Abstract
Insecticide-based vector control is key to the reduction and elimination of malaria. Although insecticide resistance is common in malaria vector populations, the operational implications are often unclear. High intensity pyrethroid resistance in the major malaria vector Anopheles funestus has been linked to control failure in Southern Africa. The aim of this study was to assess linkages between mosquito age, blood feeding and the intensity of pyrethroid resistance in two An. funestus laboratory strains that originate from southern Mozambique, namely the moderately pyrethroid resistant FUMOZ and the highly resistant FUMOZ-R. Resistance tended to decline with age. This effect was significantly mitigated by blood feeding and was most apparent in cohorts that received multiple blood meals. In the absence of insecticide exposure, blood feeding tended to increase longevity of An. funestus females and, following insecticide exposure, enhanced their levels of deltamethrin resistance, even in older age groups. These effects were more marked in FUMOZ-R compared to FUMOZ. In terms of programmatic decision-making, these data suggest that it would be useful to assess the level and intensity of resistance in older female cohorts wherever possible, notwithstanding the standard protocols for resistance testing using age-standardised samples.
Similar content being viewed by others
Introduction
Vector control is key to the reduction and elimination of malaria. A limited number of Anopheles species transmit malaria, and their control is primarily insecticide-based, relying on the deployment of insecticides via indoor residual spraying (IRS), insecticide treated nets (ITNs) and larviciding as a component of larval source management1,2,3. Until very recently, the number of insecticides available for use in malaria control was contained within only four classes—the pyrethroids, organochlorines (DDT only), carbamates and organophosphates. These collectively target only two neuronal sites within insects i.e., the voltage-gated sodium ion channel and acetylcholinesterase. Additional compounds recently developed for use in malaria control are the neonicotinoid clothianidin and the oxidative phosphorylation uncoupler chlorfenapyr4,5,6,7. These, amongst others still in the development phase, were formulated for vector control because of the burgeoning incidence of insecticide resistance in Anopheles malaria vector populations.
Insecticide resistance in vector populations is almost ubiquitous across malaria-affected regions8, and resistance management is therefore a necessary policy in many vector control programmes9, 10. Although there are several reports of resistance undermining control efficacy11,12,13,14, the operational implications of resistance are often unclear15. This is because surveillance for insecticide resistance in vector populations is typically assessed first by measuring phenotypes using standardised response-to-exposure assays16. These assays are based on the use of diagnostic insecticide concentrations that in no way resemble those used on ITNs and to spray walls during IRS operations. To address this issue, the current guidelines include the baseline diagnostic dose test followed by a stepwise assessment of resistance intensity if detected. The diagnostic dose is usually calculated as twice the lowest concentration that induces at least 99% mortality in a normally insecticide susceptible population following exposure for a set period—in most instances one hour. Resistance is thus diagnosed if mortality < 98% 24 h after the one hour exposure, and intensity can subsequently be measured in the same manner using dosages at 5X and 10X the diagnostic concentration16. These data can be used to make assessments about the potential operational impact of resistance, especially where IRS is used for control17, 18.
The standardised assays are generally effective at diagnosing the occurrence (diagnostic dose) and potential threat of resistance (intensity assessments) and are recommended for use on age-controlled samples that adequately represent the vector population from which the test mosquitoes are drawn. In order to make resistance data comparable across regions and even species, it is currently recommended that adult female mosquitoes aged less than 5 days be used for insecticide bioassays, and that these females should not have acquired any blood meals. It is however reasonable to expect that age19 and blood-feeding status20, 21 can measurably affect resistance phenotypes, as has previously been demonstrated22, and it is also important to note that it is only older, blood-fed females that transmit malaria23.
High-intensity insecticide resistant phenotypes are generally produced by multiple mechanisms, at least one of which is a major-effect factor around which stabilising co-factors have been selected24. A documented example is pyrethroid resistance in southern African populations of the major African malaria vector species Anopheles funestus. This resistant phenotype is primarily mediated by monooxygenase detoxification (cytochrome P450s CYP6P9 and CYP6P13)25,26,27 that is facilitated by increased expression of glutathione-S-Transferases (GSTs) and thickened cuticles28, 29. These collectively produce a moderate-to-high intensity resistance phenotype that has previously been linked to operational control failure in South Africa and Mozambique30, 31. Available evidence further suggests that this phenotype does not incur fitness costs in affected An. funestus populations32.
Anopheles funestus is a highly anthropophilic species and may be amongst the first anophelines to adapt to human hosts33. Adult female anophelines tend to take multiple blood meals in a single gonotrophic cycle, making them especially efficient vectors of malaria34, 35 that often carry greater parasite loads than other vector species36, 37. Furthermore, Plasmodium infection increases blood-seeking behaviour and probing38, 39. In terms of understanding malaria epidemiology where An. funestus occurs, and controlling populations of this species, important characteristics to consider are blood feeding habits, adult longevity and insecticide resistance, and how these interact with each other.
