Abstract
Microendemicity, or the condition of some species having local ranges, is a relatively common pattern in nature. However, the factors that lead to this pattern are still largely unknown. Most studies addressing this issue tend to focus on extrinsic factors associated with microendemic distributions, such as environmental conditions, hypothesising a posteriori about underlying potential speciation mechanisms, linked or not to these conditions. Here, we use a multi-faceted approach mostly focusing on intrinsic factors instead, namely diversification dynamics and speciation modes in two endemic sibling genera of leaf beetles with microendemic distributions, Taophila and Tricholapita, in a microendemicity hotspot, New Caledonia. Results suggest that the diversification rate in this lineage slowed down through most of the Neogene and consistently with a protracted speciation model possibly combined with several ecological and environmental factors potentially adding rate-slowing effects through time. In turn, species accumulated following successive allopatric speciation cycles, possibly powered by marked geological and climatic changes in the region in the last 25 million years, with daughter species ranges uncorrelated with the time of speciation. In this case, microendemicity seems to reflect a mature state for the system, rather than a temporary condition for recent species, as suggested for many microendemic organisms.
Similar content being viewed by others
Introduction
Microendemicity or narrow endemism is a relative concept that qualifies the global range of a particular species as small1. The literature on biodiversity includes many examples of microendemic species, species with narrow geographic distributions often recognisable owing to adaptations to their relatively small habitats. The interest in these species typically arises from conservation concerns, since they are perceived as more vulnerable than related species with larger ranges to threats associated with climate change2, genetic impoverishment3, outcompetition by invasive species4, or habitat disturbance5, to mention the most obvious. Many groups of organisms have examples of microendemic species, and some seem particularly prone to exhibit this pattern, including palms, frogs, millipedes, scorpions, geckos, and others6,7,8,9,10. However, what makes this biodiversity pattern particularly intriguing is that there are geographic regions that tend to concentrate these microendemisms, regions showing high rates of microendemicity across different groups of organisms. This pattern has been typically associated with special characteristics of the region, including age, isolation, topography or climate11. For example, the Espinhaço range in Brasil and the neighbouring Cerrado and Atlantic forests could be considered important plant microendemicity hotspots in South America12,13,14. The Cape Region and the Drakensberg range in South Africa are also known for plant microendemicity, but several groups of animals reflect this pattern too15,16. However, the best examples of areas characterised by generalised microendemicity across taxonomic groups are two tropical, ancient continental islands: Madagascar and New Caledonia1,17. The origin of endemic diversity in these two islands followed different paths—old and vicariant in the case of Madagascar18, recent and probably following transmarine colonisations in the case of New Caledonia19, but both island biotas seemingly underwent high diversification in situ10,20,21,22,23,24,25. Therefore, studies focusing on these areas have the potential to unveil general mechanisms responsible for geographic patterns of microendemicity.
Not much is known about the processes that underlie the pattern of regional microendemicity. For example11, proposed a mechanistic model entirely relying on predicted past climatic shifts and the topography and hydrology of Madagascar to explain species distribution patterns. A similar argument, combining climatic cycles and geographic factors, was also invoked to explain the accumulation of narrow endemics in the Mediterranean26. However, the simultaneity of geographic and taxonomic elements suggests that a more generalised answer may lie in the concourse of both extrinsic factors related to the physical and historical characteristics of the region where microendemicity is common, and intrinsic factors related to the mode of diversification of the groups that tend to exhibit microendemicity27,28.
Among extrinsic factors, two apparently opposing major theories are generally considered, one relating these patterns to geological and climatic habitat stability14,29,30, and one stressing changes in climate, orography and opportunities for dispersal as the engine for allopatric speciation11,19,26,31. However, these seemingly opposed theories can be reconciled in models that take scale into account, allowing for the persistence of lineages originated by small-scale allopatry in larger regions of relative stability29. Independent of habitat stability, some models consider species ranges a time-dependent dynamic attribute, so that narrow ranges are interpreted as a temporary characteristic of younger species32. Concerning lineage-specific or intrinsic factors, low dispersal ability and/or specialisation and associated phenotypic traits are typically invoked as explanations for microendemicity of species originated in allopatry33,34,35,36. Regardless of the actual extrinsic or intrinsic factors leading to speciation with an effect on species ranges, most microendemicity studies stress a correlation between microendemic species and allopatric ranges, deducing that allopatric speciation is a dominant explanation for the origin of microendemic biodiversity29,32,37,38. These same intrinsic and to some degree also extrinsic factors influence how organisms diversify through time39,40. Thus, the study of the tempo and mode of diversification is an important analytical tool to unravel the mechanisms leading to particular biogeographic patterns, including microendemicity7,28,29,36,41,42.
There have been some important recent contributions describing the diversity and distribution of the hyperdiverse Eumolpinae leaf beetles in one of the main microendemicity hotspots, New Caledonia22. Resulting from the improved taxonomic knowledge about these beetles, it was uncovered a generalised pattern of microendemicity for most species, generally known from a single or few geographically close localities, within a single valley or elevation43,44,45,46,47,48. These findings, concordant with reports for many other arthropod groups in the island1, prompted us to investigate the evolutionary mechanisms that could explain such a frequent outcome. Here, we will study these mechanisms in two endemic sibling genera of New Caledonian leaf beetles with similar distribution patterns in New Caledonia, Taophila Heller and Tricholapita Gómez-Zurita and Cardoso. Both genera consist of many species, most of them showing microendemic distributions47,48, and our main aim is establishing the general evolutionary process that led to the observed distribution patterns. Current knowledge on diversity and distribution patterns and the lack of temporal and spatial resolution about particular events that may or may not correlate with these patterns hinder addressing the general question why microendemicity evolves. However, we can shed some light on how it may originate in a particular group and region, as a first step towards a much-needed synthesis on this topic. Our strategy will involve the analysis of diversification in the clade of interest and an attempt to fit a model of speciation to the diversification process, in order to assess the degree in which geographic distributions and allopatry could be important factors responsible for the microendemicity patterns of these two New Caledonian leaf beetle genera.
Results
Phylogenetic backbone for diversification analyses
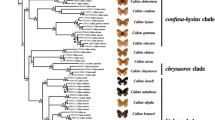
A clock-constrained Bayesian tree based on cox1 and rrnS data and calibrated based on the results of previous studies was obtained for 30 species of Taophila and Tricholapita. The tree was compatible with phylogenies published separately for each genus (Fig. 1), showing their separation during the Oligocene (29.09 Ma; 95HPD = 20.18–37.53 Ma) and their crown diversifications in the Oligocene–Miocene transition: 24.48 Ma (95HPD = 16.03–31.84 Ma) for Taophila, and 22.23 Ma (95HPD = 13.92–30.04 Ma) for Tricholapita.
Uncorrelated-lognormal clock-constrained tree of New Caledonian Eumolpinae, with focal groups Taophila and Tricholapita highlighted. Moderately (posterior probability [PP] = 0.50–0.95) and highly (PP = 0.95–1.00) supported nodes are labelled and the HPD age intervals of the latter represented by bars for each of the nodes. Terminals colour-coded according to accompanying legend based on known species distributions, where “microendemic” refers to species known from a single or geographically close localities; “reduced range” to species found in a regional set of localities; and “widespread” to species found across Grande Terre.
