Abstract
Bronchiectasis show various ventilatory disorders in pulmonary function. The characteristics and severity of patients with bronchiectasis according to these pulmonary dysfunctions are still very limited. This study aimed to evaluate the clinical, radiologic feature and the disease severity of patients with bronchiectasis according to spirometric patterns. We retrospectively evaluated 506 patients with bronchiectasis who underwent pulmonary lung function test (PFT) at a referral hospital between 2014 to 2021. The results showed that cylindrical type was the most common (70.8%) type of bronchiectasis on chest Computed tomography (CT), and 70% of patients had bilateral lung involvement. On the other hand, obstructive ventilatory disorder was the most common (51.6%), followed by normal ventilation (30%) and restrictive ventilatory disorder (18.4%). The modified Medical Research Council (mMRC) was highest in patients with obstructive ventilatory disorders, Modified Reiff score [median (interquartile range)] [6 (3–10), P < 0.001], FACED (FEV1, Age, Chronic colonization, Extension, and Dyspnea) score [3 (1–4), P < 0.001], and Bronchiectasis Severity (BSI) score [8 (5–11), P < 0.001] showed significantly highest values of obstructive ventilatory disorder rather than restrictive ventilatory disorder and normal ventilation. More than half of patients with bronchiectasis had obstructive ventilatory disorder. Bronchiectasis with obstructive ventilatory disorders has more dyspnea symptom, more disease severity and more radiologic severity. There was no significant association between spirometric pattern and radiologic type, but the more severe the radiologic severity, the more severe the lung function impairment.
Similar content being viewed by others
Introduction
Non-cystic fibrosis bronchiectasis (hereafter referred to as bronchiectasis) is a chronic respiratory disease characterized by permanent dilatation of the bronchi and chronic respiratory symptoms, such as cough, expectoration of sputum, and dyspnea. The inherent dysfunction in mucociliary clearance leads to persistent bacterial infection, chronic inflammation of the bronchial tree, and progressive tissue destruction. Thus, persistent airway destruction leads to decreased lung function and respiratory failure1,2.
Previous studies have reported that the morphological changes in bronchiectasis are associated with pulmonary dysfunction3,4,5. Research suggests that the development of bronchiectasis is promoted by the early involvement of the lymphoid follicles in the small airways, which gradually leads to the obstruction of the more distal airways6. Accordingly, it has been known that bronchiectasis is accompanied by airflow limitation7. As a result, forced expiratory volume in 1 s (FEV1) was used to evaluate pulmonary dysfunction in patients with bronchiectasis, as a same line, FEV1 was included in Bronchiectasis Severity Index (BSI) score and FACED (FEV1, Age, Chronic colonization, Extension, and Dyspnea) score to evaluate the severity and prognosis of patient with bronchiectasis8,9. Moreover, recent studies have reported that patients with bronchiectasis exhibit various ventilatory disorders on pulmonary function tests, such as obstructive, restrictive, and mixed ventilatory disorder4,10,11,12. Although various ventilatory disorders of bronchiectasis have been reported as described above, studies on the characteristics and severity of patients with bronchiectasis according to these pulmonary dysfunctions are still very limited.
Hence, we investigated the clinical and radiological features of patients with bronchiectasis according to the spirometric pattern. Moreover, we further evaluated the disease severity in patients with bronchiectasis according to spirometric patterns.
Results
Baseline characteristics
The baseline characteristics of 506 patients with bronchiectasis are presented in Table 1. The median age was 66 years interquartile range (IQR 53–79 years), and 254 (50.2%) patients were male. The median body mass index (BMI) was 23.0 (IQR 19.3–26.7). Current and ex-smokers accounted for 34.8% of the study population, and 23.7% of patients had a history of tuberculosis. The distribution of comorbidities in patients with bronchiectasis was as follows: chronic obstructive pulmonary disease (COPD) (51.6%), followed by cardiovascular disease (37.7%), asthma (16.2%), and diabetes mellitus (12.6%). Cylindrical bronchiectasis was the most common (70.8%) morphology on chest CT, and 70% of patients exhibited bilateral lung involvement. The pulmonary function parameters were as follows: forced vital capacity (FVC) % predicted, 84% (IQR 70–95); FEV1/FVC, 73% (IQR 56–90); and FEV1% predicted, 69% (IQR 55–78). Microorganisms were identified in 32.4% of patients: Pseudomonas aeruginosa (18.0%) was the most frequently identified species, followed by Klebsiella pneumoniae (8.5%), and Staphylococcus aureus (4.9%). NTM were identified in 10.1% of patients, of which Mycobacterium avium complex (MAC) was the most common type (8.3%).
