Abstract
Sodium–glucose cotransporter 2 inhibitor (SGLT2i) treatment is a therapeutic approach for type 2 diabetes mellitus (T2DM). Some reports have shown that SGLT2i treatment improves insulin resistance; however, few studies have evaluated insulin resistance by the glucose clamp method. Hepatic insulin clearance (HIC) is a new pathophysiological mechanism of T2DM. The effect of SGLT2i treatment on hepatic insulin clearance and insulin resistance is not well known. We investigated the effect of SGLT2i treatment on insulin resistance, insulin secretion, incretin levels, body composition, and hepatic insulin clearance. We conducted a meal tolerance test (MTT) and a hyperinsulinemic-euglycemic clamp test in 9 T2DM patients. Ipragliflozin (50 mg/day) was administered, and the MTT and clamp test were performed after 4 months. We calculated HIC as the postprandial C-peptide AUC-to-insulin AUC ratio. We also measured GLP-1, GIP, and glucagon levels during the MTT. Body weight and HbA1c were decreased, although not significantly, after 4 months of treatment. Postprandial glucose, fasting insulin and postprandial insulin were significantly decreased. Insulin resistance with the glucose clamp was not changed, but the HOMA-IR and insulin sensitivity indices were significantly improved. Incretin and glucagon levels were not changed. Hepatic insulin clearance was significantly increased, but whole-body insulin clearance was not changed. The FIB-4 index and fatty liver index were significantly reduced. The HOMA-beta and insulinogenic indices were not changed, but the C-peptide index was significantly increased. Although the number of patients was small, these results suggested that SGLT2i treatment improved liver function, decreased hepatic insulin resistance, and increased hepatic insulin clearance, despite the small weight reduction.
Similar content being viewed by others
Introduction
The main pathophysiology of type 2 diabetes mellitus (T2DM) is decreased insulin sensitivity and decreased insulin secretion ability1. Sodium–glucose cotransporter inhibitor treatment (SGLT2i) is an antidiabetic therapeutic approach that increases glucosuria, induces a glucose-lowering effect, and reduces body weight2. Some studies have reported that SGLT2i treatment improves insulin resistance. Dapagliflozin was found to improve muscle insulin resistance, as evaluated by the euglycemic hyperinsulinemic clamp technique, in an American population3. However, this study and other studies indicated that SGLT2i treatment increases endogenous glucose production and glucagon secretion. In one study performed by dual-tracer glucose administration combined with a mixed meal in a European population, SGLT2i treatment increased insulin sensitivity and the secretion of glucagon-like peptide-1 (GLP-1) but increased endogenous glucose production and glucagon secretion4. In another study, a glucose clamp test and an oral glucose tolerance test were performed, and SGLT2i treatment was found to improve insulin sensitivity in an American population5. These results suggest that SGLT2i treatment improves muscle insulin sensitivity but increases endogenous glucose production via glucagon secretion.
Hepatic insulin clearance (HIC) is a novel and important pathophysiological mechanism of hyperinsulinemia, metabolic syndrome, and T2DM; hepatic insulin clearance is decreased in these metabolic conditions6. One study reported that SGLT2i treatment increased insulin clearance after the meal tolerance test7. However, we previously reported that Japanese patients with poorly controlled T2DM showed increased hepatic insulin clearance8.
An American study showed that the SGLT2i canagliflozin improved liver insulin sensitivity and beta-cell function and increased hepatic insulin clearance, as evaluated by the glucose clamp method and a mixed meal tolerance test9. However, the pathophysiology of diabetes is very different between American and Asian people; Asian people exhibit a low insulin secretion ability and insulin resistance with mild obesity10,11. Therefore, we need an study of SGLT2i treatment in Asian individuals.
According to the above results, SGLT2i treatment affects insulin resistance, incretin levels, and hepatic insulin clearance; however, these effects were evaluated in different studies and, therefore, the complete mechanism in the real world, especially in Asian or Japanese people, is not well known. We evaluated the effect of SGLT2i treatment on insulin resistance and whole-body insulin clearance by the glucose clamp method, and we evaluated the effect on hepatic insulin clearance and beta-cell function by a meal tolerance test in Japanese individuals. We also measured the levels of incretins such as GLP-1 and glucose-dependent insulinotropic polypeptide (GIP), as well as glucagon levels, by a meal tolerance test. Here, we show that SGLT2i treatment improves hepatic insulin resistance, increases hepatic insulin clearance, and improves liver function in Japanese people.