Diversification model of Taophila and Tricholapita
The analysis of global changes in evolutionary rates using BAMM showed that data fitted consistently a model of progressive slow-down of net diversification rates, owing to a gradual reduction in the rate of speciation against a relatively constant estimate of extinction rate (Figs. 2A and S2). Mean speciation and extinction rates were λ = 0.120703 and µ = 0.065015, yielding a mean net diversification rate of 0.055688 events/Ma.
(A) Diversification-through-time and lineage-through-time (LTT) trajectories inferred using BAMM104 for the lineage of sibling genera Taophila and Tricholapita. Net diversification (red) decomposed in speciation (green) and extinction (yellow) components. Visual information on the trajectory of confidence intervals through time available in Fig. S1. (B) Branch-specific speciation rates in the evolution of Taophila and Tricholapita, with changes in tendency indicated by white arrowheads as obtained with the ClaDS model109. (Pictures obtained from iNaturalist and used with authors' permission: Taophila, Pierre-Louis Stenger; Tricholapita, Damien Brouste).
Testing for shifts in diversification rates produced very low Bayes factor values (BF = 0.1922) for k = 1, and increasing values of k further decreased this BF value, approaching it to 0. Such low BF values indicate rejection of the alternative over the null hypothesis or, considering the relatively small size of the dataset, at least insufficient evidence to reject the null hypothesis of no shifts in diversification rate for the whole dataset. Alternatively, the analysis of local shifts in diversification rates with CLaDS co-estimated the values of initial speciation rate, λ0 = − 2.06214; factor controlling freedom of change of daughter speciation rates, σ = 0.237885; factor controlling increasing or decreasing trends at speciation, α = 0.919254; and constant turnover, ε = 0.128263. Accordingly, the mean relative change in rate at speciation in the ingroup phylogeny was m = 0.945635. For m < 1, data is consistent with a scenario of low heterogeneity of generally decreasing diversification rates, indicating that the chance of speciation diminishes, even if weakly, as diversification proceeds (as opposed to m > 1, indicating an acceleration in speciation). Figure 2B shows this trend graphically, only reversed in the clade of Tricholapita mars and relatives, and the clade including the most recent diversification of Taophila, around the species Ta. draco and Ta. davincii.
Up to twenty-one diversification models, including constant-rate models, time-dependent, temperature-dependent, diversity-dependent and protracted speciation models were tested for the data (Table 1). The model receiving the best AICc value was protracted speciation (PS21), with relatively large AIC-based distances (8.52 < ΔAICc < 14.65) compared with other models, irrespective of the correlated factor, i.e. time, temperature or diversity. The best-fitting model proved adequate for the phylogeny, with tree-shape metrics derived from simulations under this model statistically identical to metrics obtained from the empirical tree (Fig. S3): principal Eigenvalue = 3002.55 (P = 0.552); asymmetry = − 0.13967 (P = 0.926); and peakedness = 2.109148 (P = 0.980).
Speciation modes in Taophila and Tricholapita
The ingroup mode of speciation was tested explicitly by incorporating data on the phylogeny, age of lineages, and range overlap between species. Standard age-range correlation analyses in Taophila and Tricholapita were only significant for the intercept (P = 0.0184) and the slope (P = 0.0185) of the correlation for point distribution ranges in Taophila (and marginally significant for the slope [P = 0.0799] of buffer ranges in Taophila). Otherwise, the observed distribution of slopes and intercepts were not different from random (Fig. S4). In turn, the PGLS regression between log-transformed species ages and ranges, implementing Pagel’s λ = − 0.0102, was not significant (slope = − 0.056, P = 0.630; Fig. S5), rejecting the hypothesis of younger species having smaller ranges. ARC results were also one of the summary metrics among fourteen included in the process-based DReaD simulation model of49 and Table S3 shows the estimates of summary statistics by DReaD for both genera. The accuracy of model classifications was statistically significant (κ = 0.5752, P < < 0.001), indicating that the obtained classification was better than random. In these classifications, vicariant speciation dominated in Taophila (94.71%), followed by founder (4.05%) and mixed (1.21%) models. In Tricholapita, vicariant speciation was almost exclusive (98.94%), with a mixed model as the second alternative (1.06%).
Discussion
We investigated the diversification dynamics and the geography of speciation of New Caledonian rainforest leaf beetle sibling genera Taophila and Tricholapita, characterised by mosaic-like microendemic distributions of most species. This pattern characterises many groups of organisms in New Caledonia and elsewhere, but we lack a general understanding of the conditions that lead to its development. Unveiling the mechanisms that result in these spatial patterns, and here we focused specifically on intrinsic evolutionary factors, may be crucial to understanding how microendemic diversity is generated, and more importantly how narrow species ranges are maintained, which is relevant for conservation50,51. The small size of our dataset, imposed by the limited diversity of the group of study, together with lack of fossil data, recommends much caution when interpreting results of diversification analyses owing to statistical power limitations of current analytical approaches, among others52,53,54. Thus, echoing the assessment by55, we only consider net diversification dynamics and avoid interpretations about speciation and extinction rates.
A suitable summary for the evolution of the Taophila and Tricholapita lineage generally fits a scenario of very small decline in diversification rate through time and of allopatric speciation as the dominant process explaining its diversity. Regardless of the net speciation/extinction inferred values, the diversification trend for this lineage is suggestive a priori of a reduction in diversification rate. The progressive slowdown in diversification rates is a common evolutionary trend56,57,58,59,60, and it has been reported for several organisms in New Caledonia61, including the entire diversification of Eumolpinae22. There are several accepted evolutionary scenarios that could lead to a reduction in diversification rates59, which fall into three categories, including (i) diversity-dependent, (ii) time-dependent, and (iii) protracted speciation, the latter implying that species are not immediately recognisable as separate genetic entities at the tips of a phylogeny62. Data for Taophila and Tricholapita were consistent with the latter, indicating that if a rate slowdown truly describes the diversification of this lineage, as hinted with different methods (Table 1; BAMM Figs. 2 and S1 and CLaDS Fig. S6), it is most likely related to topological conditioning of gene trees, and to the prolonged nature of the speciation stages rather than an actual trend in the diversification process. Protracted speciation implies that terminal branch lengths in a species tree may be overestimated owing to potential incipient species not being considered in the tree63. Connecting this evolutionary pattern with the diversification of a group of organisms with microendemic ranges can be difficult, since microevolutionary processes leading to speciation in large, widespread populations are expected to be rare in this system. On the other hand, limited ranges and the logical difficulties for local differentiation within a small range could potentially lead to a phylogenetic pattern consistent with protracted speciation, especially if we take into account the apparent independence of species age with their range size, implying that the lack of microevolutionary differentiation may be persistent in time. For the sake of discussion, consistent failure to sample sister species of represented lineages in the phylogeny may yield longer than average terminal branches too, but this bias seems unlikely considering our recent taxonomic revisions46,47,48. Fitting the protracted speciation model may be more suitable for datasets based on population data and not species trees, and perhaps larger datasets where estimation of parameters may be less compromised. However, in the case of microendemic insect species with ability to disperse, effective panmixia and the absence of local genetic differentiation may be common, limiting the potential of lineage-based diversification analyses. Reduced, sometimes absent mtDNA haplotype diversity in the case of Taophila48 and Tricholapita47 would support this view.