Clinical characteristics according to the spirometric pattern
As shown in Table 2, 152 patients (30.0%) with bronchiectasis had normal ventilation, 261 patients (51.6%) had obstructive ventilatory disorder, and 93 patients (18.4%) had restrictive ventilatory disorder. The median ages of patients with normal ventilation, obstructive ventilatory disorder, and restrictive ventilatory disorder were 66 (IQR 58–72), 68 (IQR 62–75), and 64 (IQR 56–71) years, respectively (P < 0.001). The proportion of males was highest in patients with obstructive ventilatory disorder, followed by those with normal ventilation and those with restrictive ventilatory disorder (62.1%, 41.4%, and 31.2%, respectively, P < 0.001). The proportion of current- or ex-smokers was higher among patients with obstructive ventilatory disorders compared to that in patients with normal ventilation or restrictive ventilatory disorders (43.7%, 28.3%, and 20.4%, respectively, P < 0.001). The prevalence of COPD (100%), asthma (21.8%), cardiovascular disease (43.7%), and diabetes mellitus (16.9%) was the highest in patients with obstructive ventilatory disorder from amongst the entire study population. Most of the microorganisms were cultured in the obstructive ventilatory disorder, followed by restrictive ventilatory disorder and normal ventilation. (41.4%, 32.3% and 15.8%, respectively, P < 0.001). The frequency of culturing for P. aeruginosa (23.8%, 17.2% and 8.6%, respectively, P < 0.001), and others (10.3%, 6.5% and 1.3%, respectively, P < 0.001) was significantly higher in the obstructive ventilatory disorder and restrictive ventilatory disorder than that in the normal ventilation. The prevalence of NTM (20.4%) was the highest in patients with restrictive ventilatory disorder from amongs the entire study population.
Respiratory symptoms, radiological features, and disease severity scales in patients with bronchiectasis according to the spirometric pattern
As shown in Table 3, the modified Medical Research Council (mMRC) was the highest in patients with obstructive ventilatory disorder, followed by those with restrictive ventilatory disorder and normal ventilation [median (IQR)] [1 (1–2), 1 (0–2), and 0 (0–1), respectively; P < 0.001]. The modified Reiff score [6 (3–10), P < 0.001)]; FACED score [3 (1–4), P < 0.001], and BSI score [8 (5–11), P < 0.001] were significantly higher in the obstructive ventilatory disorder group than those in the restrictive ventilatory disorder and normal ventilation groups. There was no significant correlation between the lung function pattern and bronchiectasis type. The proportion of bilateral lung involvement was the highest in patients with obstructive ventilatory disorder, followed by those with restrictive ventilatory disorder and normal ventilation (79.7%, 72.0%, and 52.0%, respectively, P < 0.001). The disease severity indices, i.e., the BSI and FACED scores, were the highest in patients with obstructive ventilatory disorder, followed by those with restrictive ventilatory disorder and normal ventilation [8 (5–11), 6 (4–9) and 4 (2–7), respectively, P < 0.001 for the BSI score; 3 (1–4), 1 (1–3) and 1 (0–2), respectively, P < 0.001 for the FACED score].
Correlation between the modified Reiff score and lung function
As shown in Fig. 1, the FVC %, FEV1/FVC %, and FEV1% declined significantly with the elevation in the modified Reiff score (P < 0.001 for all). The FEV1 declined significantly with the increase in the BSI score (r = − 0.442, respectively; P < 0.001) in the obstructive pulmonary disorder group. The FVC decreased significantly with the increase in the BSI score (r = − 0.363, P < 0.001) in the restrictive pulmonary disorder group (Supplementary Fig. 1).
Correlation between the modified Reiff score and lung function. (A) Relationship between the modified Reiff score and FVC %, predicted (B) Relationship between the modified Reiff score and FEV1/FVC % (C) Relationship between the modified Reiff score and FEV1 %, predicted Abbreviations: FVC, forced vital capacity; FEV1, forced expiratory volume in 1 second.
