Abstract
Transcranial random noise stimulation (tRNS) of the primary sensory or motor cortex can improve sensorimotor functions by enhancing circuit excitability and processing fidelity. However, tRNS is reported to have little effect on higher brain functions, such as response inhibition when applied to associated supramodal regions. These discrepancies suggest differential effects of tRNS on the excitability of the primary and supramodal cortex, although this has not been directly demonstrated. This study examined the effects of tRNS on supramodal brain regions on somatosensory and auditory Go/Nogo task performance, a measure of inhibitory executive function, while simultaneously recording event-related potentials (ERPs). Sixteen participants received sham or tRNS stimulation of the dorsolateral prefrontal cortex in a single-blind crossover design study. Neither sham nor tRNS altered somatosensory and auditory Nogo N2 amplitudes, Go/Nogo reaction times, or commission error rates. The results suggest that current tRNS protocols are less effective at modulating neural activity in higher-order cortical regions than in the primary sensory and motor cortex. Further studies are required to identify tRNS protocols that effectively modulate the supramodal cortex for cognitive enhancement.
Similar content being viewed by others
Introduction
Transcranial random noise stimulation (tRNS) can modulate cortical activity and enhance motor and sensory functions1. Furthermore, these effects may persist following stimulation, suggesting therapeutic value for neurological and neuropsychiatric diseases. For instance, Terney et al.2 reported that brief tRNS of the primary motor cortex (M1) increased the local motor evoked potential amplitude, a manifestation of enhanced excitability, for at least 60 min post-stimulation. Subsequent studies have also reported that tRNS is more effective than transcranial direct current stimulation (tDCS) for increasing MI excitability3,4. In addition, tRNS applied to a specific primary sensory cortex can modulate local neural activity and improve the perception of the modality subserved by that region, including the perception of somatosensory5, auditory6,7, and visual8,9,10 inputs, both during and after stimulation. Collectively, these and related findings suggest that tRNS is a more effective method for cortical modulation than other forms of transcranial electrical stimulation (tES).
In contrast to sensorimotor function, these tRNS protocols appear to have little to no effect on higher-order cognitive functions such as two-digit addition11, working memory12, and response inhibition13,14. However, such studies have focused only on behavioral responses rather than neurophysiological measures such as event-related potentials (ERPs), so the mechanisms for these differential effects remain unclear. Therefore, the present study investigated the effect of tRNS on Go/Nogo response time (RT) and error rate as behavioral measures of response inhibition and on the associated ERPs as a measure of neural excitability in the stimulated brain region.
Inhibitory control, the ability to suppress a thought or motor response, is a crucial component of executive function in daily life15,16. The inferior frontal gyrus (IFG) and dorsolateral prefrontal cortex (DLPFC) are the significant contributors to inhibitory control of responses to somatosensory17, auditory, and visual stimuli18 in functional magnetic resonance imaging (fMRI) studies. However, Brevet-Aeby19 reported that tRNS applied to the DLPFC did not improve inhibitory response accuracy despite improved Go/Nogo RT, while Brauer et al.14 reported that neither 6-Hz tACS nor tRNS of the IFG influenced Go/Nogo performance (error rate and RT) during stimulation (online) and following stimulation (offline). These discrepancies in behavioral responses to transcranial stimulation of primary and supramodal cortices may be explained by differential effects on neural excitability; however, no study has investigated the effect of tRNS on the excitability of supramodal inhibitory brain region using ERPs. In our previous study, we measured the subtracted Nogo N2 ERP (mean Nogo trial waveform minus mean Go trial waveform) in the both somatosensory and auditory cortex20 and found a positive correlation between the Nogo N2 amplitude and Go/Nogo RT for the somatosensory modality21, suggesting that a tRNS-induced increase in supramodal Nogo N2 amplitude could improve both somatosensory and auditory Go/Nogo RTs. Therefore, we evaluated the effects of tRNS on both behavioral and neurophysiological function by recording ERPs during somatosensory and auditory Go/Nogo paradigms.