Given that older, blood-fed females are the epidemiologically significant sector of a vector population, small changes in longevity can result in significant changes in malaria transmission40. Furthermore, the intensity of the pyrethroid resistance phenotype in An. funestus varies with age, and the insecticide susceptibility of An. funestus, like other mosquitoes, has been demonstrated to increase with age41, while blood feeding tends to reduce insecticide toxicity in exposed adult mosquitoes22, 42. A single blood feed42, 43 as well as multiple blood meals reduce age-related increases in insecticide susceptibility44 by reducing the amount of oxidative stress induced by insecticide exposure45.
Given these parameters, the aim of this study was to assess linkages between age, blood feeding and the intensity of pyrethroid resistance expression in An. funestus, especially in older cohorts that have passed through successive gonotrophic cycles.
Materials and methods
Mosquito strains
Two laboratory-reared strains of An. funestus were used in this study: FUMOZ, a strain colonised from Southern Mozambique in 2000, and FUMOZ-R, which was selected from FUMOZ for resistance to pyrethroids. These strains are housed in the Botha de Meillon insectary at 25 °C (±2 °C) and 80% (± 5%) humidity, with a 12:12 h day/night photoperiod including dawn and dusk transitions. Larvae were maintained on a diet of powdered Beano™ dog biscuits and yeast (3:1) as previously described46. FUMOZ has moderate pyrethroid resistance intensity, and FUMOZ-R high resistance intensity18. Resistance in these strains is primarily attributed to overexpression of CYP6P9, CYP6P13 and GSTe225, 28, and thickened cuticles29 as previously described.
The effect of blood feeding on subsequent susceptibility to insecticide exposure
Samples of FUMOZ and FUMOZ-R adult females drawn for insecticide bioassays were divided into cohorts representing 3 different nutritional regimens. The first cohort (control group) was maintained on 10% sucrose ad libitum for the duration of their lives, with no blood meals. The second cohort was offered a single blood meal either at the age of 3, 7, 11, 15, 18 or 21 days post emergence, also with ad libitum access to sucrose. The third cohort was offered blood meals at the ages of 3, 7, 11, 15, 18 and 21 days post emergence i.e. six consecutive blood meals, also with ad libitum access to sucrose. At each age point, a sub-sample of adult females from each cohort was removed for insecticide resistance bioassays. Throughout their lives, males and females in all cohorts were caged together and allowed to mate. Females in all cohorts were allowed to oviposit twice weekly for the duration of the experiment.
Those female samples scheduled for blood feeding were fed to repletion on a single human volunteer (S.V. Oliver). The females were allowed to feed only on this volunteer and no other individuals were involved in the feeding procedure. Ethical approval for the use of invertebrate organisms was waived by the University of the Witwatersrand, Faculty of Health Sciences ethics committee: (Waiver number: S Oliver 03-01-2018). This waiver specifies that S.V. Oliver is authorised to blood feed mosquitoes as an essential part of the mosquito husbandry required for the project. Informed consent was provided by S.V. Oliver.
Four hours after feeding, cohorts of blood-fed female mosquitoes were exposed to either 0.05% (1X), 0.25% (5X) or 0.5% (10x) deltamethrin16. Only fully fed females were used for the exposures. This time gap was chosen as a previous study showed that the greatest transcriptional variation, both in gene up- and down-regulation was observed 3 h post blood meal47 As such, four hours was chosen to allow time for transcript changes to take effect. It also allowed time for the fully fed mosquitoes to be collected, and the feeding status of the females to be fully ascertained. This four hour period has previously been demonstrated to be enough time to measure phenotypic changes in insecticide tolerance in An. arabiensis44 and An. funestus42. It should be noted that blood-induced increases in insecticide tolerance peak 24 h after a blood meal21, but this timeline was not experimentally feasible with the multiple blood feeding set-up. The mosquitoes were allowed to feed on the arm of the volunteer. The arm was presented on the top of the mesh cage and the mosquitoes were allowed to feed to repletion. The volunteer did not use any scented soap or fragrances on the skin before feeding. The volunteer did not consume caffeine for four hours prior to the blood feeding. Alcohol and nicotine were not variables as the volunteer consumes neither.
The exposures were performed using the standard WHO bioassay method with treated papers of the required concentrations purchased from the WHO supplier at Universiti Sains Malaysia16. Exposed mosquitoes were allowed ad libitum access to 10% sucrose, and mortality was scored 24 h post exposure. Controls included a sample of unexposed mosquitoes and a sample exposed to solvent-only treated paper. Data from assays were not used for analysis if control mortality exceeded 10%. Exposed mosquitoes were not returned to the experimental pool. For each of the exposures, 20–25 females were used per tube. Numbers per treatment are summarised in Supplementary Table 1. The experiments were conducted over a period of three months during which the samples were collected as three separate cohorts originating from three separate egg batches. Within each cohort, adult females were split into three groups i.e. unfed, single fed and multiple fed groups as described earlier. This was to ensure that any potential environmental variations would affect all treatment groups equally.