Materials and methods
Subjects
Nine type 2 diabetes patients were enrolled in the study at Tottori University Hospital between 2014 and 2017. We diagnosed type 2 diabetes by the World Health Organization (WHO) criteria12. We excluded patients with malignancy, hepatitis B or C, liver cirrhosis, pancreatitis, renal dysfunction, or current use of diabetogenic medications such as corticosteroids from the study. All patients were of Japanese ethnicity. This study had a single-arm design. Ipragliflozin (50 mg/day) was administered after the meal tolerance test (MTT) and glucose clamp test, and the MTT and clamp test were performed after 4 months. Patients were instructed to drink water to prevent dehydration.
We performed this study according to the principles of the Declaration of Helsinki. The Ethics Committee of the Tottori University Faculty of Medicine approved this study on 30/05/2014 (approval number 2439). The clinical trial number was UMIN000014521.
Trial registration: UMIN, UMIN000014521, registered (09/07/2014)Retrospectively registered, https://center6.umin.ac.jp/cgi-open-bin/ctr_e/ctr_view.cgi?recptno=R000016186.
Involvement of patients and the public
The first patient was enrolled on 3 June, 2014. The patient was recruited from the outpatient department of Tottori University Hospital. The research questions were developed by the Ethics Committee and provided to all of the patients using a procedure that was approved by the Ethics Committee. This study was a single-arm intervention study. We chose the outcomes of HOMA-IR index and glucose disposal rate as the markers of insulin resistance. All patients were recruited as the outpatient facility of Tottori University Hospital. We obtained informed consent from all of the patients using a procedure that was approved by the Ethics Committee.
Meal tolerance test (MTT)
The patients visited our hospital after overnight fasting. The patients ate a test meal developed by the Japan Diabetes Society (JDS) (460 kcal, 50% carbohydrate, 15% protein, 35% fat, and 1.6 g of salt)13. Plasma glucose, serum insulin, and C-peptide were measured at 0, 30, 60, 120, and 180 min after consumption of the test meal. We used the glucose oxidase method for glucose measurement and chemiluminescent immunoassays for insulin and C-peptide measurement (human insulin and C-peptide chemiluminescent immunoassay kits; Kyowa Medix, Japan). HbA1c was measured by high-performance liquid chromatography. HbA1c levels were converted from percentage values to the International Federation of Clinical Chemistry values (mmol/mol), and an HbA1c converter from the National Institutes of Diabetes and Digestive and Kidney Diseases was used14.
Insulin resistance was assessed by using insulin resistance indices calculated as follows:15,16
Insulin secretion was assessed by using insulin secretion indices as follows:6,15,17
We calculated hepatic insulin clearance (HIC) as follows:
\({\text{HIC }} = \, \left\{ {{\text{area under the curve }}\left( {{\text{AUC}}} \right){\text{ for Cpeptide}}, \, 0{-}{12}0{\text{ min }}} \right\}/\left\{ {{\text{AUC for insulin}}, \, 0{-}{12}0{\text{ min}}) \, } \right\}\)6. The AUC was calculated using a trapezoidal method.
GLP-1, GIP, and glucagon assays
We obtained serum samples by using blood collection tubes containing a DPP-4 inhibitor (BD™ P800, BD Japan, Japan). We performed ethanol and solid-phase extractions for the assay18. We measured intact GLP-1 levels with an ELISA kit (GLP-1 (active) ELISA, Merck Millipore, Germany). We also measured active GIP with an ELISA kit (Human GIP, Active form Assay Kit, IBL, Japan). We next measured glucagon levels with an ELISA kit (Glucagon ELISA kit, Mercodia, Sweden).
Hyperinsulinemic-euglycemic clamp
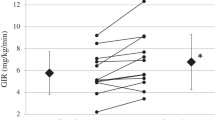
The glucose clamp method was performed as we previously reported8,19. We performed the hyperinsulinemic-euglycemic clamp method to evaluate insulin sensitivity with an artificial endocrine pancreas (STG 55, Nikkiso, Japan). We performed the protocol with a priming constant insulin infusion (100 mU/m2/min), and we maintained the plasma glucose level at 5.2 mmol/L (95 mg/dL) during the clamp. The steady-state plasma insulin level was reached at 1200 pmol/L in T2DM patients, based on previous studies20. We measured the steady-state glucose infusion rate (GIR) from 90–120 min, and we defined the mean GIR during this period as the glucose disposal rate (GDR). The GDR is generally used to mainly reflect peripheral insulin sensitivity. We next calculated the M/I value by setting the M value equal to the GDR and the I value equal to the final insulin concentration during the clamp, i.e., the peripheral insulin sensitivity adjusted by the final insulin concentration21. Whole-body insulin clearance was calculated as the insulin infusion rate divided by the steady-state insulin concentration21.
