Abstract
To evaluate the efficiency of a 20 μg hepatitis B vaccine(HepB) for disease prevention in two counties in Henan Province, China. A questionnaire was designed to examine the information of hepatitis B surface antigen (HBsAg) positive pregnant women, and their blood samples were collected to test for hepatitis B e antigen (HBeAg), hepatitis B e antibody, and hepatitis B virus (HBV) DNA. Three doses of 20 μg HepB and one dose of hepatitis B immune globulin(HBIG) were administered to newborns. Blood samples were collected from children one month after their complete immunization to test for HBsAg and hepatitis B surface antibody(HBsAb). A total of 419 HBsAg positive-pregnant women and 430 newborns were investigated. The average age of pregnant women was 29.6 ± 4.3 years, with an HBeAg positive rate of 29.1% (122/419). All newborns received their first dose of 20 μg hepatitis B vaccine and 100 IU HBIG within 12 h after birth. Six infants (1.9%, 6/319) tested positive for HBsAg and negative for HBsAb after one month of receiving the three basic doses of HepB. The geometric mean concentration(GMC) of HBsAb-positive infants was 861.6 mIU/mL, and their HBsAb antibody titers decreased with age. Immunization of children born to HBsAg-positive mothers with 20 μg HepB got the satisfactory effect on preventing mother-to-child transmission.
Similar content being viewed by others
Hepatitis B virus (HBV) infection remains a serious global public health problem1. Persistent HBV infection can cause chronic liver damage, which can lead to cirrhosis and hepatocellular carcinoma. In 2015, the World Health Organization (WHO) estimated that around 257 million people worldwide had chronic HBV infection (defined as hepatitis B surface antigen-positive) and that around 887, 000 people died of liver fibrosis or liver cancer caused by hepatitis B infection2.
Mother-to-child transmission (MTCT) is the main way of HBV transmission. About 90% of infants infected with HBV will develop chronic hepatitis B if no vaccination is done, which is substantially higher than horizontal transmission3,4. The chronic infection rate is 30–50% for children under the age of six, and 5% for adults. MTCT accounts for around 50% of new hepatitis B infections in countries with high prevalence, and one-third in countries with low prevalence5,6. Therefore, prevention of mother-to-child transmission (PMTCT) is essential to stop vertical transmission and reduce the burden of the chronic hepatitis B (CHB). In order to prevent MTCT of hepatitis B, WHO recommends that the hepatitis B vaccine (HepB) should be given within 24 h after birth, even in areas with a low prevalence of hepatitis B. Infants born to hepatitis B surface antigen (HBsAg) positive mothers, especially those who are hepatitis B e antigen (HBeAg) positive, may benefit more from receiving HepB and hepatitis B immunoglobulin (HBIG) shortly after birth7.
The first plasma-derived HBV vaccines were manufactured under the name Heptavax B (Merck) and Hevac B (Institut Pasteur) in 1982. Four years later, the first genetically engineered hepatitis B vaccine was developed with recombinant HBsAg. The yeast-derived recombinant vaccines are manufactured by expression of HBsAg protein in genetically engineered yeast cells that contain the S gene8. Because lacking glycosylation in yeast, mammalian cell-derived vaccines are produced which contained glycosylated pre-S1 and pre-S2 proteins, in addition to the major HBsAg protein9. Currently, hepatitis B vaccines are formulated to contain 5–60 μg of recombinant HBsAg protein and 10–20 μg vaccines was often used for newborns with HBsAg-positive mother.
In China, a national survey in 2006 reported that the prevalence of HBsAg among women of childbearing age was 7.6%10, while a recent study on 15 million couples in rural China revealed a 5.2% HBsAg seropositive rate among women aged 15–4911. In certain areas of southeast China, the HBsAg-positive rate among women of childbearing age even exceeded 10%12. In 2012, the Chinese government began to implement the HIV-syphilis-HBV prevention program. All newborns born to HBsAg-positive mothers were eligible to receive three doses of HepB and one dose of HBIG (100 IU).