The effect of blood feeding and insecticide exposure on subsequent adult female longevity
One hundred and fifty adult females from each of FUMOZ and FUMOZ-R were either provided with a blood meal at the age of 3 days or remained unfed. Each of these cohorts was then split into three groups: unexposed, 1X exposed (exposed to 0.05% deltamethrin using standard WHO bioassays) or 10X exposed (exposed to 0.5% deltamethrin using standard WHO bioassays). Thirty surviving females were removed from each group 24 h post exposure and placed in a cage. They were offered ad libitum access to 10% sucrose, but no further blood meals. Their respective longevities were monitored daily, with cadavers removed every day until all specimens were dead.
Statistical analysis
To determine the relative impacts of age and multiple blood meals on insecticide resistance intensity and mortality, data from FUMOZ and FUMOZ-R treatments were compared using a generalised linear model, with a quasibinomial distribution in R (v. 3.1.2). A quasibinomial distribution was chosen to account for overdispersion48. Mortality was used as the response variable, while age, bloodmeal and insecticide intensity (strain), and appropriate interactions, were used as predictor variables. The model we fit was as follows: model1 = glm(y ~ Group + Age + Treatment + Age*Treatment*Group,family = quasibinomial) where y is a bound variable alive/dead. This test was performed separately for the cohorts exposed to the standard (1X), medium (5X) and high (10X) deltamethrin dosages to determine if any predisposed advantage as a result of blood-feeding on age was carried over at higher-dose exposures to insecticides, or if this advantage was lost. For each test, the most parsimonious model was chosen as the model that best represented our data as per Crawley (2007)48.
A Kaplan–Meier estimator was used to assess longevity, with a Log-rank test used as a measure of significance at 95% confidence.
Ethical approval
This study was performed as per the ethics waiver from the University of the Witwatersrand to S Oliver: 03-01-2018.
Results
The effect of blood feeding on subsequent susceptibility to insecticide exposure
Bioassays at the standard (1X) 0.05% deltamethrin concentration
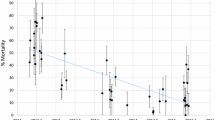
Survival was significantly decreased with increasing age in both strains. Multiple blood meals resulted in longer survival times for both strains, and there was no significant difference in survival between FUMOZ-R and FUMOZ. Females that were not fed blood experienced higher mortality than those fed blood. There was a significant two-way interaction between strain/colony and age (Group (FUMOZ)*Age), indicating that the effect of increased mortality with increased age differed between the two colonies (Table 1, Fig. 1).
Survival of Anopheles funestus FUMOZ and FUMOZ-R laboratory-reared adult females after insecticide exposure bioassays by age and blood feeding status. Average survival (%) is given for FUMOZ (white) and FUMOZ-R (grey), grouped according to age and number of blood meals following exposure to (a) 1X Deltamethrin; (b) 5X Deltamethrin; and (c) 10X Deltamethrin. Treatment groups are as follows: ‘NB’ – no blood; ‘1B’ – one blood meal; ‘MB’ – multiple blood meals; while ‘3d’, ‘7d’, ‘11d’, ‘15d’, ‘18d’ and ‘21d’ refer to 3-day old, 7-day old, 11-day, 15-day, 18-day, and 21-day old treatment groups.
Bioassays at the 5X 0.25% deltamethrin concentration
There was a significant difference in survival between the strains, with FUMOZ showing significantly higher mortality than FUMOZ-R. Increasing age resulted in a significant decrease in survival. There was no impact of blood meals on survival, and this was true whether the females had had multiple blood meals or no blood meals at all. There was, however, a significant interaction between age and blood meal, suggesting that multiple blood meals significantly improved survival of older females at this concentration (Table 1, Fig. 1). In most instances, the mortality of individuals within the resistant strain (FUMOZ-R) was lower as age increased, compared to mortality for the less resistant FUMOZ strain.
Bioassays at the 10X 0.5% deltamethrin concentration
There was no significant difference in survival between strains in general. There was, however, a significant difference by age, with older females experiencing increased mortality, when compared to their younger counterparts. The consumption of multiple bloodmeals also significantly impacted survival, with survival increasing with increased consumption of blood. There was no difference in mortality in the environmental control (unexposed) and solvent-only treated paper as there was no mortality in these treatments during the course of the 24-h period post-exposure.
There were significant two-way interactions between age and group/colony, indicating that older FUMOZ-R females experienced different mortality at this concentration when compared to older FUMOZ females i.e. older FUMOZ females died more readily than older FUMOZ-R females. The other significant two-way interaction was between colony and blood meal frequency, indicating that the influence of multiple blood meals on survival was expressed differently between colonies i.e. multiple blood fed FUMOZ-R females experienced higher survival rates by age cohort than FUMOZ females.