FIB-4 index
The liver fibrosis level was estimated by the \({\text{FIB-4 index}}:{\rm{ age }}(\left[ {{\rm{yr}}} \right]\times{\rm{AST }}\left[ {{\rm{U}}/{\rm{L}}} \right]){\rm{ }}/{\rm{ }}(\left( {{\rm{PLT }}\left[ {{\rm{1}}{0^{\rm{9}}}/{\rm{L}}} \right]} \right)\times{({\rm{ALT}}\left[ {{\rm{U}}/{\rm{L}}} \right])^{{\rm{1}}/{\rm{2}}}})\)22.
Fatty liver index (FLI)
The fatty liver index was estimated as follows:23
Body composition
Body composition was analyzed with an Inbody 770 body composition analyzer (Inbody Japan, Tokyo, Japan). Body water, percent body fat, body fat mass, lean body mass, skeletal muscle mass, trunk fat mass, and bone mineral mass were measured.
Statistical analysis
Data are expressed as the mean ± standard deviation values. We assessed differences in the mean values of clinical parameters before and after SGLT2i treatment by using the Mann–Whitney U test. We identified correlations between nonparametric clinical variables by using Spearman correlation analysis. Values of P < 0.05 were considered to be significant. PRISM8 software (GraphPad Software, San Diego, USA) was used for all analyses. We conducted a power analysis to compare the pretreatment and posttreatment HOMA-IR indices as a primary endpoint using an EZR calculator24.
Ethical approval
This study was approved by the Ethics Committee of the Faculty of Medicine, Tottori University (approval number 2439). The clinical trial number is UMIN000014521, registered 30/05/2014.
Results
The mean age of the subjects was 54.1 ± 9.1 years, and there were 2 males and 7 females. The duration of diabetes was 5.0 ± 4.1 years. The participant characteristics are shown in Table 1. Seven patients were treated with only diet therapy, 2 patients took DPP4 inhibitors and alpha-glucosidase inhibitors, and 1 patient also took metformin. Table 1 shows the effects of SGLT2i treatment for 4 months. Body weight and HbA1c showed decreasing trends, but the trend was not significant. Body composition was not significantly changed. AST, the FIB-4 index, and the fatty liver index were significantly decreased. Postprandial glucose, fasting insulin and postprandial insulin were decreased. Figure 1 shows the MTT results. Glucose was significantly decreased 120 min after the MTT. The fasting, 60-min, and 120-min insulin levels were significantly decreased. C-peptide, GLP-1, GIP, and glucagon levels were not changed. Insulin resistance with the glucose clamp was not changed, but the HOMA-IR and insulin sensitivity indices were significantly improved. Incretin levels were not changed. Hepatic insulin clearance was significantly increased. Whole-body insulin clearance was not changed. The HOMA-beta and insulinogenic indices were not changed, but the CPI was significantly increased.
We conducted a power analysis to compare the pretreatment and posttreatment HOMA-IR index as a primary endpoint. The mean pretreatment HOMA-IR index was 4.86 ± 1.14, and the mean posttreatment HOMA-IR index was 3.48 ± 1.53. The difference between the mean pretreatment and posttreatment HOMA-IR indices was 1.38 ± 0.39. We conducted a power analysis of the statistical test that was used to compare the pre- and posttreatment HOMA-IR indices. Assuming that the difference in the means was 1.38, the SD was 0.39, alpha was 0.05, the 1-beta was 0.8, and R was 0.57, the estimated power was > 99.9%.
Table 2 shows the correlations between the changes in the HOMA-IR index and clinical parameters from pretreatment to after treatment. The change in hepatic insulin clearance was significantly correlated with the change in the HOMA-IR index. The change in waist circumference was also correlated with the change in the HOMA-IR index.