The other correlative models considering time, temperature or diversity, as well as null, constant rate models, proved clearly inferior to protracted speciation for this dataset. These other models loosely clustered according to their number of free parameters, suggesting that the result of model selection could be influenced by the lack of penalty for estimating some of the parameters, e.g. extinction. However, the overall best fit of protracted speciation to the dataset, particularly with the previous considerations about microendemic distributions, does not imply that the other processes do not operate on this system, but possibly that there are statistical limitations to recognise their effects or that their effects do not lead to rate slowdowns consistently across the phylogeny63. Moreover, disentangling these effects can be challenging, even for larger datasets64. Time-dependent diversification is often connected to environmental change59, for example through a correlation between declines in speciation and gradual cooling of global temperature65, or considering the effect of past climatic stability on niche partitioning60,66. To complicate things even further, environmental effects, or more specifically the influence of temperature can potentially mimic the outcome of diversity-dependent processes too60,64,67,68.
There are multiple ways in which these interconnected processes can exert non-directed or rate-declining influence on the diversification of New Caledonian Eumolpinae, also contributing to reduction of ranges. Global cooling and episodic climatic events, with periods of aridification that reduced the primary forest in Grande Terre, may have reduced suitable niches and species ranges69,70,71,72. Similarly, the geomorphological evolution of Grande Terre transitioned from a period of relative intensity during the mid-Oligocene73,74 to relative geological stability, dominated by erosion and planation, with an exceptionally active period during the Pleistocene74,75. This geological transition, concurrent with the evolution of Taophila and Tricholapita, could represent a trend from higher to lower topological and ecological complexity, potentially correlated with a reduction in diversification rates. The idea of a time-dependent process in New Caledonia somewhat associated to niche-filling, i.e. successive colonisation and preemptive occupation of empty niches56,57,59,76,77 further constraining the occupation of space, receives some support from the rate-decline exceptions of the fine-grain CLaDS analysis of changes in diversification rates across the phylogeny. One in particular affects the Tricholapita clade including the species Tr. reidi-Tr. oceanica (see Fig. 2B). This derived group of Tricholapita colonised and remained restricted to humid forests in southern parts of Grande Terre characterised by ultramafic soils, soils with high metallic content and consequently highly specialised vegetation47,78. Both host-plant choice and the intimate association of subterranean Eumolpinae larvae with the soil make it plausible that this lineage colonised and underwent an irreversible adaptation to ultramafic soils too. Colonising and exploiting a new, previously unoccupied niche could have released diversity-dependent diversification constraints and trigger a secondary wave of diversification and speciation in this group, with the successive fragmentation of parental ranges leading to current microendemic distributions.
The previous fundamentally ecological rules only explain in part the occupation of space, but not the drivers of speciation in these genera. Knowledge about the ecology of the species of Taophila and Tricholapita is very limited, aside from field observations of some species associated with ferns and DNA-based inferences on potential host plants in different botanical families43. We cannot rule out entirely an ecological driver for the origin of new species in these genera, for example by specialisation to different host plants. However, allopatric distributions of most species and documented cases of sympatry only for species that built enough phylogenetic and evolutionary divergence48 strongly suggest that some form of geographic speciation is at play for these genera59,79.
Disentangling the processes leading to speciation is at the core of many evolutionary studies80, but in the case of geographic speciation, the changing nature of species ranges and epistemological problems in establishing ancestral species ranges have jeopardised the reliability and statistical significance of the inferred processes81,82. Nonetheless, the interest in the geography of speciation spurred the development of several analytical approaches combining distribution and phylogenetic data81,83,84,85,86. Recently49, proposed a simulation and model-selection approach designed to infer speciation modes by simultaneously analysing multiple statistics representing different aspects of the interplay between speciation, phylogeny and current species ranges, typically examined individually in most other methods. Using this approach, vicariant speciation stood out as the dominant inference for Tricholapita and Taophila, the latter also with secondary signs of dispersal speciation. Allopatric speciation is the main explanation in most cases of microendemic distributions37,87,88, and it has been repeatedly invoked as the most likely speciation mechanism for New Caledonian microendemic species32,61. However, in previous studies, this process was considered an ad hoc hypothesis to explain certain patterns, and ours would be the first where this process is inferred analytically a priori for New Caledonian microendemic organisms. Nonetheless, invoking allopatric speciation as the mechanism generating microendemic diversity has theoretical requirements that need to be considered to provide a satisfactory explanation for the observed pattern37. The mechanism requires that the range of widespread ancestral species be fragmented by vicariance, by the formation of dispersal barriers, effectively isolating daughter populations89.
The ragged orography of Grande Terre was formed through successive cycles of planation during the Miocene and as recently as the pre-Pliocene, when glacio-eustatic processes might have been involved in considerable land tilting and uplift, followed by deep river erosion and valley formation73,74. These processes were contemporaneous with the diversification of Taophila and Tricholapita and in the latter the mechanism is compatible with the vicariance succession hypothesised for basal splits in the genus47. They also offer ample opportunity for cycles of species range expansions and successive range-reduction vicariance events in line with the evolution of Taophila or derived splits in both genera48. In support of this idea, there are some species in these genera that currently show relatively large ranges and also some phylogeographic evidence suggesting they have the potential for range expansion, as deduced for T. subsericea along the northern slopes of Massif du Panié47,48. In turn, elevation gradients favour habitat heterogeneity and local insular environments which, in combination with regional climatic changes during the past 25 Ma, could have generated the conditions for successive cycles of habitat fragmentation and contributed to subsequent allopatric speciation in many organisms adapted to the rainforest. Specifically, the late Miocene represented a relatively warm period within the general cooling trend that characterised most of the Cenozoic, and responsible for dramatic changes in ecosystems globally, usually resulting in cycles of aridification in lower latitudes65. In New Caledonia, it is assumed that aridification fragmented and contracted the humid forest in refugia available thanks to landscape heterogeneity, either higher elevations or areas of microclimatic suitability69,70,71,72. Climate-driven habitat fragmentation could contribute additional vicariance events also in periods of geological stability and establish conditions that could favour speciation, including range contractions and reduction in population size90,91,92. Although a marginal inference from our analyses, speciation following dispersal cannot be entirely discarded at least in the case of the genus Taophila, implying that some ancestral species could have expanded their range across preexisting barriers with subsequent speciation.