There were also two significant three-way interactions between Group*Age*Blood meal (multi or no blood). Although difficult to disentangle, data show that older resistant FUMOZ-R females, fed multiple blood meals, or even no blood meals, experienced reduced mortality at the 10X concentration when compared to FUMOZ females (Table 1, Fig. 1).
The effect of blood feeding and insecticide exposure on subsequent adult female longevity
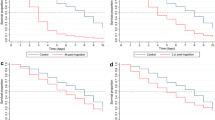
Longevity was significantly greater in unexposed FUMOZ compared to FUMOZ-R, regardless of whether they were blood-fed (Log rank test: p = 0.02, χ2 = 5.32, DF = 1) or unfed (Log rank test: p = 0.04, χ2 = 4.34, DF = 1). Exposure to the standard concentration of deltamethrin (0.05%) tended to nullify this effect in both the blood-fed and unfed cohorts (Log rank test: unfed-p = 0.08, χ2 = 3.01, DF = 1; fed-p = 0.23, χ2 = 1.41, DF = 1), as did exposure to the 10X (0.5%) deltamethrin concentration (Log rank test: unfed-p = 0.21, χ2 = 1.54, DF = 1; fed-p = 0.49, χ2 = 0.46, DF = 1) (Fig. 2).
Longevity of Anopheles funestus FUMOZ and FUMOZ-R laboratory-reared adult females by deltamethrin exposure and blood feeding status. (A) Longevity in FUMOZ and FUMOZ-R unfed, unexposed adults. (B) Longevity in FUMOZ and FUMOZ-R after a single deltamethrin exposure in adults fed a single blood meal at age 3 days and exposed to a concentration of 0.05% (1X). (C) Longevity in FUMOZ and FUMOZ-R after a single deltamethrin exposure in adults fed a single blood meal at age 3 days and exposed to a concentration of 0.5% (10X).
When examining the mean survival time to 50% mortality (St(50)), unfed and unexposed FUMOZ showed an St(50) 3 days later than FUMOZ-R. When the unfed adults were exposed to 1X deltamethrin, both strains showed an St(50) of 27 days. By contrast, when exposed to 10X deltamethrin, FUMOZ-R showed an St(50) 11 days later than FUMOZ.
When fed, unexposed FUMOZ showed an St(50) 9 days later than FUMOZ-R. When exposed to 1X deltamethrin the St(50) of FUMOZ-R was 3 days later than that of FUMOZ. The effect was even more marked when exposed to 10X deltamethrin, where FUMOZ-R showed an St(50) 9 days later than FUMOZ (Table 2).
Discussion
There is growing evidence of a direct association between insecticide resistance intensity and operational control effectiveness11, 49, 50. Although it is suggested that delayed mortality leads to sustained efficacy of long lasting insecticide treated nets (LLINs) in the presence of pyrethroid resistance15, it has recently been reported that populations in Burkina Faso show only minimal delayed mortality51. To fully understand the impact of resistance intensity on vector control operations, it is important to understand its’ effect on the survival of older, blood-fed females.
Insecticide resistance in the An. funestus strains used in this study is primarily mediated by metabolic detoxification28, 41, 52 although they differ in terms of resistance intensity18. In general, resistance tended to decline with age in both strains. This effect was significantly mitigated by blood feeding and was most apparent in FUMOZ-R. Mitigation of the loss of resistance with age was also most apparent in those cohorts that received multiple blood meals. Importantly, this mitigation effect was evident in those cohorts exposed to 5X and 10X the diagnostic dose, ultimately showing that blood feeding, and especially multiple blood feeding, tended to enhance the level and intensity of resistance to deltamethrin in both strains, and enabled the survival of a small proportion of older females even at the 10X dose.
A variety of factors can affect the longevity of adult mosquitoes. These include environmental factors such as humidity and temperature, as well as predation53. The larval environment and subsequent size of the adult also plays a role in longevity54. It has been demonstrated that insecticide resistant An. gambiae and An. arabiensis are likely to experience reduced longevity44, 55. Following this trend, the less resistant FUMOZ strain is evidently longer-lived than FUMOZ-R in the absence of exposure to insecticide. This difference however disappeared following exposure to deltamethrin at the 1X and 10X doses. In the absence of insecticide exposure, blood feeding tended to increase longevity, but this effect was lost after exposure to the diagnostic doses of insecticide. This observation was however made after an initial single blood meal, and not multiple blood meals. A comparison between the blood-fed cohorts shows that those not exposed to insecticide had greater longevity than those exposed to the 1× and 10X doses across both strains.
Although blood feeding did not necessarily increase longevity, especially following exposure to insecticide, it also did not cause any reductions in lifespan and, importantly, blood feeding bolstered the level and intensity of deltamethrin resistance in older female mosquitoes. This was most apparent in FUMOZ-R, suggesting a lower delayed mortality than FUMOZ. A recent study has independently demonstrated a reduction in delayed mortality in FUMOZ-R following repeated exposures to pyrethroid treated bed nets56.