Methods
Participants
Sixteen healthy undergraduate university male students (20.6 ± 0.9 years, height 173.5 ± 3.9 cm) participated in this study. This sample size was larger than the minimum of 14 needed for 80% power and a significance level of 0.05 based on an effect size of 0.30. Written informed consent was obtained from each participant after a full explanation of the study objectives and methods. The study was conducted in accordance with the Declaration of Helsinki and approved by the ethics committee of Niigata University of Health and Welfare, Niigata, Japan (approval number: 18090).
Random noise stimulation
High-frequency oscillatory current tRNS (100–640 Hz, 1.5 mA, 10-s rise and fall periods) was applied for 15 min using sponge-covered 5 × 5 cm rubber electrodes connected to a neuroConn DC-Stimulator (neuroConn GmbH, Ilmenau, Germany). We set the stimulation mode to “noise HF”, which is a random level of current generated for every sample (sampling rate 1280 samples/s). The random numbers are normally distributed, and the probability density function follows a bell-shaped curve2,3. Stimulating electrodes were placed bilaterally at F3 and F4 according to the international 10–20 EEG standard to stimulate the DLPFC19.
Somatosensory and auditory stimulation
Somatosensory ERPs were elicited by constant current square-wave pulses (duration 0.2 ms) delivered to the second and fifth digits of the dominant hand by ring electrodes. At each digit, the anode was placed at the distal interphalangeal joint and the cathode at the proximal interphalangeal joint. The fifth digit was stimulated in the Go condition, and the second digit in the Nogo condition. Stimulus intensity at the fifth digit was fixed at three times the participant’s sensory threshold, and that at the second digit was adjusted so that the participant reported the same sensation intensity as at the fifth digit. These stimuli elicited no pain or other unpleasant sensations. Auditory ERPs were elicited by a pure tone delivered binaurally through headphones (60 dB sound pressure level, 50 ms duration, 10 ms rise time, and 10 ms fall time). A 1000 Hz pure tone was delivered for the Go condition and a 1500 Hz pure tone for the Nogo condition. These same stimulation conditions were used in our recent study20.
Experimental paradigms
One week before electroencephalographic (EEG) measurements of ERPs, participants performed five practice sessions of both somatosensory and auditory Go/Nogo tasks, with each session consisting of 40 trials. A 1-min break was inserted between sessions to exclude the effects of short-term training. Figure 1 illustrates the experimental paradigm for EEG sessions. The actual study consisted of two separate sham and real tRNS sessions separated by at least one week to eliminate carry-over effects of stimulation, as reported in a previous study13. Sham and tRNS sessions order were randomized across participants. Participants performed separate somatosensory and then auditory Go/Nogo tasks on the same day, each consisting of 50 Go trials and 50 Nogo trials (i.e., equal 50% probabilities) presented in random order across sessions as described in our previous study20.
Individual somatosensory stimuli were presented at 2-s inter-stimulus intervals (ISIs) and auditory stimuli at 1-s ISIs to match the level of difficulty. In both somatosensory and auditory Go trials, participants were instructed to press a button as fast as possible using the dominant second digit when they perceived the Go stimulus (current or sound).
EEG recording and analysis
ERPs were recorded using a SynAmps amplifier system (Neuroscan, El Paso, TX, USA) connected to five scalp electrodes (impedance < 5 kΩ) placed at Fz, Cz, Pz, F3, and F4 according to the 10–20 system and a reference placed on the left earlobe. The sampling rate was 1000 Hz, and EEG signals were recorded with a notch filter (50 Hz). According to our previous studies, trials with responses exceeding ± 100 μV were excluded from signal averaging. Signals were then band-pass filtered offline at 0.5–60 Hz. In both the somatosensory and auditory Go/Nogo paradigms, 50 artifact-free Go and 50 artifact-free Nogo trial waveforms were averaged pre- and post-stimulation (sham or tRNS) for each participant. Responses were analyzed from 100 ms before (baseline) to 500 ms after stimulus onset using Neuroscan 4.3 software. To extract Nogo potentials, we subtracted the averaged waveform of Go trials from Nogo trials as described in previous studies22,23,24,25. The subtracted waveform, termed the somatosensory or auditory Nogo N2 component, exhibited a negative peak relative to the prestimulus baseline at ~ 200 ms after the onset of the somatosensory or auditory stimulus. The peak latencies and amplitudes of Nogo N2 waveforms were measured between 120 and 250 ms after stimulus onset at 10–20 electrode positions Fz, Cz, Pz, F3, and F4 because this waveform is shown to reach a maximum around the frontal region for both somatosensory21 and auditory20 modalities.