Discussion
Our study indicated that treatment with the SGLT2i ipragliflozin improved the HOMA-IR index, liver function, and the C-peptide index but did not significantly change muscle insulin resistance, as evaluated by the glucose clamp method, nor did it alter body weight or body composition. SGLT2i increased hepatic insulin clearance but not whole-body insulin clearance. In a previous report, tofogliflozin was found to significantly improve insulin sensitivity and peripheral glucose uptake in Japanese patients with T2DM25. These improvements were significantly correlated with the reduction in body fat mass. Our study showed a tendency toward weight reduction, but the difference was not significant. This previous study included 16 patients and was a 12-week study; therefore, we consider our study to have a small sample number. Our study showed a trend toward a reduction in body water, but body fat mass did not change; these findings were different from those of the past study. However, the liver function parameters in our study were significantly improved, despite the small weight reduction, and we consider these points to be important. Dapagliflozin reduced liver fat but did not affect insulin sensitivity in liver tissue, muscle tissue, or adipocytes26. Another recent study also reported that SGLT2i treatment increased hepatic insulin clearance, as evaluated by an oral meal tolerance test27. A previous article reported that SGLT2i treatment increased GLP-1 and glucagon secretion4 and increased glucagon secretion from alpha cells28. We did not observe significant glucagon inhibition or changes in incretin levels by SGLT2i treatment, but this may depend on the small number of patients and the fact that 2 patients were already taking DPP4i drugs.
According to these results, SGLT2i decreased postprandial glucose levels and insulin levels. The HOMA-IR index mainly reflects liver insulin resistance, the glucose clamp method mainly reflects muscle insulin resistance, and the insulin sensitivity index reflects insulin sensitivity in both liver and muscle29. Although muscle insulin resistance, as evaluated by the glucose clamp method, was not improved, liver insulin resistance, as evaluated by the HOMA-IR index, was considered to be improved. Hyperinsulinemia was ameliorated after SGLT2i treatment, and this insulin-lowering effect might be induced by increased hepatic insulin clearance. These effects resulted in improved liver function. Finally, the C-peptide index was increased, which might mean that SGLT2i treatment has a protective effect on beta cells. Although further basic research in animals and cells is needed to understand the molecular mechanisms, our results contribute to understanding the effects of SGLT2i treatment. Asian individuals, who exhibit a low insulin secretion ability and insulin resistance with mild obesity, have a T2DM pathophysiology different from that of American and European individuals10,11. All patients in our study were Japanese; therefore, our results contribute to everyday clinical work in the Asian population. A recent Japanese study indicated that the SGLT2i tofogliflozin did not change whole-body insulin clearance30. According to these results, SGLT2i treatment mainly increases hepatic insulin clearance, not whole-body insulin clearance. We consider these results to be important for understanding the effects of SGLT2i treatment and the pathophysiology of T2DM.
Our study has some limitations. The major limitation is the very small number of patients (nine). However, glucose clamp tests and meal tolerance tests, including incretins, are very complicated and expensive studies. We need a larger study. We did not observe a significant reduction in HbA1c or body weight in this study, and 4 months might be a short treatment period. We evaluated insulin resistance by the glucose clamp method. In a previous report, the GDR was evaluated by subtracting the urinary glucose excreted from the total glucose administered3. We measured urinary glucose excretion in only 2 patients; therefore, we could not evaluate urinary glucose excretion, and we must consider this point. We did not evaluate hepatic insulin resistance by the tritium-glucose method because of the difficulty of using tritium-glucose in Japan. We evaluated hepatic insulin resistance by using only the HOMA-IR index, and further study is needed. We used the MTT to avoid hyperglycemia resulting from the OGTT in patients with severe diabetes. However, a previous study used the HIC obtained from the MTT; therefore, we think our method is acceptable7. Although our main purpose was to evaluate hepatic insulin clearance, whole-body and peripheral insulin clearance are also important, as previously reported31. Unfortunately, the steady-state C-peptide levels with the glucose clamp were not measured in this study, and we need to investigate the peripheral insulin clearance evaluated during the glucose clamp test. A recent article stated that the postload CPR/IRI molar ratio does not accurately evaluate hepatic insulin clearance32. We must be careful to evaluate our results. We evaluated beta-cell function by the C-peptide index. However, the C-peptide index is affected by renal function. We excluded patients with renal disorder as determined by Cr > 1.3 mg/ml, according to a past report33, and eGFR did not show a significant change; therefore, we consider our data to be reliable. Despite these limitations, we performed glucose clamp and meal tolerance tests and incretin measurement in Japanese patients with T2DM in a real-world setting. We consider that our study might contribute to our everyday clinical work, especially in the Asian population.