There are two types of HepB available for newborns in China, each with a different HBsAg content: 10 μg and 20 μg. The 10 μg vaccine is produced using yeast or China hamster oocytes (CHO) as the expression vector, while the 20 μg vaccine is produced using CHO as the expression vector. In this study, a prospective cohort study was conducted to examine the efficacy of 20 μg HepB for PMTCT.
Results
Characteristics of the pregnant women and their infants
A total of 992 HBsAg positive pregnant women who delivered in 13 hospitals located in the two counties between May 1, 2018, and October 31, 2019. Among them, 604 (60.1%, 604/992) were interviewed when upon arrival at the hospital for delivery, and a total of 619 infants were born, with 511 receiving three doses of 20 μg HepB between one and six months apart. The blood samples of 319 infants were collected one month later, after they had completed three doses of vaccine, to test for HBsAg and HBsAb. In order to estimate the HBsAb variation of infants, blood samples of 398 infants, who had been vaccinated with 20 μg CHO HepB, were collected in June 2020, which included 287 infants who had their sero-samples collected for the first time. In total, 419 HBsAg-positive women and their 430 infants were analyzed (Fig. 1). The average age of the women was 29.6 ± 4.3 years, with the oldest and youngest women being 47 and 19 years old, respectively. Those with college education or above accounted for 54.9% (230/419). The oldest child was 24 months old, while the youngest child was seven months old. Ethnic Han women accounted for 99.1% (415/419) for the total, whereas female farmers for 24.1% (101/419). Among the women, 21.5% (90/419)were aware that they were infected with HBV in the previous year. Furthermore, 27.4% (115/419) of them had received antiviral therapy during pregnancy (Table 1). Twenty-five of the pregnant women gave birth before 37 weeks, and 14 out of the 270 pregnant women who filled out ALT values had ALT values higher than 40.
Blood sampling and testing of pregnant women
In order to detect HBV DNA, HBsAg, HBeAg, and HBeAb, 5 ml blood samples were collected from 419 pregnant women. The results revealed that all the pregnant women were HBsAg positive. Among them, 339 (80.9%, 339/419) had serum HBV DNA lower than 2 × 105 IU/mL while the remaining 80 (19.1%, 80/419) had HBV DNA higher than 2 × 105 IU/mL. HBV DNA levels tend to decrease with increasing age. The HBV DNA levels of 31% (13/42) of pregnant women aged < 25 years and 11.3% (14/124) of pregnant women aged ≥ 30 were higher than 2 × 105 IU/mL. Although the HBV DNA level in pregnant women aged ≥ 35 years was higher than that of those aged 30–35 years, the difference was not statistically significant (P = 0.649).
Civil servants had the lowest HBV DNA levels, but there were no significant differences between them and the other occupational groups. HBV DNA levels were highest in pregnant women with high school education, but the differences between different groups were not statistically significant. There were no significant differences in the distribution of HBV DNA between different ethnic groups, regardless of whether they received antiviral treatment during pregnancy.
The overall positive rate of HBeAg was 29.1% (122/419), with the positive rate decreasing with increasing age. The positive rate of HBeAg in pregnant women of the ≥ 35-year-old group was 18.3% (11/60), while the positive rate of HBeAg in the < 25-year-old group was 33.3% (14/42), with a significant difference between both groups (P = 0.043). The positive ratio of HBeAg in the 25–30-year-old group was 34.7% (67/193), with a significant difference (P = 0.016) compared to the ≥ 35-year-old group. Pregnant women with junior high school education had the highest positive ratio of HBeAg (33.7%, 32/95), but the difference was not significant compared to the other groups. Furthermore, the highest positive rate of HBeAg was found in medical staff (33.3%, 4/12), but the difference was not significant compared to the other groups. Among the 122 HBeAg-positive women, 42 received antiviral therapy during pregnancy. The HBeAg positive rate of the group receiving antiviral treatment during pregnancy was 36.8%, which was significantly higher (P = 0.034) than the untreated group (26.2%).