Data and statistical analysis
Behavioral data were obtained for Go/Nogo RT and commission error for the pre- and post-conditions in both the modalities. Parametric data (distribution confirmed using the Shapiro–Wilk test) were compared using paired-sample t-tests in both modalities. Nonparametric data were tested using Wilcoxon’s signed-rank test.
The neurophysiological data, Nogo N2 amplitudes for pre- and post-conditions in both modalities, were obtained. Parametric data were compared by two-way ANOVA with stimulation (pre vs post) and five electrode positions (Fz, Cz, Pz, F3, F4) as a within-participant factor. Nonparametric data were analyzed using Wilcoxon’s signed-rank test to compare the stimulation effects (pre vs post) by analyzing the pooled N2 amplitude of the five electrode positions.
For the two-way ANOVA analyses, we performed Mauchly’s sphericity assumption test; if it was violated, the Greenhouse–Geisser epsilon was used to correct the degrees of freedom. Statistical significance was set at p < 0.05 for all tests.
Results
DLPFC-targeted tRNS did not alter Go/Nogo RTs and commission error rates
All Go/NogoRT data were parametric while all commission error data were nonparametric. Therefore, paired-sample t-test and Wilcoxon’s signed-rank test were used to analyze Go/Nogo RT data and commission error rate data, respectively. The tRNS of the DLPFC did not significantly alter somatosensory Go/Nogo RT (235 ± 28 ms at baseline vs. 232.0 ± 30 ms post-stimulus, t (15) = 1.574, p = 0.136) or auditory Go/Nogo RT (254 ± 24 ms vs. 252 ± 27 ms, t (15) = 0.345, p = 0.734). Similarly, sham stimulation did not alter somatosensory Go/Nogo RT (232 ± 21 ms vs. 227 ± 21 ms, t (15) = 1.309, p = 0.210) or auditory Go/Nogo RT (252 ± 28 ms vs. 245 ± 21 ms, t (15) = 1.688, p = 0.112). Moreover, tRNS did not improve the somatosensory commission error rate (1.07% ± 1.60% at baseline vs. 1.03% ± 1.43% post-stimulation, p = 0.945) or auditory commission error rate (1.68% ± 1.71% vs. 0.92% ± 1.59%, p = 0.102). Sham stimulation also did not improve the somatosensory commission error rate (0.753% ± 1.04% vs. 0.459% ± 0.83%, p = 0.367) or auditory commission error rate (1.66% ± 1.75% vs. 0.98% ± 1.53%, p = 0.236).
DLPFC-targeted tRNS did not alter somatosensory Nogo N2 amplitude
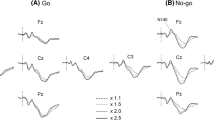
The peak amplitudes of somatosensory and auditory Nogo N2 waveforms at the five frontal and midline electrode sites Fz, Cz, Pz, F3, and F4 are summarized in Table 1, while Fig. 2A shows the grand-averaged waveforms of somatosensory Nogo N2 at three frontal electrode positions for the sham and tRNS sessions, and Fig. 2B summarizes somatosensory behaviors pre- and post-stimulation for sham and tRNS conditions. Somatosensory Nogo N2 amplitude was parametric data in both conditions. In the sham session, two-way ANOVA revealed no significant main effect of stimulation (F(1, 15) = 2.065, p = 0.171), electrode (F(2.093, 31.401) = 1.430, p = 0.255 ε = 0.523) and the interaction of stimulation × electrode (F(2.545, 38.173) = 0.358, p = 0.751 ε = 0.636) on somatosensory Nogo N2 amplitude. In the tRNS session, two-way ANOVA also revealed no significant main effect of tRNS stimulation (pre- vs. post, F(1, 15) = 0.122, p = 0.732 ε = 0.590) or electrode position (F(2.358, 35.371) = 2.796, p = 0.066 ε = 0.590), and no significant real stimulation × electrode position interaction effect (F(2.293, 34.401) = 0.328, p = 0.751 ε = 0.573) on somatosensory Nogo N2 amplitude.