Discussion
To the best of our knowledge, this is the first study to evaluate the clinical characteristics, radiological features, and disease severity according to the spirometric pattern in patients with bronchiectasis. Approximately 52% and 18% patients with bronchiectasis had obstructive and restrictive ventilatory disorders, respectively. The frequency of dyspnea, higher disease severity (including high BSI and FACED scores), higher sputum culture positivity for Pseudomonas aeruginosa, and greater radiological severity (i.e., high modified Reiff scores) were predominant in bronchiectasis accompanied by obstructive ventilatory disorder. On the other hand, bronchiectasis with restrictive ventilatory disorder was associated with a high prevalence of NTM disease. There was no significant correlation between the spirometric patterns and the type of bronchiectasis; however, lung function decreased with the increase in the modified Reiff score.
Patients with bronchiectasis frequently experience structural and functional lung damage that vary in severity2,8,9. Our notable finding was that more than half of patients (52%) with bronchiectasis showed obstructive ventilatory disorder. In previous studies, approximately up to 35% of bronchiectasis were known to experience obstructive ventilatory disorders5,12,13,14,15. Bronchiectasis is known to manifest as an obstructive ventilatory disorder due to continuous airway destruction and distortion caused by chronic inflammation and abnormal mucociliary clearance2. In our study, the bilateral lung involvement was high at 70% in patients with bronchiectasis. Bilateral lung involvement resulted in widespread inflammation of the bronchial tree and damage to the lung parenchyma, which is believed to have resulted in pulmonary impairment of obstructive ventilatory disorder. Additionally, patients with bronchiectasis may also experience restrictive ventilatory disorder. Bronchiectasis accompanied by atelectasis and pleural disease, parenchymal scarring, and peribronchial fibrosis is thought to be associated with restrictive ventilatory disorder11. In contrast with obstructive ventilatory disorder, only a few studies have described restrictive ventilatory disorder and the presumable prevalence was reported to be 8–15% among patients with bronchiectasis12,13,15. Our study also reported a similar result, i.e., 18% of patients with bronchiectasis had restrictive ventilatory disorder; however, verification through a large-scale study is necessary since it accounted for a small proportion of the study population.
Our notable finding was that bronchiectasis with obstructive ventilatory disorder was characterized by greater symptoms of dyspnea, disease severity and radiological severity. We measured the degree of dyspnea using the mMRC score, which was significantly higher in bronchiectasis with obstructive ventilatory disorder. mMRC is a subjective measure of dyspnea in COPD, which is known to be associated with the decrease in FEV116. Similarly, in our study, the degree of dyspnea as measured by the mMRC score was higher in the obstructive ventilatory disorder group with low FEV1. Moreover, we evaluated disease severity using the BSI and FACED scores. The FACED score [FEV1% predicted (F), age (A), chronic colonization by Pseudomonas aeruginosa (C), extension of the disease by radiological assessment (E) and dyspnea (D)]9 is a five-point instrument that predicts the probability of all-cause mortality after 5 years of follow-up, whereas the BSI8 is a seven-point scale that identifies patients with bronchiectasis at risk for future mortality, hospitalization, exacerbations, and deterioration in the quality of life. Our study revealed high disease severity and poor prognosis, as measured using the BSI and FACED, in bronchiectasis with obstructive ventilatory disorder. These results may be related to low BMI, low FEV1, high mMRC score, and high Pseudomonas aeruginosa infection, which are the clinical features of bronchiectasis with obstructive ventilatory disorder ascertained by our study. Although most studies have reported that the FEV1 decreases with the increase in CT severity11,17,18, no study has analyzed the relationship between CT severity and lung function in bronchiectasis with obstructive ventilatory disorder. Our study is the first to demonstrate the impairment in lung function, including FVC and FEV1, and CT severity using the modified Reiff score in bronchiectasis with obstructive ventilatory disorder. Therefore, these findings emphasize the importance of the management of lung function in bronchiectasis with obstructive ventilatory disorder.