The overall positive rate of HBeAb was 61.3% (257/419), with a gradual increase in positive rate with increasing age. The < 25-year-old group had the lowest positive rate (52.4%, 22/42), but there were no statistically significant differences between them and the other age groups. The positive rate of HBeAb was the lowest in the group with junior high school education (56.8%, 54/95), but the difference was not significant compared to the other education groups. Farmers had the lowest positive rate of HBeAb (56.3%, 54/96), while civil servants had the highest (79.4%,27/34), and the difference between them was significant (P = 0.017). The positive rate of HBeAb in the antiviral treated group was significantly lower (P = 0.044) than the non-treated group (53.5% vs. 64.3%).
Newborn vaccination, blood sample collection, and testing
All the newborns received their first dose of 20 μg hepatitis B vaccine within 12 h of birth. The second dose was administered to 284 infants within 28–35 days, and 146 infants after 35 days. For the third dose, 369 infants were immunized within 180–210 days, 54 infants were immunized over 210 days, and 3 infants were immunized 90 days after birth, while there was no information on 2 infants. One month after the third dose was administered, blood samples were collected from a total of 319 infants. Six infants were HBsAg positive (1.9%, 6/319), while the others were HBsAg-negative (98.1%). All six HBsAg positive infants born to HBeAg postive mother (6.2%,6/97). Antiviral therapy was given to 41 HBeAg-positive pregnant women during their pregnancy, with one case of MTCT (2.4%, 1/41). Among the 80 HBeAg-positive pregnant women who did not receive antiviral therapy, five women failed to PMTCT (6.3%, 5/80) and there was no significant difference between the two groups (χ2 = 0.882, P = 0.348). Out of the 313 HBsAg negative infants, 300 (95.8%, 300/313) were HBsAb positive (HBsAb ≥ 10 mIU/mL) and the geometric mean concentration (GMC) of their antibody was 861.6 mIU/mL. GMC levels higher than 100 mIU/ml were detected in 260 infants (83.1%, 260/313). The highest concentration of HBsAb was 73,150 mIU/mL.
Changes in antibody attenuation among neonates
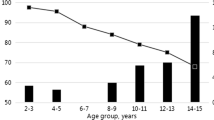
A cross-sectional survey was conducted on all children who could be contacted and had received three doses of the CHO vaccine. A total of 398 blood samples were collected to detect HBsAg and HBsAb, with all children testing negative for HBsAg, while there was a decrease in HBsAb concentration among 196 children (68.3%, 196/287). The GMC of HBsAb of 398 children decreased gradually over time after completing the vaccination program. At 1–3 months after full inoculation, the GMC was 1025 mIU/ml and the HBsAb positive rate was 100%. In the population older than 16 months, the GMC decreased to 122 mIU/ml and the HBsAb positive rate decreased to 89.6% (Fig. 2). Statistical analysis revealed that there was no significant difference in HBsAb positive percent between the space-time groups (χ2 = 6.095, P = 0.297).
The findings showed that there was no significant link between HBsAb concentration gradient in infants and the HBV infection status or HBV DNA load in pregnant women (Table2).
Characteristics of HBsAg positive children
The mothers of six HBsAg positive children were all strongly positive for HBeAg, with five of them having HBV DNA loads higher than 5 × 108 IU/mL. The HBV DNA load in one mother who received antiviral therapy during pregnancy was 2.13 × 105 IU/ml. All six children received 20 μg CHO hepatitis B vaccine and HBIG according to schedule after birth, as well as completing all three doses of the hepatitis B vaccine (Table 3). However, two children received their second dose later, at 57 and 59 days after birth, respectively. None of the six children were underweight.