Grand-averaged somatosensory Nogo N2 waveforms and task performance. (A) Grand-averaged somatosensory Nogo N2 waveforms at electrode positions F3, Fz, and F4 before sham or tRNS (pre, black lines) and after sham or tRNS (post, red lines). (B) Within-subject and mean Go/Nogo reaction times (RTs, left panels) and commission error rates (right panels) before (pre) and after (post) sham and tRNS stimulation.
DLPFC-targeted tRNS did not alter auditory Nogo N2 amplitude
Figure 3A shows the grand-averaged waveforms of auditory Nogo N2 waveforms at three frontal electrode positions for sham and tRNS sessions, and Fig. 3B summarizes auditory behaviors pre- and post-stimulation for both sham and tRNS conditions. Auditory Nogo N2 amplitude was nonparametric data in both the conditions. Wilcoxon’s signed-rank test revealed no significant main effect of sham stimulation on auditory Nogo N2 amplitude (− 4.8 μV vs − 4.0 μV, p = 0.095). Similarly, Wilcoxon’s signed-rank test revealed no significant main effect of tRNS on auditory Nogo N2 amplitude (− 2.65 μV vs − 2.90 μV, p = 0.502).
Grand-averaged auditory Nogo N2 waveforms and task performance. (A) Grand-averaged auditory Nogo N2 waveforms at electrode positions F3, Fz, and F4 before sham or tRNS (pre, black lines) and after sham or tRNS (post, red lines). (B) Within-subject and mean Go/Nogo reaction times (RTs, left panels) and commission error rates (right panels) before (pre) and after (post) sham and tRNS.
Discussion
The results of psychophysical stochastic resonance experiment revealed that noise stimulation for sensory area improved sensory detection performance in vision, audition, and touch26. Longtin et al.27 reported the role of noise in neural encoding and the phase-locked response of sensory neurons to weak periodic stimulation. However, the effects of noise stimulation on the activity of higher-order cortical regions and behaviors are debated. Taking these reports into consideration, we investigated the effect of tRNS applied to the DLPFC on somatosensory and auditory Nogo N2 amplitude and Go/Nogo task performance but found no significant changes compared to sham stimulation. Therefore, we suggest that tRNS does not influence response inhibition because it does not appropriately modulate the DLPFC circuits contributing to this function.
tRNS is a relatively new brain modulation technique, and the underlying mechanisms are still relatively unexplored1,28,29. Transcranial stimulation is believed to modulate neural activity and behavior directly by facilitating or inhibiting neuronal firing, referred to as an online effect, and by inducing lasting changes in synaptic and circuit function (neuroplasticity) as an offline effect29. One potential online effect of tRNS is the repetitive opening of voltage-gated sodium channels and ensuing neuronal excitation2,30. Random noise may serve to boost weak signals, thereby enhancing processing fidelity, a phenomenon referred to as stochastic resonance26,31. We initially speculated that the offline effect of tRNS could modulate somatosensory and auditory Nogo N2 amplitudes and Go/Nogo RTs through such neuroplastic effects on DLPFC neurons but observed no such changes.
We propose several potential explanations for this lack of effect. The most parsimonious explanation is that tRNS does not alter the neuronal or circuit functions of supramodal cortices as effectively as it modulates sensory and motor cortex activities. Indeed, tRNS over the SI increased both S1 activity and gradient orientation discrimination task performance5. Similarly, tRNS over the auditory cortex increased the near-threshold stimulus detection rate in the temporal domain and reduced the peak latencies of P50 and N1 components, indicating accelerated auditory sensory processing6. Also, tRNS targeted at lateral occipitotemporal cortices enhanced face perception compared to motor cortex stimulation9.