Another interesting finding of our study was that there was no significant correlation between the spirometric pattern and radiological type of bronchiectasis. However, the increase in the radiological severity, which was measured by the modified Reiff score, was accompanied by the increase in the severity of impairment of lung function. Bronchiectasis is known to manifest as a combination of three morphologies, viz., cylindrical, varicose, and cystic19. Few studies have investigated the relationship between the type of bronchiectasis and the spirometric pattern. In contrast with our study, other studies have suggested that cylindrical bronchiectasis is associated with obstructive ventilatory disorder15,20. However, it is difficult to generalize the results of studies that incorporated a small patient population. Thus, large-scale research on the association between the radiological features of bronchiectasis and lung function is warranted. Our study also showed that the severity of lung function impairment increased with the radiological extent of bronchiectasis. Inflammation and destruction of the lung parenchyma and airways become more severe with the expansion in the extent of bronchiectasis, which may lead to a decrease in lung function. Other studies have also reported similar results11,17. Thus, we can infer that the extent of bronchiectasis, rather than the type of bronchiectasis, affects lung function.
This study had several limitations. First, it was conducted at a single center in South Korea. Second, its retrospective and observational design could have introduced potential biases. For example, the proportion of patients with obstructive ventilatory disorder was high in this bronchiectasis population, possibly because this study was conducted in patients with bronchiectasis who underwent pulmonary function testing. Third, the BSI and FACED scores were high in the bronchiectasis with obstructive ventilatory disorder group. FEV1 is included in the BSI and FACED evaluation criteria, which may be a confounding factor. However, our study showed that low BMI, high mMRC score, and high prevalence of Pseudomonas aeruginosa infection were clinical features associated with the severity of bronchiectasis with obstructive ventilatory disorder, even after excluding these confounding factors.
In conclusion, among patients with bronchiectasis, 52% had obstructive ventilatory disorder and 18% had restrictive ventilatory disorder. Bronchiectasis with obstructive ventilatory disorder had more dyspnea, and greater disease and radiological severity. The extent of bronchiectasis rather than radiologic type of bronchiectasis affects lung function impairment, however more research is needed.
Methods
Study setting and patients population
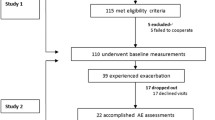
We retrospectively reviewed the medical records of 1369 patients at Chungbuk National University Hospital (a 793-bed referral hospital) in Cheongju, Republic of Korea, in whom bronchiectasis was diagnosed between January 2014 to August 2021. Of those patients, those not undergoing pulmonary function test (n = 863) were excluded, a total of 506 patients were included in the study. We classified patients with bronchiectasis into three groups according to spirometric patterns (Fig. 2).
Ethics declarations
The study protocol was approved by the Institutional Review Board of Chungbuk National University Hospital (IRB No. 2021-09-023) and was conducted in accordance with the amended Declaration of Helsinki (as revised in 2013). Patient information was anonymized and de-identified prior to analysis. Therefore, the need for informed consent was waived owing to the retrospective nature of the study.
Pulmonary function tests
Pre-bronchodilator and post-bronchodilator spirometry was performed according to American Thoracic Society/European criteria21. The absolute values of FEV1 and forced vital capacity (FVC) were recorded, and the percentage of predicted values for FEV1 and FVC were calculated using an automatic calculator with a reference equation obtained from a representative Korean sample22. Normal ventilation was defined as post-bronchodilator FEV1/FVC ≥ 0.70 and FVC ≥ 80% predicted. Obstructive ventilatory disorder was defined as post-bronchodilator FEV1/FVC < 0.70. Restrictive ventilatory disorder was defined as FEV1/FVC ≥ 0.7 and FVC < 80% predicted23.
Bronchiectasis severity: radiological and disease severity
The radiological severity of bronchiectasis was measured using the modified Reiff score24. Chest computed tomography (CT) was used to evaluate the radiological extent of bronchiectasis in all participants. The number of lobes involved (the lingula was considered to be a separate lobe) and degree of dilatation (tubular: 1, varicose: 2, and cystic: 3) were determined by three researchers, including two pulmonologists (SHK and BY) and one radiologist (JYY), based on the consensual interpretation of the chest CT image. The FACED and BSI scores were calculated to assess the clinical status and severity of the bronchiectasis, based on previous studies8,9.