Reimmunization of children with low antibody levels
If the HBsAb concentration was lower than 10 mIU/ml when the sero-sample was collected the first time, three additional doses of HepB were administered. At the time of the first sero-sample, a total of 13 children were HBsAg negative and had HBsAb levels lower than 10 mIU/ml. During the second blood collection after the booster immunization, a total of ten sero-samples were collected. There were elevated HBsAb levels in 6 children (60%, 6/10), with a maximum of 681 mIU/ml and a minimum of 60.9 mIU/ml, while four children had negative HBsAb and HBsAg (40%, 4/10) (Table 4).
Discussion
CHB is a major public health problem that affects around 257 million people worldwide, including 65 million women of reproductive age, and causes 800,000 deaths yearly. Adults infected with HBV often have an acute infection, that leads to a self-limited disease; only 2 to 3% of them will develop CHB. However, infants with acute infection have a 95% risk of developing CHB. As a result, vertical mother-to-child transmission from HBsAg-positive mothers accounts for approximately 50% of the global disease burden13. There are two main ways of MTCT, one is intrauterine transmission that occurs during pregnancy and the other is blood transmission that occurs during and after delivery. WHO recommends that HBsAg-positive pregnant women with higher HBV DNA or positive HBeAg should receive antiviral treatment in order to reduce intrauterine transmission. WHO also recommends that all infants should receive a birth dose of the hepatitis B vaccine immediately after birth, followed by two further doses to complete the primary series of vaccinations.
Since the 1990s, HBV vaccination in children has been widely implemented in more than 200 countries, leading to significant reductions in MTCT and global CHB incidence. Previous reports have shown that HBV transmission in newborns of HBeAg-negative mothers can be almost completely inhibited by the timely completion of the whole HBV vaccination course14, however, about 10% newborns born to HBsAg and HBeAg double positive mothers may be infected with HBV through MTCT. In previous studies, HBsAg antigen expressed by CHO cells was found to be more stable15, and the immune effect was better than that of yeast after use in adults16. Furthermore, vaccines prepared with HBsAg containing PreS1 expressed in CHO have a good immune response in immunodeficient persons17. Therefore, it may also be interesting to understand the effects of HepB with high concentration derived from CHO cells on MTCT prevention. The results of this study showed that administration of three doses of 20 μg HepB (plus HBIG) achieved a totally prevention success rate of 98.1% and the HBeAg-negtive mother achieved 100% prevention and HBeAg-positive mother achieved 93.8% for prevention success rate. Previous retrospective study had showed that 5 μg HepB (plus HBIG) achieved 94.4% and the 10 μg achieved 96.5% on blocking MTCT18,19. 20 μg HepB (plus HBIG) derived from CHO cell seems to have a greater advantage on preventing MTCT. WHO had set the goal of eliminating hepatitis on the whole world by 2030 in 2021, including MTCT blocking rate reached to 98% by the way of vaccination20. So finding more effective vaccine was also a daunting task.
The occurrence of HBV MTCT is closely related to the presence of HBV infection in the mother. Several studies reported that pregnant women who were HBeAg positive or had an HBV DNA level higher than 2 × 105 IU/ml had a significantly increased probability of MTCT4,21. According to the 2014 HBV sero-survey of China, the average HBsAg positive rate among Chinese women under the age of 29 was 2.6%22. Due to the differences in economy and lifestyle, women of childbearing age living in different provinces of China often have varying HBsAg-positive rates. In certain provinces in southeastern China, the HBsAg positive rate among pregnant women aged 15–49 years is as high as 11.2%12. In 2012, a 3.7% HBsAg positive rate among women of childbearing age in the central Chinese province of Henan was reported23.