Further, several studies have reported that tRNS effectively increase corticospinal tract excitability compared to tDCS3,4. Thus, tRNS may enhance excitability and oscillations more reliably when directed at sensory and motor regions through the induction of stochastic resonance26. Several recent studies have suggested that tRNS can modulate oscillations in the sensory cortex6,7,32, although not visual evoked potentials (VEPs) N1, N2, and P2 in the visual cortex32. Thus, tRNS to the supramodal cortex may have no electrophysiologically or behaviorally detectable effects.
Previous studies have suggested modest or no effects of tRNS targeted at the frontal cortex. For example, Bieck et al.11 found little to no effect of tRNS targeted at parietal and frontal cortices on two-digit addition, while Mulquiney et al.12 reported that tRNS of the left DLPFC did not improve working memory, although tDCS could. To our knowledge, only two behavioral studies have investigated the effect of tRNS on inhibitory control using the Go/Nogo paradigm, and the results were contradictory. Brauer et al.14 found that tRNS at 1 mA for 20 min over the unilateral IFG had no effect on Go/Nogo RT or commission errors, while Brevet-Aeby et al.19 found that tRNS applied to the bilateral DLPFC at 2 mA improved Go/Nogo RT but not commission error rate. However, neither study examined changes in ERPs or other neurophysiological indices of neural activity.
In contrast, Sallard et al.13 reported that tRNS applied over the bilateral IFG did not impact Go/Nogo performance, while concomitant MEG analysis revealed a significant increase in the beta band (20 Hz) spectral power following real tRNS. This finding suggests that tRNS may modulate cortical oscillations but not ERPs or VEPs, which reflect the summation of excitatory postsynaptic potentials and inhibitory postsynaptic potentials33. Therefore, tRNS may not affect synaptic activity in higher brain regions such as the DLPFC. However, future studies are required to assess if tRNS intensity, duration, timing, and (or) set number can modulate the activity of supramodal cortical regions in ways that improve higher brain functions.
In conclusion, tRNS appears to enhance oscillatory activity online in the primary sensory and motor cortex, thereby improving perception and motor task performance. However, the current tRNS protocol does not modulate synaptic activity offline in supramodal regions and thus fails to enhance higher brain functions.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Potok, W., van der Groen, O., Bächinger, M., Edwards, D. & Wenderoth, N. Transcranial random noise stimulation modulates neural processing of sensory and motor circuits, from potential cellular mechanisms to behavior: A scoping review. eNeuro 9. https://doi.org/10.1523/eneuro.0248-21.2021 (2022).
Terney, D., Chaieb, L., Moliadze, V., Antal, A. & Paulus, W. Increasing human brain excitability by transcranial high-frequency random noise stimulation. J. Neurosci. 28, 14147–14155. https://doi.org/10.1523/jneurosci.4248-08.2008 (2008).
Inukai, Y. et al. Comparison of three non-invasive transcranial electrical stimulation methods for increasing cortical excitability. Front. Hum. Neurosci. 10. https://doi.org/10.3389/fnhum.2016.00668 (2016).
Moliadze, V., Fritzsche, G. & Antal, A. Comparing the efficacy of excitatory transcranial stimulation methods measuring motor evoked potentials. Neural Plast. 2014, 837141. https://doi.org/10.1155/2014/837141 (2014).
Saito, K. et al. Comparison of transcranial electrical stimulation regimens for effects on inhibitory circuit activity in primary somatosensory cortex and tactile spatial discrimination performance. Behav. Brain Res. 375, 112168. https://doi.org/10.1016/j.bbr.2019.112168 (2019).
