Abstract
When facing particular combinations of stimuli and responses, people create temporary event-files integrating the corresponding stimulus and response features. Subsequent repetition of one or more of these features retrieves the entire event-file, which impairs performance if not all features are repeated (partial-repetition costs). In the literature, different decay functions have been reported presumably dependent on the type of feature that is repeated (e.g. target vs. distractor features). Here, we use a variant of the S1R1-S2R2 and distractor-response binding task and analyze for the first time target-based and distractor-based event-file decay functions within the same task and sample. While we found evidence for decay functions and also stronger retrieval due to target than distractor repetitions, slopes of the decay functions were comparable suggesting that the decay process itself is equal irrespective of the type of stimulus feature that is repeated. Our study thereby confirms overarching approaches that summarize paradigm specific findings with the same set of core processes.
Similar content being viewed by others
Introduction
Humans organize perception and action in integrated representations of individual, distributed features. These integrated representations of sensorimotor episodes are compiled by binding features of actions and concurrent stimulation into event-files1,2,3,4,5,6. The entire set of features can be retrieved upon re-encountering one of the features, thus creating a cognitive short-cut for efficient action control as the retrieved response features might facilitate responding in the current situation.
In the laboratory, the influence of such event-file binding on action execution is usually measured with sequential tasks. At the first (the prime) event, stimulus- and response features are integrated, so that repetition of any of these features at the second (or probe) event can retrieve the previous episode thereby influencing responses to the probe. These so-called ‘binding effects’ occur via target stimuli and response integrations (e.g.7), but also for distractor stimuli (e.g.,8,9), effect stimuli (e.g.,10), and responses. Even tasks11, and control states12 can be integrated into event-files.
For the present purpose, it is important to note that depending on the type of binding effect, the duration for that the effect can be measured is variable. While response-irrelevant distractor stimuli are reliably integrated with responses (e.g.,8,13,14), distractor-response binding effects equally reliably decay within two seconds after integration15,16,17,18. Bindings between target stimuli and responses can still be measured four seconds after integration19, but seem to have fully decayed after about 5 s20. Neurophysiological evidence suggests that a decay of the neurophysiological correlates of an event file representation starts ~ 500 ms after an event file was built21. In contrast, bindings between individual responses do not seem to decay to a measurable extent within the first six seconds after integration22, also considering the neurophysiological level21,23, and data on control-state bindings seem to be in line with such response bindings24.
In this article we focus on stimulus binding effects. The differences in the stability between distractor-based and target-based binding effects is so far not well understood. There are arguments that attention might play a role here25, but one particular tacit assumption has never been challenged, namely that the decay function for all stimulus features is essentially the same. The recent binding and retrieval in action control (BRAC) framework argues to explain a plethora of paradigms with only few core processes6 and tacitly assumes that the decay function and the retrieval process is the same for all features. This assumption is what we want to test and challenge here.
To this end, we combine the S1R1-S2R2 task7 with the distractor-response binding task8 so that in the same task target-based and distractor-based binding effects can be measured. Attention allocation to target stimuli should further their integration and particularly the retrieval26 observable as a main effect for binding type (i.e. target-binding is expected to be way larger than distractor-binding). Varying the response-stimulus-interval (RSI) between prime and probe display should—according to previous studies16,20—lead to decreased binding effects for both types of effects (observable as a main effect for RSI). The crucial question is whether the decay is comparable (that is the interaction between RSI and binding type or the slope of the decay function). If the same decay function underlies distractor-based and target-based bindings we should not find an interaction. This would suggest that decay processes are independent of the type of stimulus features, which would lend support for overarching approaches that summarize paradigm specific findings with the same set of core processes—such as BRAC.