It is suggested that this effect is related to oxidative stress. A previous study found that age-related changes in pyrethroid resistance were not associated with cytochrome P450 transcription, regardless of blood feeding status41. By contrast, FUMOZ-R has an increased capacity to withstand oxidative stress compared to insecticide susceptible An. funestus. This is mediated by high catalase and glutathione peroxidase activity45. In this scenario, multiple blood feeding reduces the oxidative burden in insecticide resistant mosquitoes, producing the downstream effect of reducing the oxidative burden induced by exposure to insecticide. This has previously been demonstrated by increased glutathione peroxidase and catalase activity in insecticide resistant An. arabiensis that had been provided multiple blood meals45. The significantly higher oxidative stress defence capacity of FUMOZ-R may underlie the ability of older females to survive deltamethrin exposure at the 10X dose.
Although this study was performed on laboratory strains, it has previously been shown that the insecticide resistance intensity of FUMOZ-R is comparable to that of wild Zambian An. funestus, and likely comparable to populations of this species in Mozambique from where the FUMOZ and FUMOZ-R strains are derived18. The high intensity resistance in these populations has been linked to reduced efficacy of pyrethroid impregnated bed nets under laboratory conditions14, 18, 31, 57,58,59,60,61.
The effect of continued exposure of mosquitoes to sub-lethal doses of insecticide is poorly understood, especially in the context of multiple blood meals. This study demonstrates that insecticide resistance intensity is amplified by multiple blood feeding. This highlights the importance of intensity screenings as part of routine surveillance because intensity assessments can be used to predict the operational significance of resistance in the field16, 17. The study also shows that the presence of high-intensity resistance has differential effects on the life history parameters of affected mosquitoes.
It is concluded that blood feeding tends to increase the longevity of An. funestus females and enhances their levels and intensity of pyrethroid resistance, even in older age groups. This is important in terms of measuring insecticide resistance in wild vector populations and making inferences about the possible impact of resistance on operational efficacy, especially because it cannot be assumed that older females will not be able to withstand the same levels of insecticide intoxication as younger females. In terms of programmatic decision-making, it should therefore be considered useful to assess the level and intensity of resistance in older female cohorts wherever possible, notwithstanding the standard protocols for resistance testing using age-standardised samples16.
References
Benelli, G. & Beier, J. C. Current vector control challenges in the fight against malaria. Acta Trop. 174, 91–96. https://doi.org/10.1016/j.actatropica.2017.06.028 (2017).
Schmidt, M., Hrabcova, V., Jun, D., Kuca, K. & Musilek, K. Vector control and insecticidal resistance in the African Malaria mosquito Anopheles gambiae. Chem. Res. Toxicol. 31, 534–547. https://doi.org/10.1021/acs.chemrestox.7b00285 (2018).
Walker, K. & Lynch, M. Contributions of Anopheles larval control to malaria suppression in tropical Africa: review of achievements and potential. Med. Vet. Entomol. 21, 2–21. https://doi.org/10.1111/j.1365-2915.2007.00674.x (2007).
Agumba, S. et al. Diagnostic dose determination and efficacy of chlorfenapyr and clothianidin insecticides against Anopheles malaria vector populations of western Kenya. Malaria J. 18, 243. https://doi.org/10.1186/s12936-019-2858-z (2019).
Dagg, K. et al. Evaluation of toxicity of clothianidin (neonicotinoid) and chlorfenapyr (pyrrole) insecticides and cross-resistance to other public health insecticides in Anopheles arabiensis from Ethiopia. Malaria J. 18, 49. https://doi.org/10.1186/s12936-019-2685-2 (2019).
Oliver, S. V. et al. Evaluation of the pyrrole insecticide chlorfenapyr against pyrethroid resistant and susceptible Anopheles funestus (Diptera: Culicidae). Trop. Med. Int. Health TM IH 15, 127–131. https://doi.org/10.1111/j.1365-3156.2009.02416.x (2010).
Raghavendra, K. et al. Chlorfenapyr: a new insecticide with novel mode of action can control pyrethroid resistant malaria vectors. Malaria J. 10, 16. https://doi.org/10.1186/1475-2875-10-16 (2011).
Knox, T. B. et al. An online tool for mapping insecticide resistance in major Anopheles vectors of human malaria parasites and review of resistance status for the Afrotropical region. Parasites Vectors 7, 76. https://doi.org/10.1186/1756-3305-7-76 (2014).
Hemingway, J. The role of vector control in stopping the transmission of malaria: threats and opportunities. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 369, 20130431, doi:https://doi.org/10.1098/rstb.2013.0431 (2014).
WHO. Guidelines for malaria vector control. https://www.who.int/malaria/publications/atoz/9789241550499/en/ (2019).
Etang, J. et al. When intensity of deltamethrin resistance in Anopheles gambiae s.l. leads to loss of Long Lasting Insecticidal Nets bio-efficacy: a case study in north Cameroon. Parasites Vectors 9, 132, doi:https://doi.org/10.1186/s13071-016-1420-x (2016).