An alternation during most of the Late Cenozoic of phases of relief formation and/or habitat fragmentation with periods dominated by erosion and/or habitat expansion in New Caledonia could explain in part the cycles of species range expansions, isolation of populations through vicariance and eventual speciation in allopatry with ever contracting ranges (Fig. 3a). But this alone is not sufficient to explain the accumulation of species with reduced ranges, and the concourse of environmental effects may have been required as well. The interplay of geological and environmental drivers can be hypothesised based on the apparent match of peaks of cladogenesis following dry periods, in turn responsible for the fragmentation of New Caledonian rainforests (Fig. 3b). The species surviving these geological and climatic cycles could have a limited potential for range expansions because of neighbouring species through some form of density-dependent processes, such as competitive exclusion93,94, perhaps reinforced by other negative biotic interactions, including reproductive interference95. This model would explain the gradual build-up of species diversity attaining microendemic distributions through the combined effects of isolation and negative biotic interactions. The idea that microendemicity reflects the maturity of a system approaching carrying capacity, contrasts with alternative hypotheses where microendemicity is associated with initial stages of speciation prior to species range expansions32. Indeed, most sister species pairs in our analysis have ranges of different size (Figs. 1 and S7) and the lack of a significant association between species age and range also argue against a pure effect of time alone on these ranges. Moreover, the model does not conflict with the possibility of sympatry, a pattern observed in several instances of Taophila and Tricholapita, which could have been attained secondarily when phenotypic and ecological differences between neighbouring species relaxed competition and other negative interactions. Secondary sympatry for these genera seems particularly frequent in few high elevation spots throughout the island, such as Aoupinié or Mandjélia47,48. This distribution pattern could be expected if these areas acted as climatic refugia during the Pleistocene and following the contraction of the rainforest as proposed for other organisms32,71.
(a) Cartoon summary of the main spatial processes explained in the main text conducing to gradual occupation of habitat through successive speciation events and confinement to microendemic ranges in a scenario with reduced effects of extinction. (b) Histogram of the succession and variation in the number of phylogenetic splits along the evolution of Taophila and Tricholapita since the Oligocene, with gray horizontal bars showing confidence time intervals for these splits (shown in no particular order), together with contemporaneous palaeoenvironmental changes in temperature and precipitation regimes (pale yellow intervals and black bars on top) and major chronological periods of geomorphic evolution of Grande Terre (white, blue and red intervals), both based on the synthetic model of74.
The study of diversification is challenging for small clades, lacking the analytical power to provide definitive answers. However, the knowledge gained about possible modes of diversification in these cases can be at least and with every caution a starting point towards informed hypotheses for a better understanding of their diversity55. The study of diversification and speciation of the New Caledonian radiation of Taophila and Tricholapita revealed there is nothing extraordinary or unexpected about the putative intrinsic processes that led to their current microendemic species distributions. But their analytical characterisation is essential to incorporate these processes as ad hoc elements of an eventual explanation of spatial patterns. Our full understanding of the causes of microendemicity shall benefit from integrating micro- and macroevolutionary processes and explicit knowledge about both intrinsic and extrinsic factors, as illustrated by geological and climatic evolution matching cladogenesis in this group, how speciation occurred and what was the fate of the daughter species relative to inherited area and environmental conditions.
Methods
Study groups
Studies on microendemicity can be biased because of sampling artifacts1 and incorrect taxonomies96. Here, we selected two endemic sibling genera of New Caledonian Eumolpinae where these potential drawbacks are minimised: Taophila and Tricholapita. These genera were revised recently using material from our 2007–2008 research collection (IBB-CSIC, Spain) and Prof. M. Wanat's collection (Museum of Natural History of Wrocław University, Poland), obtained through intensive sampling of phytophagous beetles in New Caledonia between 2004 and 2010. Establishing species ranges mainly depends on sampling effort97,98,99. Admittedly, exploring new localities in New Caledonia, where much of the island’s wilderness is inaccessible, could potentially refine our knowledge both on species diversity and distributions. However, our current ideas about species ranges and boundaries seem consistent with information for other groups100, representing a relatively solid starting point to start questioning about the relationship between species ranges and evolution. Taophila consists of 21 species, 18 of which show reduced, mostly allopatric ranges, 14 of them known from a single locality48. In turn, Tricholapita includes twelve species, all with restricted and usually allopatric ranges and seven known from a single locality46,47. Relevant genetic data were available for all species of Tricholapita and 18 out of 21 species of Taophila (Table S1).
Clock-constrained phylogeny
We used mtDNA gene trees based on partial cytochrome c oxidase subunit 1 (cox1) and small rRNA subunit (rrnS) genes as a proxy to Taophila and Tricholapita species phylogenies (most source material were dry collection specimens, which failed to yield nuclear gene PCR products). In these genera, mtDNA data alone were reliable for species delimitation and relationships, with nearly perfect matches between taxa and species monophyly deduced from multiple individuals, a single instance of incongruence due to interspecific hybridisation between locally sympatric species48, and species phylogenetic clusters compatible with morphological species groups47,48. Representative sequences from one individual per species as well as several outgroups (Table S1; Fig. 1) were obtained from published studies22,43,46,47,48. Sequences were aligned for each marker separately using the G-INS-i algorithm in MAFFT 7.3101, and concatenated in a single alignment to infer a time-constrained ultrametric tree in BEAST 1.8.4102. Data were analysed using unlinked103 evolutionary models for each marker, with site-heterogeneity in the rates of evolution, including a proportion of invariable sites, nucleotide frequencies estimated from the sequences, a Birth–Death speciation tree model and uncorrelated lognormal relaxed clocks for each marker. The tree was time-calibrated using two calibration points derived from clade ages inferred by22, who used the same phylogenetic markers and calibration hypotheses based on either marker-specific rates or on biogeographic considerations that yielded highly consistent age estimates. Specifically, we used rate-based inferences for the stem age of Tricholapita, dated at 32.6 Ma (95% Highest Posterior Density [95HPD] = 26.2–39.9 Ma), and the crown age of “Clade I” in22, including representatives of the groups of Ta. sagittarii and Ta. subsericea48, dated at 26.3 Ma (95HPD = 10.9–31.9 Ma). These two calibration nodes were modelled to cover the respective 95HPD confidence intervals as normal distributions with means 33.0 and 22.0, and standard deviations of 4.0 and 6.5, respectively. Bayesian inference used a MCMC search with 75 million generations, sampling trees and parameters every 7500 steps, and final estimation of parameters and maximum clade credibility tree were obtained after discarding 10% of the initial results.