In this study, the positive rate of HBeAg in HBsAg-positive pregnant women was 29.1% in China, whereas the positive rate ranged from 11.2 to 35.1% in other previously published studies24,25,26. In some European countries, the rate was considerably higher than 50%27,28. Many studies have reported that the positive rate of HBeAg in HBsAg-positive women significantly decreases with increasing age. Our findings were consistent with previous studies, as we observed a gradual decrease in the positive rate of HBeAg with the increasing age of pregnant women. In this study, the success rate of PMTCT of infants born to HBeAg-positive pregnant women was 93.8%, and no children born to HBeAg-negative pregnant women were infected. Six mothers who gave birth to children with positive HBsAg and negative HBsAb had HBV DNA levels higher than 2 × 105 IU/ml, with five of them having levels higher than 5 × 108 IU/ml. Maternal HBeAg positivity and an increased HBV DNA level were risk factors for HBV MTCT. As previously mentioned, maternal HBeAg positivity may even be an independent risk factor29. Previous study had found that more than 1% of HBV infections in newborns who had vaccinated timely were due to intrauterine infections. However, positive HBeAg and higher HBV DNA level of pregnant women were risk factors related to intrauterine infections. Therefore, more attention should also be paid to health education of pregnant women. Timely and continuously antiviral treatment before pregnancy, in order to reduce HBV DNA level, and even realize the serological conversion of HBeAg, may be more meaningful for improving the blocking rate of mother-to-child infection.
Previous studies have reported that antiviral therapy during pregnancy can effectively reduce the serum HBV DNA level, even reaching the serological conversion of HBeAg and HBeAb30,31. In this study, the HBeAg positive rate of pregnant women who received antiviral therapy was significantly higher (P = 0.034) than those who did not (36.8% VS 26.4%). This was potentially due to people who received antiviral treatment having more severe clinical symptoms, i.e., they had positive HBeAg and a higher HBV DNA level. The antiviral treatment aimed to treat CHB rather than prevent MTCT. Among the six cases that failed to prevent MTCT, one pregnant woman received antiviral therapy during her pregnancy (16.7%, 1/6), but her HBV DNA level was still higher than 2 × 105 IU/ml. There were no significant differences in MTCT between the group receiving antiviral treatment and those who did not. The treatment procedures for pregnant women to evaluate the effect of MTCT, rather than the care procedure for CHB in general, still require improvement. On the other hand, the sample size of the preventing failure of MTCT was very small in this study, which couldn’t truly reflect the therapeutic effect and may bring large sampling error. So perhaps a larger sample size is necessary to get real results.
The HBsAb titer of children was an important indicator to determine whether they possessed immunity to HBV infection. In this study, the GMC of HBsAb in children with successful HBV prevention was not associated with the maternal HBV infection status. One month after the complete immunization, the GMC was 861.6mIU/mL. The GMC of HBsAb gradually decreased with increasing age, and 17 months after the entire immunization process, the positive rate of HBsAb also decreased from 100 to 89.1%. Federica Chiara et al. had showed that the HepB administered before one year of age tends to cause a significant decrease in the positive rate and the GMT of HBsAb32. It is probably related to the differences between innate immunity and adaptive immunity. Other finding showed that if the response to the HepB is positive, the decrease of the positive rate of HBsAb even below 10 mIU/ml does not mean a loss of protection33. But for the infants with HBsAg-postitive mother, a post-vaccination serological test (PVST) was recommended before the occurrence of a rapid decline 4–6 months following complete immunization. According to WHO guidelines, infants whose mothers are HBsAg positive can receive three injections of enhanced immunization if they are negative for both HBsAg and HBsAb after the immunization program. In this study, ten children who had HBsAb lower than 10 mIU/ml after completing the vaccination program received another three doses of 20 μg HepB as a booster. Although six children had HBsAb higher than 10 mIU/ml, the other four still had HBsAb lower than 10 mIU/ml. These results suggested that timely detection of antibodies after complete immunization, as well as revaccination for children with HBsAb lower than 10 mIU/ml, were critical for the success of PMTCT.
Conclusion
20 μg HepB was effective in preventing vertical transmission of HBV. It is crucial to enhance health education for childbearing age women so that they may participate in early HBsAg screening and be aware of their HBV infection status. Although several technical guidelines have recommended antiviral therapy for HBsAg-HBeAg positive pregnant women or those with a higher HBV DNA level, a more viable treatment procedure for greater prevention of HBV MTCT is still lacking in China, even worldwide.