Lopez-Monroy, B. et al. Frequency and intensity of pyrethroid resistance through the CDC bottle bioassay and their association with the frequency of kdr mutations in Aedes aegypti (Diptera: Culicidae) from Mexico. Pest Manag. Sci. https://doi.org/10.1002/ps.4916 (2018).
Omondi, S. et al. Quantifying the intensity of permethrin insecticide resistance in Anopheles mosquitoes in western Kenya. Parasites Vectors 10, 548. https://doi.org/10.1186/s13071-017-2489-6 (2017).
Pwalia, R. et al. High insecticide resistance intensity of Anopheles gambiae (s.l.) and low efficacy of pyrethroid LLINs in Accra, Ghana. Parasites Vectors 12, 299, doi:https://doi.org/10.1186/s13071-019-3556-y (2019).
Thomas, M. B. & Read, A. F. The threat (or not) of insecticide resistance for malaria control. Proc. Natl. Acad. Sci. USA 113, 8900–8902. https://doi.org/10.1073/pnas.1609889113 (2016).
WHO. Test procedures for insecticide resistance monitoring in malaria vector mosquitoes. http://www.who.int/iris/handle/10665/250677 (2016).
Bagi, J. et al. When a discriminating dose assay is not enough: measuring the intensity of insecticide resistance in malaria vectors. Malaria J. 14, 210. https://doi.org/10.1186/s12936-015-0721-4 (2015).
Venter, N. et al. Benchmarking insecticide resistance intensity bioassays for Anopheles malaria vector species against resistance phenotypes of known epidemiological significance. Parasites Vectors 10, 198. https://doi.org/10.1186/s13071-017-2134-4 (2017).
Jones, C. M. et al. Aging partially restores the efficacy of malaria vector control in insecticide-resistant populations of Anopheles gambiae s.l. from Burkina Faso. Malaria J. 11, 24, doi:https://doi.org/10.1186/1475-2875-11-24 (2012).
Barlow, F. & Hadaway, A. B. Effects of age, sex and feeding on the susceptibility of mosquitoes to insecticides. Ann. Trop. Med. Parasitol. 50, 438–443. https://doi.org/10.1080/00034983.1956.11685786 (1956).
Reiter, P., Eliason, D. A., Francy, D. B., Moore, C. G. & Campos, E. G. Apparent influence of the stage of blood meal digestion on the efficacy of ground applied ULV aerosols for the control of urban Culex mosquitoes. I. Field evidence. J. Am. Mosquito Control Assoc. 6, 366–370 (1990).
Halliday W.R. & R., F. Why does DDT toxicity change after a blood meal in adult female Culex pipiens? Pesticide Biochem. Physiol. 28, 172–181 (1987).
Ohm, J. R. et al. Rethinking the extrinsic incubation period of malaria parasites. Parasites Vectors 11, 178. https://doi.org/10.1186/s13071-018-2761-4 (2018).
Brooke, B. D. & Koekemoer, L. L. Major effect genes or loose confederations? The development of insecticide resistance in the malaria vector Anopheles gambiae. Parasites Vectors 3, 74. https://doi.org/10.1186/1756-3305-3-74 (2010).
Amenya, D. A. et al. Over expression of a cytochrome P450 (CYP6P9) in a major African malaria vector, Anopheles funestus, resistant to pyrethroids. Insect Mol. Biol. 17, 19–25. https://doi.org/10.1111/j.1365-2583.2008.00776.x (2008).
Matambo, T. S., Paine, M. J., Coetzee, M. & Koekemoer, L. L. Sequence characterization of cytochrome P450 CYP6P9 in pyrethroid resistant and susceptible Anopheles funestus (Diptera: Culicidae). Genet. Mol. Res. GMR 9, 554–564. https://doi.org/10.4238/vol9-1gmr719 (2010).
Wondji, C. S. et al. Two duplicated P450 genes are associated with pyrethroid resistance in Anopheles funestus, a major malaria vector. Genome Res. 19, 452–459. https://doi.org/10.1101/gr.087916.108 (2009).
Irving, H., Riveron, J. M., Ibrahim, S. S., Lobo, N. F. & Wondji, C. S. Positional cloning of rp2 QTL associates the P450 genes CYP6Z1, CYP6Z3 and CYP6M7 with pyrethroid resistance in the malaria vector Anopheles funestus. Heredity 109, 383–392. https://doi.org/10.1038/hdy.2012.53 (2012).
Wood, O., Hanrahan, S., Coetzee, M., Koekemoer, L. & Brooke, B. Cuticle thickening associated with pyrethroid resistance in the major malaria vector Anopheles funestus. Parasites Vectors 3, 67. https://doi.org/10.1186/1756-3305-3-67 (2010).