In conclusion, SGLT2i treatment improved liver function, decreased hepatic insulin resistance, and increased hepatic insulin clearance, despite the small weight reduction.
Data availability
No additional data are available.
Abbreviations
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- AUC:
-
Area under the curve
- BMI:
-
Body mass index
- CPI:
-
C-peptide index
- CPR:
-
C-peptide immunoreactivity
- eGFR:
-
Estimated glomerular filtration rate
- FLI:
-
Fatty liver index
- GDR:
-
Glucose disposal rate
- GIP:
-
Glucose-dependent insulinotropic polypeptide
- GLP-1:
-
Glucagon-like peptide-1
- GGTP:
-
γ-Glutamyl transpeptidase
- HDL:
-
High-density lipoprotein
- HIC:
-
Hepatic insulin clearance
- HOMA-beta:
-
Homeostatic model assessment for beta-cell function
- HOMA-IR:
-
Homeostatic model assessment for insulin resistance
- LDL:
-
Low-density lipoprotein
- SGLT2i:
-
Sodium–glucose cotransporter 2 inhibitor
References
Kahn, S. E., Cooper, M. E. & Del Prato, S. Pathophysiology and treatment of type 2 diabetes: perspectives on the past, present, and future. Lancet 383, 1068–1083 (2014).
Ohkura, T. Ipragliflozin: a novel sodium–glucose cotransporter 2 inhibitor developed in Japan. World J. Diabetes. 6, 136–144 (2015).
Merovci, A. et al. Dapagliflozin improves muscle insulin sensitivity but enhances endogenous glucose production. J. Clin. Invest. 124, 509–514 (2014).
Ferrannini, E. et al. Metabolic response to sodium–glucose cotransporter 2 inhibition in type 2 diabetic patients. J. Clin. Invest. 124, 499–508 (2014).
Mudaliar, S. et al. Changes in insulin sensitivity and insulin secretion with the sodium glucose cotransporter 2 inhibitor dapagliflozin. Diabetes Technol. Ther. 16, 137–144 (2014).
Pivovarova, O. et al. Hepatic insulin clearance is closely related to metabolic syndrome components. Diabetes Care 36, 3779–3785 (2013).
Ekholm, E. et al. Combined treatment with saxagliptin plus dapagliflozin reduces insulin levels by increased insulin clearance and improves beta-cell function. Endocr. Pract. 23(3), 258–265 (2017).
Okura, T. et al. Hepatic insulin clearance is increased in patients with high HbA1c type 2 diabetes: a preliminary report. BMJ Open Diabetes Res. Care. 8, e001149 (2020).
Cusi, K. et al. Effect of canagliflozin treatment on hepatic triglyceride content and glucose metabolism in patients with type 2 diabetes. Diabetes Obes. Metab. 21, 812–821 (2019).
Hsu, W. C., Araneta, M. R., Kanaya, A. M., Chiang, J. L. & Fujimoto, W. BMI cut points to identify at-risk Asian Americans for type 2 diabetes screening. Diabetes Care 38, 150–158 (2015).
Okura, T. et al. Body mass index ≥23 is a risk factor for insulin resistance and diabetes in Japanese people: A brief report. PLoS ONE 13, e0201052 (2018).
Alberti, K. G. & Zimmet, P. Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet. Med. 15, 539–553 (1998).
Yoshino, G. et al. The test meal A: A pilot model for the international standard of test meal for an assessment of both postprandial hyperglycemia and hyperlipidemia. J. Jpn. Diabetes Soc. 49, 361–371 (2006) ((In Japanese)).
National Institutes of Diabetes and Digestive and Kidney Diseases, the HbA1c converter, [article online]. 1999. Available from: http://www.ngsp.org/convert1.asp.
Matthews, D. R. et al. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28, 412–419 (1985).
Matsuda, M. & DeFronzo, R. A. Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care 22, 1462–1470 (1999).
Iwata, M. et al. Genetic risk score constructed using 14 susceptibility alleles for type 2 diabetes is associated with the early onset of diabetes and may predict the future requirement of insulin injections among Japanese individuals. Diabetes Care 35, 1763–1770 (2012).