Material and methods
Study population
The Xinzheng county and Jiefang district of Jiaozuo in Henan province in central China were selected as the survey points based on the previous prevalence rate of HBsAg-positive women of childbearing age. All HBsAg-positive pregnant women who gave birth between May 1, 2018, and Oct 31, 2019, in the two areas were interviewed. Their infants (those that survived) were vaccinated with three doses of 20 μg HepB at intervals of 0, 1, and 6 months, as well as one dose of 100 IU HBIG, all of which were provided for free.
The HBV infection status and information of HBsAg-positive women
Pregnant women were screened for HBsAg using the ELISA method when they visited the hospital for perinatal health care in China. All the pregnant women who participated in the survey were interviewed and given a questionnaire to fill out. Personal information about the pregnant women, such as age, gestational age, history of therapy, and other factors, were included in the questionnaire. Before delivery, 5 ml of blood sample was taken from the expecting mothers. All serum samples were delivered to the Henan provincial Center for Disease Control and Prevention to test for HBsAg, HBeAg, and HBeAb using ELISA test kits (Beijing Wantai Biological Pharmacy Enterprise Co., Ltd.). The amount of HBV DNA in the collected serum was measured by real-time PCR using the HBV DNA test kit (DAAN Gene Co., Ltd. China).
The vaccination and serology test of infants
The HepB used in this study contained 20 μg HBsAg in each dose (Jintan Biotechnology Co., Ltd, China). All newborns received the birth dose vaccine and 100 IU HBIG within 12 h after birth, followed by the second dose vaccine within 28–35 days after birth, and the third dose vaccine between 180 and 210 days after birth. One month after completing the basic immunization program, 3 ml blood samples (the first blood samples) were collected to test for HBsAb and HBsAg. In June 2020, 3 ml blood samples (the second blood samples) were collected from all enrolled HBsAg negative infants to detect HBsAb and HBsAg.
Statistical analysis
All data were double entered into an EPI Data 3.02 software database and cross-checked against the original records. After accuracy verification, data were analyzed at HNCDC using the SPSS 19.0 software for Windows (SPSS Inc., Chicago. IL). The sociodemographic characteristics of the participants and their infection status in terms of HBsAg positivity were described using proportions and ratios. The chi-square test or Fisher exact probability test was used to determine the statistical significance of the associations, with a significance level of P < 0.05. The chi-square analysis was used to analyze the differences.
Ethical approval statement
The survey was approved by the Henan provincial Center for Disease Control and Prevention Ethics Committee. Each participant was informed of the survey’s purpose, as well as their right to have their information kept confidential. The informed consent was obtained as following: the pregnant women's informed consent were signed and confirmed by themself and the informed consent of the newborns were signed by their parents (or other legal guardians entrusted by their parents). In addition, all interviews, laboratory tests, and notification of results were provided free of charge to the participants. The test results were enveloped, sealed, and personally delivered to each participant by the local doctor. This study was performed in accordance with relevant guidelines and regulations.
Data availability
All data generated or analysed during this study are included in this published article.
References
MacLachlan, J. H. & Cowie, B. C. Hepatitis B virus epidemiology. Cold Spring Harb. Perspect. Med. 5, 1–12. https://doi.org/10.1101/cshperspect.a021410 (2015).
WHO. Global hepatitis report, 2017, <https://www.who.int/publications/i/item/global-hepatitis-report-2017> (2017).
Wiseman, E. et al. Perinatal transmission of hepatitis B virus: an Australian experience. Med. J. Aust. 190, 489–492 (2009).
Gentile, I. & Borgia, G. Vertical transmission of hepatitis B virus: challenges and solutions. Int. J. Women’s Health 6, 605–611. https://doi.org/10.2147/IJWH.S51138 (2014).
Gentile, I., Zappulo, E., Buonomo, A. R. & Borgia, G. Prevention of mother-to-child transmission of hepatitis B virus and hepatitis C virus. Expert Rev. Anti Infect. Ther. 12, 775–782. https://doi.org/10.1586/14787210.2014.920254 (2014).