Experiment
Method
Participants
Distractor-response binding effects for horizontally flanking distractors ranged between dz = 0.7 and dz = 1.0 in past studies27,28. Binding effects of response and target feature are typically even larger than those between response and distractor feature (see20). Yet, effect sizes in decay rates in previous studies ranged between dz = 0.3 and dz = 1.2 so we planned for a sample size, sufficient to find at least a medium sized effect, dz = 0.4, assuming alpha = 0.05 and a power of 1 − ß = 0.90. A power analysis with the program G*Power revealed that at least 55 participants were necessary29. We recruited fifty-six students (46 women) from the University of Trier. Participants’ median age was 24 years with a range from 18 to 38 years. All participants took part in exchange for partial course credit or monetary compensation. The experiment followed the ethical guidelines of the University of Trier and was in accordance with the guidelines of the German Psychology Association (DGPs). We adhered the recommendations of the German Research Council (DFG) that waive the need for ethic approval of behavioral, non-invasive experiments with non-emotional stimuli. Informed consent was obtained. This study was not preregistered.
Design
The design comprised four within-subjects factors, namely response relation (response repetition vs. response change), target relation (repetition vs. change), distractor relation (repetition vs. change) and response-stimulus interval (RSI) between prime response R1 and presentation of probe stimulus S2 (500 ms vs. 2000 ms).
Materials
The experiment was conducted using the E-prime 2.0 software. Instructions were shown in white on black background on a standard TFT screen. Target stimuli were the letters D and K, presented in red, distractors were the letters G and H, presented in green. R1 response identity (left or right) was indicated via three left or right pointing arrows (<<<or>>>). All letters subtended a horizontal visual angle of 0.6° to 0.7° and a vertical visual angle of 0.6°. Each array of left or right pointing arrows had a horizontal visual angle of 1.7° and a vertical visual angle of 0.5°. Viewing distance was approximately 60 cm.
Procedure
Participants were tested individually in soundproof chambers. Instructions were given on the screen. Participants were instructed to place their left index finger on the D key and their right index finger on the K key of a standard computer keyboard. Participants always saw the letter D or the letter K that was horizontally flanked by two additional letters (either G on both sides or H on both sides). These letter-triplets were presented in the center of the display. The procedure was adapted from7. Participants’ task was to execute a pre-cued response R1 upon presentation of S1 and to respond to the identity of the central target letter by pressing a key with the corresponding finger upon presentation of S2. Participants were instructed to react as quickly and as correctly as possible.
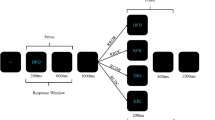
Each trial was started self-paced by the participant and featured a S1R1-S2R2 sequence with the following events (see Fig. 1a). After participants started the trial by pressing the space bar, three arrows (<<<or>>>) appeared for 1500 ms in the middle of the screen, indicating the response required upon S1 presentation. After a 1000 ms blank interval, the S1 letter triplet appeared and stayed on the screen until the participant responded. Then a fixation cross was presented for either 500 ms or 2000 ms (according to the RSI condition), before the S2 letter-triplet appeared and stayed on the screen until the participant responded to the target identity. In response repetition trials, the same response was indicated by the cue (R1) and required by the identity of the target letter in S2 (R2); in response change trials, R1 and R2 differed. In target repetition trials, the S2 target was presented as the central letter in S1; in target change trials the S2 target differed from the central letter in S1. Note that because R1 was cued while R2 was a discrimination response, it is possible to separately repeat or change targets and responses. In distractor repetition trials the flanking letters were repeated from S1 to S2 and in distractor change trials the flanking letters differed between S1 and S2. The orthogonal variation of the factors response relation, target relation and distractor relation resulted in eight conditions: response repetition with target repetition and distractor repetition (RrTrDr), response repetition with target repetition and distractor change (RrTrDc), response repetition with target change and distractor repetition (RrTcDr), response repetition with target change and distractor change (RrTcDc), response change with target repetition and distractor repetition (RcTrDr), response change with target repetition and distractor change (RcTrDc), response change with target change and distractor repetition (RcTcDr), and response change with target change and distractor change (RcTcDc). Each participant completed one experimental block including a 500 ms RSI (128 trials) and another including a 2000 ms RSI (128 trials) between R1 execution and S2 presentation onset. Block order was counterbalanced across participants. Before the experiment started, participants worked through 32 practice trials with the first block’s RSI. Before the second block started, participants worked through an additional 8 practice trials that included the RSI of the second block. Practice trials were a subset of the experimental trials.