Diversification analyses
The time-constrained tree topology was used to study different aspects of diversification in Taophila and Tricholapita. Potential heterogeneity in evolutionary rates through time was studied based on Bayesian Analysis of Macroevolutionary Mixtures (BAMM104). BAMM analyses were run for the whole ingroup dataset (n = 30) and the speciation-extinction test consisted of 20 million simulation steps, sampling data every 1000th generation, estimating taxonomic coverage as the fraction of available taxa from the known species catalogues, with additional priors (Table S2) obtained using the function setBAMMpriors in the R package BAMMTools 2.0.2105. Global rate shifts were tested with BAMM considering models with up to k = 5 shifts, using Bayes Factors to compare against a null model without shifts in net diversification rate. These analyses make inferences about the diversification process considering global rates and major shifts across phylogenies, relying on strong assumptions about species representation in the sample106. We alternatively implemented a Bayesian approach that analyses branch-specific changes in diversification rates at each speciation event under a birth–death diversification process, the ClaDS model107, as implemented in the RPANDA 1.9 package108. This approach complements some of the sample assumptions of the standard BAMM approach by explicitly modelling hidden speciation events, e.g., events missing in the phylogeny either because of extinction or sampling issues60. The Julia language implementation of the ClaDS model109 used here considers a scenario with variable extinction rate across lineages modelled by a constant turnover parameter, proven superior to other potential alternatives, such as excluding extinction or considering a constant extinction rate across lineages107. Finally, we evaluated the fit of the deduced diversification process to different diversification models using a maximum-likelihood framework, testing for potential effects of biotic (e.g., competition) or abiotic (e.g., temperature) factors on this rate. Specifically, we tested 21 models (see Table 1 for details), including two constant-rate models (pure-birth, CR1; birth–death, CR2), eight models with rates varying through time (pure-birth, TD3–4; birth–death, TD5–10), eight temperature-dependent models (pure-birth, ED11–12; birth–death, ED13–18), two diversity-dependent models with constant extinction (DD19–20); and protracted speciation (PS21). Time- and temperature-dependent models were fitted using RPANDA 1.9108, integrating palaeotemperature data derived from δ18O measurements from110 equations; diversity-dependent models using DDD 4.4111; and protracted speciation using ProSSE functions112 as implemented in the package diversitree 0.9.16113, which is the most robust current approach to reduce the error boundary in parameter estimates for this model in species trees when lineage-level phylogenies are not available112. Best-fitting models were selected based on differences of AICc estimates. The relatively small size of the dataset could impair the ability of these tests to accurately discern alternative diversification models39. Therefore, as a safeguard for model selection, we performed adequacy tests of best-fitting models using the R package BoskR114, which assesses shape similarity between empirical trees and a set of trees simulated under the tested model based on metrics derived from the tree Laplacian spectrum, interpreting uncorrected (to avoid inflation of type I errors115) significant differences as model inadequacy116. The simulation of protracted speciation is not implemented in BoskR, thus 1000 trees were simulated using the tree.prosse function of the diversitree package, fixing the number of tips, maximum root age and diversification rate to match the values deduced from the original tree, and subsequently used as input for the BoskR analysis.
Speciation analyses
Microendemicity is a biodiversity pattern defined by geographic distributions, and may or may not be the result of the actual speciation process that generated the microendemisms. We explored explicitly the geography of speciation in Taophila and Tricholapita using standard age-range correlation (ARC) analyses, specifically testing the range overlap between species81 using the enmtools.aoc function in the R package ENMTools 1.0.1117 and two different measures of species ranges, considering point localities, as proposed by83, or fixed-radius buffered ranges. Specifically, these ranges considered circular buffers with radius = 8 km around data points (following, e.g.,32) obtained from geographic coordinates of occurrence data46,47,48, calculated and transformed into raster data on a 0.5-min resolution grid with ENMTools. We analysed another temporal aspect of species distributions, addressing the question whether recent speciation events could be associated to smaller ranges. This was tested through phylogenetic generalised least squares (PGLS) regression between terminal branch ages in the dated phylogeny and species ranges as deduced from the raster data. The PGLS regression was studied using the gls function of the nlme package in R118, based on an estimation of Pagel’s119 lambda. A preliminary analysis of the original variables showed a certain degree of positive skewness (Fig. S1), thus final analyses used the logarithmic transformation of the variables. The analysis used the full dataset after checking for outliers based on potential departures of homoscedasticity and normally-distributed residuals.
The model of speciation best fitting the phylogeny and species geographic ranges was inferred using a process-based simulation model that considers 14 summary metrics related to the speciation modes under the assumption supported by simulations that phylogenetic and geographic patterns retain signals of the history of speciation49. The model allows distinguishing among five speciation modes, including allopatric or vicariant, sympatric, parapatric, by dispersal and founder events, and a mixed mode including the previous four. The analysis of speciation mode used the phylogenies for each genus separately, and species ranges estimated as above, and it was run with the set of functions and the DReaD simulation established by49. This analysis included inference of summary metrics relevant for discrimination of speciation modes, likelihood-free model selection based on machine-learning linear discriminant analysis (LDA) and cross-validation accuracy test for model classification, and tests were performed with the R packages klaR 0.6-15120 and caret 6.0121. Here, LDA model selection assigns posterior probabilities to each model class to rank their potential contribution to the observed pattern.
All the statistical analyses described were run in the R environment122 (or the Julia environment123, when indicated).
Data availability
All the sequences and distribution data used for this study are publicly available as data associated to articles Papadopoulou et al. (2013), Gómez-Zurita and Cardoso (2014), Platania et al. (2020), Gómez-Zurita et al. (2020) and Platania and Gómez-Zurita (2022), and are under sequence accession numbers of the European Nucleotide Archive (https://www.ebi.ac.uk/ena/browser/home) listed in Table S1 of the Supplementary Information. Scripts and data files used for the analyses are included as part of the Supplementary Information of this article.
References
Caesar, M., Grandcolas, P. & Pellens, R. Outstanding micro-endemism in New Caledonia: More than one out of ten animal species have a very restricted distribution range. PLoS ONE 12, e0181437 (2017).
Hadjou Belaid, A., Maurice, S., Freville, H., Carbonell, D. & Imbert, E. Predicting population viability of the narrow endemic Mediterranean plant Centaurea corymbosa under climate change. Biol. Cons. 223, 19–33 (2018).
Purvis, A., Gittleman, J. L., Cowlishaw, G. & Mace, G. M. Predicting extinction risk in declining species. Proc. R. Soc. Lond. B 267, 1947–1952 (2000).
Ding, C. Z., Jiang, X. M., Xie, Z. C. & Brosse, S. Seventy-five years of biodiversity decline of fish assemblages in Chinese isolated plateau lakes: Widespread introductions and extirpations of narrow endemics lead to regional loss of dissimilarity. Div. Distr. 23, 171–184 (2017).
Thomas, C. D. Translocation of species, climate change, and the end of trying to recreate past ecological communities. Trends Ecol. Evol. 26, 216–221 (2011).
Pintaud, J.-C. & Jaffré, T. Patterns of diversity and endemism in palms on ultramafic rocks in New Caledonia. S. Afr. J. Sci. 97, 548–550 (2001).
Wollenberg, K. C., Vieites, D. R., Glaw, F. & Vences, M. Speciation in little: The role of range and body size in the diversification of Malagasy mantellid frogs. BMC Evol. Biol. 11, 217 (2011).
Ochoa-Ochoa, L. M., Rodríguez, P., Mora, F., Flores-Villela, O. & Whittaker, R. J. Climate change and amphibian diversity patterns in Mexico. Biol. Cons. 150, 94–102 (2012).
Bryson, R. W. Jr., Savary, W. E., Zellmer, A. J., Bury, R. B. & McCormack, J. E. Genomic data reveal ancient microendemism in forest scorpions across the California Floristic Province. Mol. Ecol. 25, 3731–3751 (2016).
Skipwith, P. L., Bauer, A. M., Jackman, T. R. & Sadlier, R. A. Old but not ancient: Coalescent species tree of New Caledonian geckos reveals recent post-inundation diversification. J. Biogeogr. 43, 1266–1276 (2016).
Wilmé, L., Goodman, S. M. & Ganzhorn, J. U. Biogeographic evolution of Madagascar’s microendemic biota. Science 312, 1063–1065 (2006).
Echternacht, L., Sano, P. T., Trovó, M. & Dubuisson, J.-Y. Phylogenetic analysis of the Brazilian microendemic Paepalanthus subgenus Xeractis (Eriocaulaceae) inferred from morphology. Bot. J. Linn. Soc. 167, 137–152 (2011).
Bitencourt, C. & Rapini, A. Centres of endemism in the Espinhaço Range: Identifying cradles and museums of Asclepiadoideae (Apocynaceae). Syst. Biodiv. 11, 525–536 (2013).