Liu, C. P. et al. Factors associated with mother-to-child transmission of hepatitis B virus despite immunoprophylaxis. Intern. Med. 54, 711–716. https://doi.org/10.2169/internalmedicine.54.3514 (2015).
WHO. Guidelines for the prevention, care and treatment of persons with chronic hepatitis B infection, <https://www.who.int/publications/i/item/9789241549059> (2015).
Pattyn, J., Hendrickx, G., Vorsters, A. & Van Damme, P. Hepatitis B vaccines. J. Infect. Dis. 224, S343–S351. https://doi.org/10.1093/infdis/jiaa668 (2021).
Kim, H. S. et al. Phylogenetic analyses of HBV pre-s/s genes in mother-child pairs with long-term infection by presumed vertical transmission. J. Korean Med. Sci. 29, 564–569. https://doi.org/10.3346/jkms.2014.29.4.564 (2014).
Liang, X. et al. Evaluation of the impact of hepatitis B vaccination among children born during 1992–2005 in China. J. Infect. Dis. 200, 39–47. https://doi.org/10.1086/599332 (2009).
Wang, Y. et al. Prevalence of chronic hepatitis B and status of HBV care among rural women who planned to conceive in China. Sci. Rep. 7, 12090. https://doi.org/10.1038/s41598-017-12005-2 (2017).
Zheng, H. et al. The epidemiology of hepatitis B virus infection in women of reproductive age in highly endemic areas in China. J. Viral Hepat. 25, 88–96. https://doi.org/10.1111/jvh.12757 (2018).
Franco, E. et al. Hepatitis B: Epidemiology and prevention in developing countries. World J. Hepatol. 4, 74–80. https://doi.org/10.4254/wjh.v4.i3.74 (2012).
Lu, Y. et al. Hepatitis B vaccine alone may be enough for preventing hepatitis B virus transmission in neonates of HBsAg (+)/HBeAg (-) mothers. Vaccine 35, 40–45. https://doi.org/10.1016/j.vaccine.2016.11.061 (2017).
Diminsky, D., Moav, N., Gorecki, M. & Barenholz, Y. Physical, chemical and immunological stability of CHO-derived hepatitis B surface antigen (HBsAg) particles. Vaccine 18, 3–17. https://doi.org/10.1016/s0264-410x(99)00149-8 (1999).
Zhang, W. et al. Surface antibody and cytokine response to recombinant Chinese hamster ovary cell (CHO) hepatitis B vaccine. Vaccine 29, 6276–6282. https://doi.org/10.1016/j.vaccine.2011.06.045 (2011).
Shouval, D., Roggendorf, H. & Roggendorf, M. Enhanced immune response to hepatitis B vaccination through immunization with a Pre-S1/Pre-S2/S vaccine. Med. Microbiol. Immunol. 204, 57–68. https://doi.org/10.1007/s00430-014-0374-x (2015).
Wang, F. et al. Effectiveness of prevention of mother-to-child transmission practice in three provinces of Southern China. Hum. Vaccin. Immunother. 11, 2061–2067. https://doi.org/10.1080/21645515.2015.1023972 (2015).
Wang, F. et al. Post-vaccination serologic testing of infants born to hepatitis B surface antigen positive mothers in 4 provinces of China. Vaccine 35, 4229–4235. https://doi.org/10.1016/j.vaccine.2017.06.019 (2017).
Organization, W. H. Global guidance on criteria and processes for validation: elimination of mother-to child transmission of HIV, syphilis and hepatitis B virus. ( World Health Organization, 2021). Licence: CC BY-NC-SA 3.0 IGO.
Kang, W. et al. Risk factors associated with immunoprophylaxis failure against mother to child transmission of hepatitis B virus and hepatitis B vaccination status in Yunnan province, China. Vaccine 32, 3362–3366. https://doi.org/10.1016/j.vaccine.2014.04.045 (2014).