(a) Sequence of events in one trial. Participants executed a pre-cued and prepared response upon presentation of S1 and gave a discrimination response to S2 (the central letter). Flanking distractor letters were to be ignored. This is an example for a response repetition trial with distractor change. White is depicted in black and black is depicted in white. Stimuli are not drawn to scale. (b) Target-based binding effects and distractor-based binding effects in milliseconds as a function of RSI (blue indicates RSI 500 ms while orange indicates RSI 2000 ms) depicted at the level of individual effects, at the group level via boxplots, and as a frequency distribution. Binding effects are computed as the interaction of stimulus relation × response relation. * indicates p-values < 0.05.
Results
Individual reaction time distributions were smoothed by applying non-parametric outlier detection to the lower and upper part of the distribution (according to30). In addition, only trials with correct prime and probe responses were considered for RTs. These criteria led to the exclusion of 13.1% (SD 12.6%) of all trials.
RTs and error rates were combined into inverse efficiency scores (see Supplement for RT and error data) as probe error rate was only 3.6% (SD 2.1%)31. Target-binding effects were built by computing the interaction of response relation × target relation, distractor-binding effects were built by computing the interaction of response relation × distractor relation. Target-based binding effects were significant for both RSIs, t(54) = 8.12, p < 0.001, for the RSI 500 ms, and t(54) = 7.411, p < 0.001, for the RSI 2000 ms interval while distractor-based binding effects were significant for the RSI 500 ms condition, t(55) = 2.18, p = 0.033, but not for the RSI 2000 ms condition, t(55) = 0.33, p = 0.741. These binding effects were then submitted to a 2 (stimulus type: target vs. distractor) × 2 (RSI: 500 vs. 2000 ms) repeated measures ANOVA. The main effect for stimulus type was significant, F(1,54) = 65.613, p < 0.001, ω2 = 0.359, showing that binding effects were larger for targets than for distractors (this can be seen when you compare the average target binding effect with the average distractor binding effect as reported for the t-tests above). The main effects of RSI was also significant, F(1,54) = 8.132, p = 0.006, ω2 = 0.054, showing that binding effects were larger for shorter RSIs than for longer RSIs, thus replicating previous findings on decay functions (this can be seen when you compare the binding effects at short RSI with the ones at long RSI as reported for the t-tests above). RSI and stimulus type did not interact, F(1,54) = 2.435, p = 0.124, ω2 = 0.012 (see Fig. 1b). To corroborate these findings a Bayesian repeated measures ANOVA was run. A model that included only both main effects was the best model (BFM = 4.88), while adding the interaction term did not differ from the best model (BF10 = 0.661; according to BF conventions32). This pattern reflects that the differences between target binding effects at short and long RSIs is comparable to the differences between distractor binding effects at short and long RSIs.
Discussion
As expected we observed and replicated that targets produce larger binding effects than distractors (25, e.g.,26) and that binding effects on average diminish over time16,20. More intriguing is that we did not find an interaction of stimulus type and RSI suggesting that the decay function for stimulus features is essentially the same for targets and distractors that means the binding effects for targets and distractors diminish to a comparable degree over time albeit they start at different levels. Put differently, the tacit assumption of only one decay function for stimulus features that is inherent in most action control approaches was confirmed. The observed differences for distractor-based and target-based binding effects can be attributed to differences in attention allocation—obviously relevant stimuli receive more attention and hence might have just a head-start for the binding and retrieval while the decay function itself is independent of the stimulus type.
Yet, we have to acknowledge that a study with more test power might find differences in the decay function. The crucial test for the different decay functions is the Bayesian analysis and this analysis suggested that the model without the interaction term is the best while adding the interaction term does not improve the model fit—still the evidence for this null effect was rather small or anecdotal according to Schönbrodt and Wagenmarkers33. Further research with more test power and more time points might pinpoint possible differences in stimulus dependent decay functions. However, our study suggests that if such differences exist they are small.