Casimiro, S. L., Hemingway, J., Sharp, B. L. & Coleman, M. Monitoring the operational impact of insecticide usage for malaria control on Anopheles funestus from Mozambique. Malaria J. 6, 142. https://doi.org/10.1186/1475-2875-6-142 (2007).
Coetzee, M. & Koekemoer, L. L. Molecular systematics and insecticide resistance in the major African malaria vector Anopheles funestus. Annu. Rev. Entomol. 58, 393–412. https://doi.org/10.1146/annurev-ento-120811-153628 (2013).
Okoye, P. N., Brooke, B. D., Hunt, R. H. & Coetzee, M. Relative developmental and reproductive fitness associated with pyrethroid resistance in the major southern African malaria vector Anopheles funestus. Bull. Entomol. Res. 97, 599–605. https://doi.org/10.1017/s0007485307005317 (2007).
Charlwood, J. D. et al. Density independent feeding success of malaria vectors (Diptera: Culicidae) in Tanzania. Bull. Entomol. Res. 85, 29–35. https://doi.org/10.1017/S0007485300051981 (2009).
Gillies, M. T. & Meillon, B. D. The anophelinae of Africa south of the Sahara (Ethiopian Zoogeographical Region). Publ. S. Afr. Inst. Med. Res. 54 (1968).
Sinka, M. E. et al. The dominant Anopheles vectors of human malaria in Africa, Europe and the Middle East: occurrence data, distribution maps and bionomic precis. Parasites Vectors 3, 117. https://doi.org/10.1186/1756-3305-3-117 (2010).
Kaindoa, E. W. et al. Interventions that effectively target Anopheles funestus mosquitoes could significantly improve control of persistent malaria transmission in south-eastern Tanzania. PLoS ONE 12, e0177807. https://doi.org/10.1371/journal.pone.0177807 (2017).
Lwetoijera, D. W. et al. Increasing role of Anopheles funestus and Anopheles arabiensis in malaria transmission in the Kilombero Valley, Tanzania. Malaria J. 13, 331. https://doi.org/10.1186/1475-2875-13-331 (2014).
Ferguson, H. M. & Read, A. F. Mosquito appetite for blood is stimulated by Plasmodium chabaudi infections in themselves and their vertebrate hosts. Malaria J. 3, 12. https://doi.org/10.1186/1475-2875-3-12 (2004).
Wekesa, J. W., Copeland, R. S. & Mwangi, R. W. Effect of Plasmodium falciparum on blood feeding behavior of naturally infected Anopheles mosquitoes in western Kenya. Am. J. Trop. Med. Hyg. 47, 484–488. https://doi.org/10.4269/ajtmh.1992.47.484 (1992).
Garrett-Jones, C. & Shidrawi, G. R. Malaria vectorial capacity of a population of Anopheles gambiae: an exercise in epidemiological entomology. Bull. World Health Organ. 40, 531–545 (1969).
Christian, R. N. et al. Age-related pyrethroid resistance is not a function of P450 gene expression in the major African malaria vector, Anopheles funestus (Diptera: Culicidae). Genet. Mol. Res. GMR 10, 3220–3229. https://doi.org/10.4238/2011.December.21.4 (2011).
Spillings, B. L., Coetzee, M., Koekemoer, L. L. & Brooke, B. D. The effect of a single blood meal on the phenotypic expression of insecticide resistance in the major malaria vector Anopheles funestus. Malaria J. 7, 226. https://doi.org/10.1186/1475-2875-7-226 (2008).
Hauser, G., Thiévent, K. & Koella, J. C. The ability of Anopheles gambiae mosquitoes to bite through a permethrin-treated net and the consequences for their fitness. Sci. Rep. 9, 8141. https://doi.org/10.1038/s41598-019-44679-1 (2019).
Oliver, S. V. & Brooke, B. D. The effect of multiple blood-feeding on the longevity and insecticide resistant phenotype in the major malaria vector Anopheles arabiensis (Diptera: Culicidae). Parasites Vectors 7, 390. https://doi.org/10.1186/1756-3305-7-390 (2014).
Oliver, S. V. & Brooke, B. D. The role of oxidative stress in the longevity and insecticide resistance phenotype of the major malaria vectors Anopheles arabiensis and Anopheles funestus. PLoS ONE 11, e0151049. https://doi.org/10.1371/journal.pone.0151049 (2016).
Hunt, R. H., Brooke, B. D., Pillay, C., Koekemoer, L. L. & Coetzee, M. Laboratory selection for and characteristics of pyrethroid resistance in the malaria vector Anopheles funestus. Med. Vet. Entomol. 19, 271–275. https://doi.org/10.1111/j.1365-2915.2005.00574.x (2005).
Marinotti, O., Nguyen, Q. K., Calvo, E., James, A. A. & Ribeiro, J. M. Microarray analysis of genes showing variable expression following a blood meal in Anopheles gambiae. Insect Mol. Biol. 14, 365–373. https://doi.org/10.1111/j.1365-2583.2005.00567.x (2005).