Cui, F. et al. Prevention of chronic hepatitis B after 3 decades of escalating vaccination policy, China. Emerg. Infect. Dis. 23, 765–772. https://doi.org/10.3201/eid2305.161477 (2017).
Yong Hao, G. et al. The prevalence of hepatitis B infection in central China: An adult population-based serological survey of a large sample size. J. Med. Virol. 89, 450–457. https://doi.org/10.1002/jmv.24649 (2017).
Ding, Y., Sheng, Q., Ma, L. & Dou, X. Chronic HBV infection among pregnant women and their infants in Shenyang, China. Virol. J. 10, 17. https://doi.org/10.1186/1743-422X-10-17 (2013).
Kang, W. et al. Risk factors related to the failure of prevention of hepatitis B virus mother-to-child transmission in Yunnan, China. Vaccine 35, 605–609. https://doi.org/10.1016/j.vaccine.2016.12.016 (2017).
Zheng, H., Cui, F. & Gong, X. Staus of the hepatitis B virus surface antigen and e antigen prevalence among reproductive women in China. Chin. J. Vaccin. Immun. 16, 496–499 (2010).
Beasley, R. P., Trepo, C., Stevens, C. E. & Szmuness, W. The e antigen and vertical transmission of hepatitis B surface antigen. Am. J. Epidemiol. 105, 94–98. https://doi.org/10.1093/oxfordjournals.aje.a112370 (1977).
Chamroonkul, N. & Piratvisuth, T. Hepatitis B during pregnancy in endemic areas: screening, treatment, and prevention of mother-to-child transmission. Paediatr. Drugs 19, 173–181. https://doi.org/10.1007/s40272-017-0229-1 (2017).
Yonghao, G. et al. A retrospective study of hepatitis B mother-to-child transmission prevention and postvaccination serological test results of infants at risk of perinatal transmission in two counties of middle China. J. Viral Hepat. 24, 687–695. https://doi.org/10.1111/jvh.12694 (2017).
Bruni, R., Taliani, G. & Ciccaglione, A. R. Antiviral treatment of HBV positive pregnant women: An additional tool to reduce perinatal transmission. Pathog. Glob. Health 110, 275–276. https://doi.org/10.1080/20477724.2016.1255375 (2016).
Fan, L., Owusu-Edusei, K. Jr., Schillie, S. F. & Murphy, T. V. Cost-effectiveness of active-passive prophylaxis and antiviral prophylaxis during pregnancy to prevent perinatal hepatitis B virus infection. Hepatology 63, 1471–1480. https://doi.org/10.1002/hep.28310 (2016).
Chiara, F. et al. Hepatitis B vaccination at three months of age: A successful strategy?. Vaccine 31, 1696–1700. https://doi.org/10.1016/j.vaccine.2013.01.046 (2013).
Chiara, F. et al. Hepatitis B vaccination of adolescents: Significance of non-protective antibodies. Vaccine 32, 62–68. https://doi.org/10.1016/j.vaccine.2013.10.074 (2013).
Funding
Natural Science Foundation of Henan Province (222300420466). Research Program of Medical Science and Technology of Henan Province (2018010028). Hepatitis B Prevention and Control Research project of China foundation for hepatitis prevention and control (YGFK20170023).
Author information
Authors and Affiliations
Contributions
We confrm that there is no overlap with previous publications, and we confrm that the manuscript, including related data, figures, and tables, has not been published previously and that the manuscript is not under consideration elsewhere. We also confrm that all authors have made substantial contributions to all of the following: (1) the conception and design of the study, or acquisition of data, or analysis and interpretation of data, (2) drafing the article or revising it critically for important intellectual content, (3) final approval of the version to be submitted.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yonghao, G., Yanping, C., Qiaohua, D. et al. The effectiveness of 20 μg hepatitis B vaccine used for the prevention of HBV vertical transmission. Sci Rep 12, 11759 (2022). https://doi.org/10.1038/s41598-022-15744-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-15744-z
- Springer Nature Limited