The different decay functions for stimulus-based versus response-based bindings as noted in the introduction might be due to the fact that these bindings serve different functions. Following research on hierarchical action representations (see34,35), response-response bindings might enable the formation of complex action representations, which are supposed to group sub-elements of an extended action by chunking multiple simple responses. For such higher-level representations, temporal stability is relevant, because these representations merge temporally distant events. However, at the level of stimulus representations, quick disintegration of stimulus–response bindings should prevent interference between individual episodes (see20).
The tacit assumption of only one decay function for stimulus features in action control as suggested by the BRAC framework6 or the theory of event-coding (TEC; 1) was confirmed. This is an important theoretical advance as overarching approaches trying to summarize and integrate action control paradigms need evidence for core processes that are not paradigm- or stimulus-specific. Stimuli are stimuli—differences between them are due to attentional allocation or their role as response-relevant or response-irrelevant stimuli etc. but the underlying decay and retrieval function is the same (Supplementary Tables).
Data availability
Raw data and aggregated data are available on OSF, Link: https://doi.org/10.17605/OSF.IO/Z6HQ8.
References
Hommel, B. Event files: Feature binding in and across perception and action. Trends Cogn. Sci. 8(11), 494–500 (2004).
Hommel, B., Müsseler, J., Aschersleben, G. & Prinz, W. The Theory of Event Coding (TEC): A framework for perception and action planning. Behav. Brain Sci. 24(5), 849–878 (2001).
Kahneman, D. & Treisman, A. M. Changing views of attention and automaticity. In Varieties of Attention (eds Parasuraman, R. & Davies, D. R.) 29–61 (Academic Press, 1984).
Kahneman, D., Treisman, A. M. & Gibbs, B. J. The reviewing of object files: Object-specific integration of information. Cogn. Psychol. 24(2), 175–219 (1992).
Logan, G. D. Toward an instance theory of automatization. Psychol. Rev. 95(4), 492–527 (1988).
Frings, C. et al. Binding and retrieval in action control (BRAC). Trends Cogn. Sci. 24(5), 375–387 (2020).
Hommel, B. Event files: Evidence for automatic integration of stimulus-response episodes. Vis. Cogn. 5(1–2), 183–216 (1998).
Frings, C., Rothermund, K. & Wentura, D. Distractor repetitions retrieve previous responses to targets. Q. J. Exp. Psychol. 60(10), 1367–1377 (2007).
Schreiter, M. L., Chmielewski, W. X., Ward, J. & Beste, C. How non-veridical perception drives actions in healthy humans: Evidence from synaesthesia. Philos. Trans. R. Soc. Lond. B Biol. Sci. 374(1787), 20180574 (2019).
Herwig, A. & Waszak, F. Action-effect bindings and ideomotor learning in intention- and stimulus-based actions. Front. Psychol. 3, 444 (2012).
Waszak, F., Hommel, B. & Allport, A. Task-switching and long-term priming: Role of episodic stimulus–task bindings in task-shift costs. Cogn. Psychol. 46(4), 361–413 (2003).
Dignath, D., Johannsen, L., Hommel, B. & Kiesel, A. Reconciling cognitive-control and episodic-retrieval accounts of sequential conflict modulation: Binding of control-states into event-files. J. Exp. Psychol. Hum. Percept. Perform. 45(9), 1265–1270 (2019).
Giesen, C. & Rothermund, K. Affective matching moderates S-R binding. Cogn. Emot. 25(2), 342–350 (2011).
Moeller, B., Rothermund, K. & Frings, C. Integrating the irrelevant sound. Exp. Psychol. 59(5), 258–264 (2012).
Pastötter, B., Moeller, B. & Frings, C. Watching the brain as it (un)binds: Beta synchronization relates to distractor-response binding. J. Cogn. Neurosci. 33(8), 1581–1594 (2021).
Frings, C. On the decay of distractor-response episodes. Exp. Psychol. 58(2), 125–131 (2011).
Moeller, B. & Frings, C. Dissociation of binding and learning processes. Atten. Percept. Psychophys. 79(8), 2590–2605 (2017).
Moeller, B., Pfister, R., Kunde, W. & Frings, C. A common mechanism behind distractor-response and response-effect binding?. Atten. Percept. Psychophys. 78(4), 1074–1086 (2016).