Inglis, P. W. & Cavalcanti, T. B. A molecular phylogeny of the genus Diplusodon (Lythraceae), endemic to the campos rupestres and cerrados of South America. Taxon 67, 66–82 (2018).
Hamer, M. L. & Slotow, R. H. Conservation application of existing data for South African millipedes (Diplopoda). Afr. Entom. 10, 29–42 (2002).
Linder, H. P. et al. The partitioning of Africa: Statistically defined biogeographical regions in sub-Saharan Africa. J. Biogeogr. 39, 1189–1205 (2012).
Vences, M., Wollenberg, K. C., Vieites, D. R. & Lees, D. C. Madagascar as a model region of species diversification. Trends Ecol. Evol. 24, 456–465 (2009).
Vences, M. Origin of Madagascar’s extant fauna: A perspective from amphibians, reptiles and other non-flying vertebrates. Ital. J. Zool. 71, 217–228 (2004).
Grandcolas, P. et al. New Caledonia: A very old Darwinian island? Phil. Trans. R. Soc. 363, 3309–3317 (2008).
Miraldo, A., Wirta, H. & Hanski, I. Origin and diversification of dung beetles in Madagascar. Insects 2, 112–127 (2011).
Pillon, Y. Time and tempo of diversification in the flora of New Caledonia. Bot. J. Linn. Soc. 170, 288–298 (2012).
Papadopoulou, A., Cardoso, A. & Gómez-Zurita, J. Diversity and diversification of Eumolpinae (Coleoptera: Chrysomelidae) in New Caledonia. Zool. J. Linn. Soc. 168, 473–495 (2013).
Anso, J. et al. Old lineage on an old island: Pixibinthus, a new cricket genus endemic to New Caledonia shed light on gryllid diversification in a hotspot of biodiversity. PLoS ONE 11, e0150920 (2016).
Hackel, J. et al. Grass diversification in Madagascar: In situ radiation of two large C3 shade clades and support for a Miocene to Pliocene origin of C4 grassy biomes. J. Biogeogr. 45, 750–761 (2018).
Burbrink, F. T. et al. The origins and diversification of the exceptionally rich gemsnakes (Colubroidea: Lamprophiidae: Pseudoxyrhophiinae) in Madagascar. Syst. Biol. 68, 918–936 (2019).
García-Vázquez, D., Bilton, D. T., Foster, G. N. & Ribera, I. Pleistocene range shifts, refugia and the origin of widespread species in western Palaearctic water beetles. Mol. Phylogenet. Evol. 114, 122–136 (2017).
Vitales, D. et al. The explosive radiation of Cheirolophus (Asteraceae, Cardueae) in Macaronesia. BMC Evol. Biol. 14, 118 (2014).
Valero, K. C. W. Evidence for an intrinsic factor promoting landscape genetic divergence in Madagascan leaf-litter frogs. Fr. Genet. 6, 155 (2015).
Hollingsworth, P. R. Jr. & Near, T. J. Temporal patterns of diversification and microendemism in eastern highland endemic barcheek darters (Percidae: Etheostomatinae). Evolution 63, 228–243 (2009).
Harrison, S. & Noss, R. Endemism hotspots are linked to stable climatic refugia. Ann. Bot. 119, 207–214 (2017).
Nattier, R. et al. Climate and soil type together explain the distribution of microendemic species in a biodiversity hotspot. PLoS ONE 8, e80811 (2013).
Nattier, R. et al. Secondary sympatry caused by range expansion informs on the dynamics of microendemism in a biodiversity hotspot. PLoS ONE 7, e48047 (2012).
Lavergne, S., Thompson, J. D., Garnier, E. & Debussche, M. The biology and ecology of narrow endemic and widespread plants: A comparative study of trait variation in 20 congeneric pairs. Oikos 107, 505–518 (2004).
Rowley, J. J. L. et al. Undiagnosed cryptic diversity in small, microendemic frogs (Leptolalax) from the Central Highlands of Vietnam. PLoS ONE 10, e0128382 (2015).
Grismer, L. L. et al. Twelve new species of Cyrtodactylus Gray (Squamata: Gekkonidae) from isolated limestone habitats in east-central and southern Myanmar demonstrate high localized diversity and unprecedented microendemism. Zool. J. Linn. Soc. 182, 862–959 (2018).
Younger, J. L. et al. Hidden diversity of forest birds in Madagascar revealed using integrative taxonomy. Mol. Phylogenet. Evol. 124, 16–26 (2018).
Miraldo, A. & Hanski, I. A. Competitive release leads to range expansion and rampant speciation in Malagasy dung beetles. Syst. Biol. 63, 480–492 (2014).
Faille, A., Bourdeau, C., Bellés, X. & Fresneda, J. Allopatric speciation illustrated: The hypogean genus Geogrechus Jeannel, 1919 (Coleoptera: Carabidae: Trechini), with description of four new species from the eastern Pyrenees (Spain). Arthr. Syst. Phylog. 73, 439–455 (2015).
Morlon, H. Phylogenetic approaches for studying diversification. Ecol. Lett. 17, 508–525 (2014).
Lewitus, E. & Morlon, H. Detecting environment-dependent diversification from phylogenies: A simulation study and some empirical illustrations. Syst. Biol. 67, 576–593 (2018).
Pabijan, M., Wollenberg, K. C. & Vences, M. Small body size increases the regional differentiation of populations of tropical mantellid frogs (Anura: Mantellidae). J. Evol. Biol. 25, 2310–2324 (2012).
Andújar, C. et al. Speciation below ground: Tempo and mode of diversification in a radiation of endogean ground beetles. Mol. Ecol. 26, 6053–6070 (2017).
Gómez-Zurita, J. & Cardoso, A. Systematics of the New Caledonian endemic genus Taophila Heller (Coleoptera: Chrysomelidae, Eumolpinae) combining morphological, molecular and ecological data, with description of two new species. Syst. Ent. 39, 111–126 (2014).
Gómez-Zurita, J. Insights on the genus Acronymolpus Samuelson with new synonymies and exclusion of Stethotes Baly from the fauna of New Caledonia (Coleoptera, Chrysomelidae, Eumolpinae). ZooKeys 720, 65–75 (2017).
Gómez-Zurita, J. Description of Kumatoeides gen. nov. (Coleoptera: Chrysomelidae, Eumolpinae) from New Caledonia. Zootaxa 4521, 89–115 (2018).
Gómez-Zurita, J., Platania, L. & Cardoso, A. A new species of the genus Tricholapita nom. nov. and stat. nov. (Coleoptera: Chrysomelidae, Eumolpinae) from New Caledonia. Zootaxa 4857, 85–94 (2020).
Platania, L., Cardoso, A. & Gómez-Zurita, J. Diversity and evolution of New Caledonian endemic genus Taophila subgenus Lapita (Coleoptera: Chrysomelidae: Eumolpinae). Zool. J. Linn. Soc. 189, 1123–1154 (2020).
Platania, L. & Gómez-Zurita, J. Integrative taxonomic revision of the New Caledonian endemic genus Taophila Heller (Coleoptera: Chrysomelidae, Eumolpinae). Insect Syst. Evol. 53, 111–184 (2022).