Microbiology
Spontaneous sputum or lower tract specimens (bronchoalveolar lavage) were obtained from all patients. The specimens were subjected to microbiological analyses according to standard methods25. Conventional semi-qualitative bacterial and fungal cultures were performed. All samples underwent initial Gram staining prior to sputum culture, if the Murray and Washington criteria were met26. Nontuberculous mycobacteria (NTM) lung disease was diagnosed using the microbiological criteria provided by the American Thoracic Society and Infectious Disease Society of America; (1) two positive sputum cultures, (2) one positive bronchial wash or lavage, (3) compatible mycobacterial histological features such as granulomatous inflammation, and positive results on acid-fast bacilli lung biopsy and/or lung biopsy culture, and (4) more than one positive sputum culture or bronchial wash27. Chronic cavitary pulmonary aspergillosis is one or more pulmonary cavities possibly containing one or more aspergillomas with serological or microbiological evidence implicating Aspergillus spp. with significant pulmonary symptoms and overt radiological progression over at least 3 months of observation28.
Statistical analysis
Data were presented as the median and interquartile range (IQR) for continuous variables and frequency (percentage) for categorical variables. Continuous variables were compared using the Kruskal–Wallis test and the Pearson chi-squared test or Fisher exact test was used for categorical variables. Moreover, P-values for comparing the three ventilatory disorder groups were adjusted using Bonferroni correction. Spearman's rho correlation method was used to confirm the existence of a correlation between the BSI score and lung function tests. All tests were two-sided, and P-values < 0.05 were considered statistically significant. All statistical analyses were conducted using IBM SPSS Statistics for Windows (version 21.0; IBM Corp., Armonk, NY, USA) and STATA (version 15; Stata Corp., College Station, TX, USA).
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Cole, P. Inflammation: A two-edged sword—The model of bronchiectasis. Eur. J. Respir. Dis. Suppl. 147, 6–15 (1986).
Chalmers, J. D., Chang, A. B., Chotirmall, S. H., Dhar, R. & McShane, P. J. Bronchiectasis. Nat. Rev. Dis. Primer. 4, 1–18 (2018).
Ooi, G. C. et al. High-resolution CT quantification of bronchiectasis: Clinical and functional correlation. Radiology 225, 663–672 (2002).
Sheehan, R. E. et al. A comparison of serial computed tomography and functional change in bronchiectasis. Eur. Respir. J. 20, 581–587 (2002).
Lee, J.-H., Kim, Y.-K., Kwag, H.-J. & Chang, J.-H. Relationships between high-resolution computed tomography, lung function and bacteriology in stable bronchiectasis. J. Korean. Med. Sci. 19, 62–68 (2004).
Whitwell, F. A study of the pathology and pathogenesis of bronchiectasis. Thorax 7, 213 (1952).
Culiner, M. M. Obliterative bronchitis and bronchiolitis with bronchiectasis. Dis. Chest. 44, 351–361 (1963).
Chalmers, J. D. et al. The bronchiectasis severity index. An international derivation and validation study. Am. J. Respir. Crit. Care. Med. 189, 576–585 (2014).
Martínez-García, M. Á. et al. Multidimensional approach to non-cystic fibrosis bronchiectasis: The FACED score. Eur. Respir. J. 43, 1357–1367 (2014).
Guan, W. J. et al. Characterization of lung function impairment in adults with bronchiectasis. PLoS One 9, e113373 (2014).
Habesoglu, M. A., Tercan, F., Ozkan, U. & Fusun, E. O. Effect of radiological extent and severity of bronchiectasis on pulmonary function. Multidiscip. Respir. Med. 6, 1–7 (2011).
Radovanovic, D. et al. A comprehensive approach to lung function in bronchiectasis. Respir. Med. 145, 120–129 (2018).
Miller, M. R. et al. Standardisation of spirometry. Eur. Respir. J. 26, 319–338 (2005).
Choi, J. K., Paek, D. & Lee, J. O. Normal predictive values of spirometry in Korean population. Tuberc. Respir. Dis. 58, 230–242 (2005).
Sim, Y. S. et al. Spirometry and bronchodilator test. Tuberc. Respir. Dis. 80, 105–112 (2017).