Rufener, K. S., Ruhnau, P., Heinze, H. J. & Zaehle, T. Transcranial random noise stimulation (tRNS) shapes the processing of rapidly changing auditory information. Front. Cell. Neurosci. 11, 162. https://doi.org/10.3389/fncel.2017.00162 (2017).
Van Doren, J., Langguth, B. & Schecklmann, M. Electroencephalographic effects of transcranial random noise stimulation in the auditory cortex. Brain Stimul. 7, 807–812. https://doi.org/10.1016/j.brs.2014.08.007 (2014).
Herpich, F., Contò, F., van Koningsbruggen, M. & Battelli, L. Modulating the excitability of the visual cortex using a stimulation priming paradigm. Neuropsychologia 119, 165–171. https://doi.org/10.1016/j.neuropsychologia.2018.08.009 (2018).
Romanska, A., Rezlescu, C., Susilo, T., Duchaine, B. & Banissy, M. J. High-frequency transcranial random noise stimulation enhances perception of facial identity. Cerebral Cortex (New York, N.Y. : 1991) 25, 4334–4340. https://doi.org/10.1093/cercor/bhv016 (2015).
Fertonani, A., Pirulli, C. & Miniussi, C. Random noise stimulation improves neuroplasticity in perceptual learning. J. Neurosci. 31, 15416–15423. https://doi.org/10.1523/jneurosci.2002-11.2011 (2011).
Bieck, S. M., Artemenko, C., Moeller, K. & Klein, E. Low to no effect: Application of tRNS during two-digit addition. Front. Neurosci. 12, 176. https://doi.org/10.3389/fnins.2018.00176 (2018).
Mulquiney, P. G., Hoy, K. E., Daskalakis, Z. J. & Fitzgerald, P. B. Improving working memory: Exploring the effect of transcranial random noise stimulation and transcranial direct current stimulation on the dorsolateral prefrontal cortex. Clin. Neurophysiol. 122, 2384–2389. https://doi.org/10.1016/j.clinph.2011.05.009 (2011).
Sallard, E., Buch, E. R., Cohen, L. G. & Quentin, R. No evidence of improvements in inhibitory control with tRNS. Neuroimage: Rep. 1, 100056. https://doi.org/10.1016/j.ynirp.2021.100056 (2021).
Brauer, H., Kadish, N. E., Pedersen, A., Siniatchkin, M. & Moliadze, V. No modulatory effects when stimulating the right inferior frontal gyrus with continuous 6 Hz tACS and tRNS on response inhibition: A behavioral study. Neural Plast. 2018, 3156796. https://doi.org/10.1155/2018/3156796 (2018).
Zheng, D., Oka, T., Bokura, H. & Yamaguchi, S. The key locus of common response inhibition network for no-go and stop signals. J. Cogn. Neurosci. 20, 1434–1442. https://doi.org/10.1162/jocn.2008.20100 (2008).
Aron, A. R. et al. Converging evidence for a fronto-basal-ganglia network for inhibitory control of action and cognition. J. Neurosci. 27, 11860–11864. https://doi.org/10.1523/jneurosci.3644-07.2007 (2007).
Nakata, H. et al. Somato-motor inhibitory processing in humans: an event-related functional MRI study. Neuroimage 39, 1858–1866. https://doi.org/10.1016/j.neuroimage.2007.10.041 (2008).
Walther, S., Goya-Maldonado, R., Stippich, C., Weisbrod, M. & Kaiser, S. A supramodal network for response inhibition. NeuroReport 21, 191–195. https://doi.org/10.1097/WNR.0b013e328335640f (2010).
Brevet-Aeby, C., Mondino, M., Poulet, E. & Brunelin, J. Three repeated sessions of transcranial random noise stimulation (tRNS) leads to long-term effects on reaction time in the Go/No Go task. Clin. Neurophysiol. 49, 27–32. https://doi.org/10.1016/j.neucli.2018.10.066 (2019).
Yamashiro, K. et al. Modality-specific improvements in sensory processing among baseball players. Sci. Rep. 11, 2248. https://doi.org/10.1038/s41598-021-81852-x (2021).