Skeels, A. & Cardillo, M. Reconstructing the geography of speciation from contemporary biodiversity data. Am. Nat. 193, 240–255 (2019).
Mittermeier, R. A. et al. Hotspots Revisited (University of Chicago Press, 2005).
Mouillot, D. et al. Rare species support vulnerable functions in highdiversity ecosystems. PLoS Biol. 11, e1001569 (2013).
Quental, T. B. & Marshall, C. R. Diversity dynamics: Molecular phylogenies need the fossil record. Trends Ecol. Evol. 25, 434–441 (2010).
Louca, S. & Pennell, M. W. Extant timetrees are consistent with a myriad of diversification histories. Nature 580, 502–505 (2020).
Louca, S. & Pennell, M. W. Why extinction estimates from extant phylogenies are so often zero? Curr. Biol. 31, 3168–3173 (2021).
Chazot, N. et al. Conserved ancestral tropical niche but different continental histories explain the latitudinal diversity gradient in brush-footed butterflies. Nat. Comm. 12, 5717 (2021).
McPeek, M. A. The ecological dynamics of clade diversification and community assembly. Am. Nat. 172, E270–E284 (2008).
Phillimore, A. B. & Price, T. D. Density-dependent cladogenesis in birds. PLoS Biol. 6, e71 (2008).
Morlon, H., Potts, M. D. & Plotkin, J. B. Inferring the dynamics of diversification: A coalescent approach. PLoS Biol. 8, e1000493 (2010).
Moen, D. & Morlon, H. Why does diversification slow down? Trends Ecol. Evol. 29, 190–197 (2014).
Condamine, F. L., Rolland, J. & Morlon, H. Assessing the causes of diversification slowdowns: Temperature-dependent and diversity-dependent models receive equivalent support. Ecol. Lett. 22, 1900–1912 (2019).
Espeland, M. & Murienne, J. Diversity dynamics in New Caledonia: Towards the end of the museum model? BMC Evol. Biol. 11, 254 (2011).
Avise, J. C. & Walker, D. Pleistocene phylogeographic effects on avian populations and the speciation process. Proc. R. Soc. Lond. B 265, 457–463 (1998).
Etienne, R. S. & Rosindell, J. Prolonging the past counteracts the pull of the present: Protracted speciation can explain observed slowdowns in diversification. Syst. Biol. 61, 204–213 (2012).
Pannetier, T., Martinez, C., Bunnefeld, L. & Etienne, R. S. Branching patterns in phylogenies cannot distinguish diversity-dependent diversification from time-dependent diversification. Evolution 75, 25–38 (2021).
Herbert, T. D. et al. Late Miocene global cooling and the rise of modern ecosystems. Nat. Geosci. 9, 843–847 (2016).
Dynesius, M. & Jansson, R. Evolutionary consequences of changes in species’ geographical distributions driven by Milankovitch climate oscillations. Proc. Natl. Acad. Sci. USA 97, 9115–9120 (2000).
Boucher, F. C., Zimmermann, N. E. & Conti, E. Allopatric speciation with little niche divergence is common among alpine Primulaceae. J. Biogeogr. 43, 591–602 (2016).
Etienne, R. S., Pigot, A. L. & Phillimore, A. B. How reliably can we infer diversity-dependent diversification from phylogenies? Methods Ecol. Evol. 7, 1092–1099 (2016).
Jaffré, T., Morat, P., Veillon, J.-M. & MacKee, H. S. Changements dans la végétation de la Nouvelle-Calédonie au cours du Tertiaire: la végétation et la flore des roches ultrabasiques. Bull. Mus. Natl. Hist. Nat. 9, 365–391 (1987).
Jaffré, T. & Veillon, J.-M. Structural and floristic characteristics of a rain forest on schist in New Caledonia: A comparison with an ultramafic rain forest. Bull. Mus. Natl. Hist. Nat. 17, 201–226 (1995).
Lowry, P.P. Diversity, endemism, and extinction in the flora of New Caledonia: A review. In Peng, C.I., Lowry, P.P. (eds.), Proceedings of the International Symposium on Rare, Threatened, and Endangered Floras of Asia and the Pacific. Academica Sinica, Taipei, pp. 181–206 (1998).
Pintaud, J.-C., Jaffré, T. & Puig, H. Chorology of new caledonian palms and possible evidence of Pleistocene rain forest refugia. Comp. R. Acad. Sci. 324, 453–463 (2001).
Lillie, A. R. & Brothers, R. N. The geology of New Caledonia. N. Z. Jl. Geol. Geophys. 13, 145–183 (1970).
Chevillotte, V., Chardon, D., Beauvais, A., Maurizot, P. & Colin, F. Long-term tropical morphogenesis of New Caledonia (Southwest Pacific): Importance of positive epeirogeny and climate change. Geomorphology 81, 361–375 (2006).
Sevin, B. et al. Post-obduction evolution of New Caledonia. Geol. Soc. Lond. Mem. 51, 147–188 (2020).
Glor, R. E. Phylogenetic insights on adaptive radiation. Annu. Rev. Ecol. Evol. Syst. 41, 251–270 (2010).
Rovito, S. M., Vásquez-Almazán, C. R., Papenfuss, T. J., Parra-Olea, G. & Wake, D. B. Biogeography and evolution of Central American cloud forest salamanders (Caudata: Plethodontidae: Cryptotriton), with the description of a new species. Zool. J. Linn. Soc. 175, 150–166 (2015).
Pillon, Y., Munzinger, J., Amir, H. & Lebrun, M. Ultramafic soils and species sorting in the flora of New Caledonia. J. Ecol. 98, 1108–1116 (2010).
Pigot, A. L., Phillimore, A. B., Owens, I. P. F. & Orme, C. D. L. The shape and temporal dynamics of phylogenetic trees arising from geographic speciation. Syst. Biol. 59, 660–673 (2010).
Coyne, J. A. & Orr, H. A. Speciation (Sinauer Associates, 2004).
Fitzpatrick, B. M. & Turelli, M. The geography of mammalian speciation: Mixed signals from phylogenies and range maps. Evolution 60, 601–615 (2006).
Losos, J. B. & Glor, R. E. Phylogenetic comparative methods and the geography of speciation. Trends Ecol. Evol. 18, 220–227 (2003).
Cardillo, M. & Warren, D. L. Analysing patterns of spatial and niche overlap among species at multiple resolutions. Glob. Ecol. Biogeogr. 25, 951–963 (2016).
Barraclough, T. G. & Vogler, A. P. Detecting the geographic pattern of speciation from species level phylogenies. Am. Nat. 155, 419–434 (2000).
Phillimore, A. B. et al. Sympatric speciation in birds is rare: Insights from range data and simulations. Am. Nat. 171, 646–657 (2008).
Anacker, B. L. & Strauss, S. Y. The geography and ecology of plant speciation: Range overlap and niche divergence in sister species. Proc. R. Soc. Lond. B 281, 20132980 (2014).
Ribera, I. et al. The geography of speciation in narrow-range endemics of the ‘Haenydra’ lineage (Coleoptera, Hydraenidae, Hydraena). J. Biogeogr. 38, 502–516 (2011).
Kunprom, C., Plant, A. R. & Pramual, P. Population genetic structure and microendemism in aquatic Empididae (Diptera) in transient tufa biotopes on tropical karst. Entom. Sci. 24, 127–136 (2021).