Hommel, B. & Colzato, L. S. Visual attention and the temporal dynamics of feature integration. Vis. Cogn. 11(4), 483–521 (2004).
Hommel, B. & Frings, C. The disintegration of event files over time: Decay or interference?. Psychon. Bull. Rev. 27(4), 751–757 (2020).
Takacs, A., Mückschel, M., Roessner, V. & Beste, C. Decoding stimulus-response representations and their stability using EEG-based multivariate pattern analysis. Cereb. Cortex Commun. 1(1), 016 (2020).
Moeller, B. & Frings, C. Response-response bindings do not decay for 6 seconds after integration: A case for bindings’ relevance in hierarchical action control. J. Exp. Psychol. Hum. Percept. Perform. 47(4), 508–517 (2021).
Geißler, C. F., Frings, C. & Moeller, B. Illuminating the prefrontal neural correlates of action sequence disassembling in response-response binding. Sci. Rep. 11(1), 22856 (2021).
Schiltenwolf, M. et al. Contextualized cognitive control: Retrieved or disrupted control-states? Manuscript submitted for publication (2022).
Henson, R. N., Eckstein, D., Waszak, F., Frings, C. & Horner, A. J. Stimulus-response bindings in priming. Trends Cogn. Sci. 18(7), 376–384 (2014).
Ihrke, M., Behrendt, J., Schrobsdorff, H., Michael Herrmann, J. & Hasselhorn, M. Response-retrieval and negative priming. Exp. Psychol. 58(2), 154–161 (2011).
Frings, C. & Rothermund, K. To be or not to be…included in an event file: Integration and retrieval of distractors in stimulus-response episodes is influenced by perceptual grouping. J. Exp. Psychol. Learn. Mem. Cogn. 37(5), 1209–1227 (2011).
Moeller, B. & Frings, C. Attention meets binding: Only attended distractors are used for the retrieval of event files. Atten. Percept. Psychophys. 76(4), 959–978 (2014).
Faul, F., Erdfelder, E., Lang, A.-G. & Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 39(2), 175–191 (2007).
Tukey, J. W. Exploratory Data Analysis. Addison-Wesley Series in Behavioral Science Quantitative Methods (Addison-Wesley, 1977).
Townsend, J. T. & Ashby, F. G. Stochastic Modeling of Elementary Psychological Processes (CUP Archive, 1983).
Wagenmakers, E.-J. et al. Bayesian inference for psychology. Part II: Example applications with JASP. Psychon. Bull. Rev. 25(1), 58–76 (2018).
Schönbrodt, F. D. & Wagenmakers, E.-J. Bayes factor design analysis: Planning for compelling evidence. Psychon. Bull. Rev. 25(1), 128–142 (2018).
Lashley, K. S. The problem of serial order in behavior. In Cerebral Mechanisms in Behavior: The Hixon Symposium Vol. 115 (ed. Jeffress, L. A.) 112–147 (Wiley, 1952).
Cooper, R. P. & Shallice, T. Hierarchical schemas and goals in the control of sequential behavior. Psychol. Rev. 113(4), 887–916 (2006) (discussion 917–31).
Funding
Open Access funding enabled and organized by Projekt DEAL. The research reported in this article was supported by grants of the Deutsche Forschungsgemeinschaft (FOR 2790) to CF. CB and AM were supported by grants of the Deutsche Forschungsgemeinschaft (FOR 2698). Correspondence concerning this article may be addressed to Christian Frings, University of Trier, Faculty of Behavioral Sciences, Department of Psychology, Universitätsring 15, D-54296 Trier, Germany (Email: chfrings@uni-trier.de).
Author information
Authors and Affiliations
Contributions
C.F. funding acquisition, conceptualization, writing (original draft), formal analysis, review and editing, supervision. B.M. investigation, data curation, writing (original draft). B.P., C.B., A.M. writing, review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Frings, C., Moeller, B., Beste, C. et al. Stimulus decay functions in action control. Sci Rep 12, 20139 (2022). https://doi.org/10.1038/s41598-022-24499-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-24499-6
- Springer Nature Limited
This article is cited by
-
Towards a systematization of brain oscillatory activity in actions
Communications Biology (2023)