Yabe, D. et al. Effects of DPP-4 inhibitor linagliptin and GLP-1 receptor agonist liraglutide on physiological response to hypoglycaemia in Japanese subjects with type 2 diabetes: a randomized, open-label, 2-arm parallel comparative, exploratory trial. Diabetes Obes. Metab. 19, 442–447 (2017).
Ohkura, T. et al. 20/(fasting C-peptide x fasting plasma glucose) is a simple and effective index of insulin resistance in patients with type 2 diabetes mellitus: a preliminary report. Cardiovasc. Diabetol. 12, 21 (2013).
Tamura, Y. et al. Effects of diet and exercise on muscle and liver intracellular lipid contents and insulin sensitivity in type 2 diabetic patients. J. Clin. Endocrinol. Metab. 90, 3191–3196 (2005).
DeFronzo, R. A., Tobin, J. D. & Andres, R. Glucose clamp technique: a method for quantifying insulin secretion and resistance. Am. J. Physiol. 237, 214–223 (1979).
Sterling, R. K. et al. APRICOT Clinical Investigators Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology 43, 1317–1325 (2006).
Bedogni, G. et al. The Fatty Liver Index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 6, 33 (2006).
Kanda, Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant. 48, 452–458 (2013).
Matsuba, R., Matsuba, I., Shimokawa, M., Nagai, Y. & Tanaka, Y. Tofogliflozin decreases body fat mass and improves peripheral insulin resistance. Diabetes Obes. Metab. 20, 1311–1315 (2018).
Latva-Rasku, A. et al. The SGLT2 inhibitor dapagliflozin reduces liver fat but does not affect tissue insulin sensitivity: a randomized, double-blind, placebo-controlled study with 8-week treatment in type 2 diabetes patients. Diabetes Care 42, 931–937 (2019).
Matsubayashi, Y. et al. Association of increased hepatic insulin clearance and change in serum triglycerides or beta-hydroxybutyrate concentration via the sodium/glucose-cotransporter 2 inhibitor tofogliflozin. Diabetes Obes. Metab. 22, 947–956 (2020).
Suga, T. et al. SGLT1 in pancreatic α cells regulates glucagon secretion in mice, possibly explaining the distinct effects of SGLT2 inhibitors on plasma glucagon levels. Mol. Metab. 19, 1–12 (2019).
Muniyappa, R., Lee, S., Chen, H. & Quon, M. J. Current approaches for assessing insulin sensitivity and resistance in vivo: advantages, limitations, and appropriate usage. Am. J. Physiol. Endocrinol. Metab. 294, E15-26 (2008).
Sato, M. et al. Short-term SGLT2 inhibitor administration does not alter systemic insulin clearance in type 2 diabetes. Biomedicines. 9, 1154 (2021).
Elahi, D. et al. Feedback inhibition of insulin secretion by insulin: relation to the hyperinsulinemia of obesity. N Engl. J. Med. 306, 1196–1202 (1982).
Piccinini, F. & Bergman, R. N. The measurement of insulin clearance. Diabetes Care 43, 2296–2302 (2020).
Funakoshi, S. et al. Analysis of factors influencing pancreatic beta-cell function in Japanese patients with type 2 diabetes: association with body mass index and duration of diabetic exposure. Diabetes Res. Clin. Pract. 82, 353–358 (2008).
Acknowledgements
We thank Ms. Satomi Endo for her excellent technical assistance.
Funding
This study was funded by Astellas Pharma, Inc. No grant number. The company was not involved in the study design, patient selection, data aggregation/analysis, interpretation of results, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed significantly. T.O. participated in the design of the study and performed the statistical analysis. Y.F., R.N., S.K., Y.I., M.A., K.M., S.K., K.M., S.I., H.O., and E.U. collected the data. M.K., T.I., S.T., and K.Y. conceived the study, participated in its design and coordination, and helped to draft the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Okura, T., Fujioka, Y., Nakamura, R. et al. The sodium–glucose cotransporter 2 inhibitor ipragliflozin improves liver function and insulin resistance in Japanese patients with type 2 diabetes. Sci Rep 12, 1896 (2022). https://doi.org/10.1038/s41598-022-05704-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-05704-y
- Springer Nature Limited
This article is cited by
-
Fasting hepatic insulin clearance reflects postprandial hepatic insulin clearance: a brief report
Diabetology & Metabolic Syndrome (2023)
-
Empagliflozin improves insulin sensitivity in patients with recent acute coronary syndrome and newly detected dysglycaemia
Cardiovascular Diabetology (2023)