Platnick, N. I. & Nelson, G. A method of analysis of historical biogeography. Syst. Zool. 27, 1–16 (1978).
Pennington, R. T. et al. Historical climate change and speciation: Neotropical seasonally dry forest plants show patterns of both Tertiary and Quaternary diversification. Phil. Trans. R. Soc. Lond. B 359, 515–538 (2004).
Guillaumet, A., Crochet, P. A. & Pons, J. M. Climate-driven diversification in two widespread Galerida larks. BMC Evol. Biol. 8, 1–22 (2008).
Lanfear, R., Kokko, H. & Eyre-Walker, A. Population size and the rate of evolution. Trends Ecol. Evol. 29, 33–41 (2014).
Waters, J. M. Competitive exclusion: Phylogeography’s ‘elephant in the room’? Mol. Ecol. 20, 4388–4394 (2011).
Waters, J. M., Fraser, C. I. & Hewitt, G. M. Founder takes all: Density-dependent processes structure biodiversity. Trends Ecol. Evol. 28, 78–85 (2013).
Gröning, J. & Hochkirch, A. Reproductive interference between animal species. Q. Rev. Biol. 83, 257–282 (2008).
Wesener, T. & Conrad, C. Local hotspots of endemism or artifacts of incorrect taxonomy? The status of micoendemic pill millipede species of the genus Glomeris in northern Italy (Diplopoda, Glomerida). PLoS ONE 11, e0162284 (2016).
Ahrends, A. et al. Funding begets biodiversity. Divers. Distrib. 17, 191–200 (2011).
Ward, D. F. More than just records: Analysing natural history collections for biodiversity planning. PLoS ONE 7, e50346 (2012).
Taylor-Smith, B., Morgan-Richards, M. & Trewick, S. A. Patterns of regional endemism among New Zealand invertebrates. N. Z. J. Zool. 47, 1–19 (2019).
Wulff, A. S. et al. Conservation priorities in a Biodiversity Hotspot: Analysis of narrow endemic plant species in New Caledonia. PLoS ONE 8, e73371 (2013).
Katoh, K. & Standley, D. M. MAFFT Multiple Sequence Alignment software Version 7: Improvements in performance and usability. Mol. Biol. Evol. 30, 772–780 (2013).
Drummond, A. J., Suchard, M. A., Xie, D. & Rambaut, A. Bayesian phylogenetics with BEAUti and the BEAST 1.7. Mol. Biol. Evol. 29, 1969–1973 (2012).
Tamura, K. & Nei, M. Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol. Biol. Evol. 10, 512–526 (1993).
Rabosky, D. L. Automatic detection of key innovations, rate shifts, and diversity-dependence on phylogenetic trees. PLoS ONE 9, e89543 (2014).
Rabosky, D. L. et al. BAMMtools: An R package for the analysis of evolutionary dynamics on phylogenetic trees. Methods Ecol. Evol. 5, 701–707 (2014).
Cusimano, N. & Renner, S. S. Slowdowns in diversification rates from real phylogenies may not be real. Syst. Biol. 59, 458–464 (2010).
Maliet, O., Hartig, F. & Morlon, H. A model with many small shifts for estimating species-specific diversification rates. Nat. Ecol. Evol. 3, 1086–1092 (2019).
Morlon, H. et al. RPANDA: An R package for macroevolutionary analyses on phylogenetic trees. Methods Ecol. Evol. 7, 589–597 (2016).
Maliet, O. & Morlon, H. Fast and accurate estimation of species-specific diversification rates using data augmentation. Syst. Biol. 71, 353–366 (2022).
Epstein, S., Buchsbaum, R., Lowenstam, H. A. & Urey, H. C. Revised carbonate-water isotopic temperature scale. Geol. Soc. Am. Bull. 64, 1315–1326 (1953).
Etienne, R. S. et al. Diversity-dependence brings molecular phylogenies closer to agreement with the fossil record. Proc. R. Soc. Lond. B 279, 1300–1309 (2012).
Hua, X., Herdha, T. & Burden, C. J. Protracted speciation under the state-dependent speciation and extinction approach. Syst. Biol. 71, 1362–1377 (2022).
FitzJohn, R. G. Diversitree: Comparative phylogenetic analyses of diversification in R. Meth. Ecol. Evol. 3, 1084–1092 (2012).
Schwery, O., O'Meara, B.C. 2020. Preprint: BoskR: Testing adequacy of diversification models using tree shape. bioRxiv, 22/12/2020, https://doi.org/10.1101/2020.12.21.423829
Pennell, M. W., FitzJohn, R. G., Cornwell, W. K. & Harmon, L. J. Model adequacy and the macroevolution of angiosperm functional traits. Am. Nat. 186, E33–E50 (2015).
Lewitus, E. & Morlon, H. Natural constraints to species diversification. PLoS Biol. 14, e1002532 (2016).
Warren, D., Dinnage, R. ENMTools: Analysis of niche evolution using niche and distribution models. R package version 1.0.1. (2020) https://CRAN.R-project.org/package=ENMTools.
Pinheiro, J., Bates, D., DebRoy, S., Sarkar, D., R Core Team. nlme: Linear and Nonlinear Mixed Effects Models. R package version 3.1-155, (2022) https://CRAN.R-project.org/package=nlme.
Pagel, M. Inferring the historical patterns of biological evolution. Nature 401, 877–884 (1999).
Weihs, C., Ligges, U., Luebke, K. & Raabe, N. klaR, analyzing german business cycles. In Data Analysis and Decision Support (eds Baier, D. et al.) 335–343 (Springer, 2005).
Kuhn, M. Caret: Classification and Regression Training. R package version 6.0-71 (2016). https://CRAN.R-project.org/package=caret.
R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. Vienna, Austria. (2021) https://www.R-project.org.
Bezanson, J., Edelman, A., Karpinski, S. & Shah, V. B. Julia: A fresh approach to numerical computing. SIAM Rev. 59, 65–98 (2017).
Acknowledgements
We are indebted to Alexander Skeels (Swiss Federal Research Institute for Forest, Birmensdorf) for sharing unpublished scripts to run their speciation model simulations and tests, and to Orlando Schwery (University of Idaho, Moscow, USA) for assistance and sharing unpublished BoskR scripts to study model adequacy as well as discussion about the interpretation of results, and to Xia Hua (Australian National University, Canberra) for assistance with her recently published ProSSE functions.
Funding
Work funded by the Spanish “Agencia Estatal de Investigación” and the European Regional Development Fund of the European Commission (Grants CGL2017-83324-P and PID2021-123668NB-I00) and associated predoctoral scholarship PRE2018-083172 to LP.
Author information
Authors and Affiliations
Contributions
L.P. and J.G.Z. were responsible for every stage of the production of this manuscript. The figures included in the article were originally drawn by the authors in all of their parts, unless stated in the foot caption, where formal permission was requested and attached to the submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Platania, L., Gómez-Zurita, J. Analysis of intrinsic evolutionary factors leading to microendemic distributions in New Caledonian leaf beetles. Sci Rep 13, 6909 (2023). https://doi.org/10.1038/s41598-023-34104-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-34104-z
- Springer Nature Limited