Cawley, M. J. in The R Book 511–526 (2007).
Awolola, T. S. et al. Pyrethroids resistance intensity and resistance mechanisms in Anopheles gambiae from malaria vector surveillance sites in Nigeria. PLoS ONE 13, e0205230. https://doi.org/10.1371/journal.pone.0205230 (2018).
Orjuela, L. I. et al. Insecticide resistance and its intensity in populations of malaria vectors in Colombia. BioMed. Res. Int. 2018, 9163543. https://doi.org/10.1155/2018/9163543 (2018).
Hughes, A., Lissenden, N., Viana, M., Toe, K. H. & Ranson, H. Anopheles gambiae populations from Burkina Faso show minimal delayed mortality after exposure to insecticide-treated nets. Parasites Vectors 13, 17. https://doi.org/10.1186/s13071-019-3872-2 (2020).
Brooke, B. D. et al. Bioassay and biochemical analyses of insecticide resistance in southern African Anopheles funestus (Diptera: Culicidae). Bull. Entomol. Res. 91, 265–272. https://doi.org/10.1079/ber2001108 (2001).
Smith, C. E. The significance of mosquito longevity and blood-feeding behaviour in the dynamics of arbovirus infections. Med. Biol. 53, 288–294 (1975).
Barreaux, A. M. G., Stone, C. M., Barreaux, P. & Koella, J. C. The relationship between size and longevity of the malaria vector Anopheles gambiae (s.s.) depends on the larval environment. Parasites Vectors 11, 485, doi:https://doi.org/10.1186/s13071-018-3058-3 (2018).
Otali, D. et al. Increased production of mitochondrial reactive oxygen species and reduced adult life span in an insecticide-resistant strain of Anopheles gambiae. Bull. Entomol. Res. 104, 323–333. https://doi.org/10.1017/s0007485314000091 (2014).
Grossman, M. K., Oliver, S. V., Brooke, B. D. & Thomas, M. B. Use of alternative bioassays to explore the impact of pyrethroid resistance on LLIN efficacy. Parasites Vectors 13, 179. https://doi.org/10.1186/s13071-020-04055-9 (2020).
Glunt, K. D. et al. Long-lasting insecticidal nets no longer effectively kill the highly resistant Anopheles funestus of southern Mozambique. Malaria J. 14, 298. https://doi.org/10.1186/s12936-015-0807-z (2015).
Riveron, J. M. et al. Escalation of pyrethroid resistance in the malaria vector Anopheles funestus induces a loss of efficacy of piperonyl butoxide-based insecticide-treated nets in mozambique. J. Infect. Dis. 220, 467–475. https://doi.org/10.1093/infdis/jiz139 (2019).
Tchakounte, A. et al. Exposure to the insecticide-treated bednet PermaNet 2.0 reduces the longevity of the wild African malaria vector Anopheles funestus but GSTe2-resistant mosquitoes live longer. PLoS ONE 14, e0213949, doi:https://doi.org/10.1371/journal.pone.0213949 (2019).
Toe, K. H. et al. Increased pyrethroid resistance in malaria vectors and decreased bed net effectiveness, Burkina Faso. Emerg. Infect. Dis. 20, 1691–1696. https://doi.org/10.3201/eid2010.140619 (2014).
Riveron, J. M. et al. Multiple insecticide resistance in the major malaria vector Anopheles funestus in southern Ghana: implications for malaria control. Parasites Vectors 9, 504. https://doi.org/10.1186/s13071-016-1787-8 (2016).
Acknowledgements
Dr Rodney Hull is thanked for his valuable comments on the drafting of the manuscript.
Funding
This project was funded by a National Research Foundation Competitive Support for Unrated researchers grant [SRUG190313423259] as well as National Health Research Development Grants [2017-2DEV08SOL01 and 2017-2DEV08SOL02] to SVO.
Author information
Authors and Affiliations
Contributions
S.V.O. and B.D.B. conceived the project. S.V.O. performed the experiments; S.V.O. and C.L.L. performed the data analysis; and S.V.O. produced the initial draft of the manuscript. B.D.B. produced the final version of the manuscript. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Oliver, S.V., Lyons, C.L. & Brooke, B.D. The effect of blood feeding on insecticide resistance intensity and adult longevity in the major malaria vector Anopheles funestus (Diptera: Culicidae). Sci Rep 12, 3877 (2022). https://doi.org/10.1038/s41598-022-07798-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-07798-w
- Springer Nature Limited
This article is cited by
-
Temperature, mosquito feeding status and mosquito density influence the measured bio-efficacy of insecticide-treated nets in cone assays
Parasites & Vectors (2024)
-
Pyrethroid-treated bed nets impair blood feeding performance in insecticide resistant mosquitoes
Scientific Reports (2023)
-
Natural plant diet impacts phenotypic expression of pyrethroid resistance in Anopheles mosquitoes
Scientific Reports (2022)