Yamashiro, K. et al. Skill-specific changes in somatosensory nogo potentials in baseball players. PLoS One 10, e0142581. https://doi.org/10.1371/journal.pone.0142581 (2015).
Smith, J. L. & Douglas, K. M. On the use of event-related potentials to auditory stimuli in the Go/NoGo task. Psychiatry Res. 193, 177–181. https://doi.org/10.1016/j.pscychresns.2011.03.002 (2011).
Nakata, H. et al. Effects of ISI and stimulus probability on event-related go/nogo potentials after somatosensory stimulation. Exp. Brain Res. 162, 293–299. https://doi.org/10.1007/s00221-004-2195-4 (2005).
Falkenstein, M., Hoormann, J. & Hohnsbein, J. ERP components in Go/Nogo tasks and their relation to inhibition. Acta Physiol. (Oxf) 101, 267–291. https://doi.org/10.1016/s0001-6918(99)00008-6 (1999).
Thorpe, S., Fize, D. & Marlot, C. Speed of processing in the human visual system. Nature 381, 520–522. https://doi.org/10.1038/381520a0 (1996).
Moss, F., Ward, L. M. & Sannita, W. G. Stochastic resonance and sensory information processing: A tutorial and review of application. Clin. Neurophysiol. 115, 267–281. https://doi.org/10.1016/j.clinph.2003.09.014 (2004).
Longtin, A., Bulsara, A. & Moss, F. Time-interval sequences in bistable systems and the noise-induced transmission of information by sensory neurons. Phys. Rev. Lett. 67, 656–659. https://doi.org/10.1103/PhysRevLett.67.656 (1991).
Reed, T. & Cohen Kadosh, R. Transcranial electrical stimulation (tES) mechanisms and its effects on cortical excitability and connectivity. J. Inherit. Metab. Dis. 41, 1123–1130. https://doi.org/10.1007/s10545-018-0181-4 (2018).
Antal, A. & Herrmann, C. S. Transcranial alternating current and random noise stimulation: Possible mechanisms. Neural. Plast. 2016, 3616807. https://doi.org/10.1155/2016/3616807 (2016).
Schoen, I. & Fromherz, P. Extracellular stimulation of mammalian neurons through repetitive activation of Na+ channels by weak capacitive currents on a silicon chip. J. Neurophysiol. 100, 346–357. https://doi.org/10.1152/jn.90287.2008 (2008).
Stacey, W. C. & Durand, D. M. Stochastic resonance improves signal detection in hippocampal CA1 neurons. J. Neurophysiol. 83, 1394–1402. https://doi.org/10.1152/jn.2000.83.3.1394 (2000).
Ghin, F., O’Hare, L. & Pavan, A. Electrophysiological aftereffects of high-frequency transcranial random noise stimulation (hf-tRNS): An EEG investigation. Exp. Brain Res. 239, 2399–2418. https://doi.org/10.1007/s00221-021-06142-4 (2021).
Ibáñez, A. et al. Gesture influences the processing of figurative language in non-native speakers: ERP evidence. Neurosci. Lett. 471, 48–52. https://doi.org/10.1016/j.neulet.2010.01.009 (2010).
Funding
This study was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Number 22H03473 and a Grant-in-aid for Advanced Research from Niigata University of Health and Welfare.
Author information
Authors and Affiliations
Contributions
Edited the manuscript: K.Y., B.Y., T.F., G.O., and D.S. Conceived and designed the experiments: K.Y. and D.S. Performed the experiments: K.Y., K.I., T.M., and S.A. Analyzed the data: K.Y., T.M., and S.A. Contributed reagents/materials/analysis tools: K.Y., D.S. Wrote the paper: K.Y.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yamashiro, K., Ikarashi, K., Makibuchi, T. et al. Transcranial high-frequency random noise stimulation does not modulate Nogo N2 and Go/Nogo reaction times in somatosensory and auditory modalities. Sci Rep 13, 3014 (2023). https://doi.org/10.1038/s41598-023-30261-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-30261-3
- Springer Nature Limited