Reiff, D. B., Wells, A. U., Carr, D. H., Cole, P. J. & Hansell, D. M. CT findings in bronchiectasis: Limited value in distinguishing between idiopathic and specific types. AJR. Am. J. Roentgenol. 165, 261–267 (1995).
Patel, J. B., Cockerill, F. & Bradford, P. A. Performance standards for antimicrobial susceptibility testing. In Twenty-Fifth Informational Supplement (2015).
Murray, P. R. & Washington, J. A. Microscopic and bacteriologic analysis of expectorated sputum. Mayo Clin. Proc. 50, 339–344 (1975).
Griffith, D. E. et al. An official ATS/IDSA statement: Diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am. J. Respir. Crit. Care. Med. 175, 367–416 (2007).
Yang, B. et al. Factors associated with bronchiectasis in Korea: A national database study. Ann. Transl. Med. 8, 1350 (2020).
Lee, H. et al. Characteristics of bronchiectasis in Korea: First data from the Korean Multicentre Bronchiectasis Audit and Research Collaboration registry and comparison with other international registries. Respirology 26, 619–621 (2021).
Lynch, D. A. et al. Correlation of CT findings with clinical evaluations in 261 patients with symptomatic bronchiectasis. AJR. Am. J. Roentgenol. 173, 53–58 (1999).
Bestall, J. C. et al. Usefulness of the Medical Research Council (MRC) dyspnoea scale as a measure of disability in patients with chronic obstructive pulmonary disease. Thorax 54, 581–586 (1999).
Roberts, H. R. et al. Airflow obstruction in bronchiectasis: Correlation between computed tomography features and pulmonary function tests. Thorax 55, 198–204 (2000).
Ibrahim, R. M., Elnekeidy, A., Rizk, A., Yossef, A. & Abdelrahman, S. Correlation between a proposed MDCT severity score of bronchiectasis and pulmonary function tests. Egypt. J. Radiol. Nucl. Med. 47, 413–420 (2016).
Milliron, B., Henry, T. S., Veeraraghavan, S. & Little, B. P. Bronchiectasis: Mechanisms and imaging clues of associated common and uncommon diseases. Radiographics 35, 1011–1030 (2015).
Nicotra, M. B., Rivera, M., Dale, A. M., Shepherd, R. & Carter, R. Clinical, pathophysiologic, and microbiologic characterization of bronchiectasis in an aging cohort. Chest 108, 955–961 (1995).
Denning, D. W. et al. Chronic pulmonary aspergillosis: Rationale and clinical guidelines for diagnosis and management. Eur. Respir. J. 47, 45–68 (2016).
Acknowledgements
This research was supported by the grant from the National Research Foundation (NRF) of Korea (No.2020R1A5A2017476 to E.-G.K.) and NRF grant funded by the Korea government (MSIT) (No.2022R1F1A1074749 to B.Y.). This research was also supported by Basic Science Research Program through the NRF funded by the Ministry of Education (2021R1I1A3058091) and the grant from Osong Medical Innovation foundation funded by Cheongju City.
Author information
Authors and Affiliations
Contributions
Conceptualization: K.H.C., E.G.K., B.Y. Investigation: S.H.K., B.Y. Formal analysis: S.H.K., B.Y., J.Y.Y. Methodology: K.H.C., S.H.K., B.Y. Supervision: E.G.K., J.Y.Y. Writing-original draft: K.H.C., S.H.K., B.Y. Writing-review and editing: K.H.C., S.H.K., B.H.Y., J.Y.Y., J.Y.C., H.K., Y.M.S., K.M.L.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, SH., Yang, B., Yoo, J. et al. Clinical characteristics, radiological features, and disease severity of bronchiectasis according to the spirometric pattern. Sci Rep 12, 13167 (2022). https://doi.org/10.1038/s41598-022-17085-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-17085-3
- Springer Nature Limited
This article is cited by
-
The clinical characteristics of non-cystic fibrosis bronchiectasis patients with positive serum tumor markers: a retrospective study
BMC Pulmonary Medicine (2024)
-
Correlating Reiff scores with clinical, functional, and prognostic factors: characterizing noncystic fibrosis bronchiectasis severity: validation from a nationwide multicenter study in Taiwan
European Journal of Medical Research (2024)