psychometric performance of the NEI VFQ-25 in visually normal Latinos: The Los Angeles Latino Eye Study. Invest. Ophthalmol. Vis. Sci. 44, 1470–1478 (2003).
Oliver, G. F. et al. Current ophthalmology practice patterns for syphilitic uveitis. Br. J. Ophthalmol. 103, 1645–1649 (2019).
Moradi, A. et al. Clinical features and incidence rates of ocular complications in patients with ocular syphilis. Am. J. Ophthalmol. 159, 334-343.e1 (2015).
Murphy, C. C., Hughes, E. H., Frost, N. A. & Dick, A. D. Quality of life and visual function in patients with intermediate uveitis. Br. J. Ophthalmol. 89, 1161–1165 (2005).
Hirneiss, C. et al. The NEI VFQ-25 vision-related quality of life and prevalence of eye disease in a working population. Graefes Arch. Clin. Exp. Ophthalmol. 248, 85–92 (2010).
Bollemeijer, J. G. et al. Clinical manifestations and outcome of syphilitic uveitis. Invest. Ophthalmol. Vis. Sci. 57, 404–411 (2016).
Tsuboi, M. et al. Prognosis of ocular syphilis in patients infected with HIV in the antiretroviral therapy era. Sex. Transm. Infect. 92, 605–610 (2016).
Acknowledgements
The authors wish to thank Ms. Janet Matthews for administrative support in the preparation of this manuscript.
Funding
This study was supported by a grant from the Brazilian National Council for Scientific and Technological Development (CNPq: 310099/2021-8 to JMF). The funder had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Contributions
The study was planned by M.S.F.S., J.R.S. and J.M.F. Data were collected by M.S.F.S. and R.M. Data analyses were performed, and display items were prepared by T.E.A. The manuscript was drafted by M.S.F.S., and edited and critically revised by J.R.S. and J.M.F. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Silva, M.S.F., Arantes, T.E., Moreto, R. et al. Vision-related quality of life in patients treated for ocular syphilis. Sci Rep 13, 13413 (2023). https://doi.org/10.1038/s41598-023-40289-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-40289-0
- Springer Nature Limited