Abstract
The removal and chemical speciation changes of heavy metals in the sewage sludge during the single bioleaching and combined bioleaching/Fenton-like processes were compared in this study. The improvement in the dewaterability of the treated sludge was also investigated. The single bioleaching led to a removal of Zn, Cu, Cd, Cr, Mn, Ni, As and Pb of 67.28%, 50.78%, 64.86%, 6.32%, 56.15%, 49.83%, 20.78% and 10.52% in 10 days, respectively. The chemical speciation analysis showed that the solubilization of heavy metals in mobile forms (exchangeable/acid soluble and reducible forms) and oxidizable form was the main reason for their removal. Subsequent Fenton-like treatment was carried out at different bioleaching stages when the bioleached sludge dropped to certain pH values (4.5, 4.0 and 3.0), by adding H2O2 at different dosages. The highest removal ratio of Zn, Cu, Cd, Cr, Mn and Ni could reach 75.53%, 52.17%, 71.91%, 11.63%, 66.29% and 65.19% after combined bioleaching/Fenton-like process, respectively, with appropriate pH and H2O2 dosages in less than 6 days. The solubilization efficiencies of these heavy metals in mobile forms were further improved by Fenton-like treatment. The removal efficiencies of As and Pb decreased due to their transformation into insoluble forms (mostly residual fraction) after Fenton treatment. The capillary suction times (CST) of the raw sludge (98.7 s) decreased by 79.43% after bioleaching and 87.44% after combined process, respectively.
Similar content being viewed by others
Introduction
The amount of sewage sludge is increasing fast worldwide due to the growing wastewater quantity and stringent environmental regulations. Land application of sewage sludge after appropriate treatment has become an important disposal alternative due to its high content of organic and inorganic nutrients1,2,3. However, the presence of contaminants in sewage sludge, especially heavy metals which are toxic and non-biodegradable, limits its utilization as fertilizer or soil amendment4,5,6,7.
Bioleaching has been considered as an environmentally friendly and promising method for the removal of heavy metals from contaminated sediment and sludge5,8,9. Chemoautotrophic iron-oxidizing and sulfur-oxidizing strains can generate sulfuric acid or ferric ions using S0 or Fe2+ as substrate, leading to a highly acidic environment and generation of soluble metal sulfate, and thus enable the metals to move from solid phase to liquid phase5,10. The rate of microbial sulfur and ferrous ion oxidation is the limiting step governing the efficiency of bioleaching process, and it has been reported that long time length (6–12 days) is usually required to obtain high solubilization efficiency and thus makes this process non-economically viable11,12.
Fenton and Fenton-like reaction as advanced oxidation processes have been widely applied to conditioning sludge13,14. Powerful oxidizing agent hydroxyl radical can be produced under acidic condition through the reaction of hydrogen peroxide (H2O2) catalyzed by ferrous (Fe2+) or ferric (Fe3+) ions. Previous studies indicated that Fenton oxidation could improve sludge dewatering by degrading extracellular polymeric substances (EPS) and releasing bound water, and enhance anaerobic biodegradability of the sewage sludge13,14,15. Fenton oxidation was also reported to efficiently release heavy metals from sewage sludge16. However, due to the high buffering capacity of the sewage sludge, large amounts of inorganic acid are required to achieve the desired acidic condition for Fenton reaction, leading to high operational cost. During the bioleaching process, the pH value of the bioleached sludge could decline to an optimal value for Fenton oxidation, and ferrous ion added as substrate for iron-oxidizing bacteria17 could also act as catalyst for Fenton reaction. The subsequent Fenton reaction after bioleaching can accelerate the heavy metals dissolution and sludge dewatering, and thus shorten the operating period for leaching. However, studies on the combination of bioleaching and Fenton reaction are yet limited. Fontmorin and Sillanpaaand investigated the efficiency of combined bioleaching/Fenton-like process for heavy metals removal and dewaterability improvement of the sludge10. Zhu et al. obtained high removal efficiency of Cu, Zn, Pb and Cd after 5-day bioleaching and subsequent Fenton like treatment18. However, little attention was paid to the change of chemical speciation of heavy metals. According to BCR extraction procedure19, the chemical speciation of metals could be classified as exchangeable/acid soluble, reducible, oxidizable and residual forms. The negative impact of heavy metals in sludge is largely determined by their chemical speciation and distributions20,21,22. However, the chemical form distributions of heavy metals in sewage sludge during the combined process of bioleaching and Fenton oxidation are still poorly understood. In addition, the efficiency of Fenton oxidation is mainly depended on the reaction condition, therefore, it is also crucial to investigate the influence of reaction pH and H2O2 dosage in the combined bioleaching/Fenton-like process, which has also been neglected in previous studies.
In this study, the single bioleaching and combined bioleaching/Fenton-like processes were applied to remove heavy metals (Zn, Cu, Cd, Cr, Mn, Ni, As and Pb) from sewage sludge. The solubilization efficiency, chemical speciation changes of heavy metals, and dewaterability of sludge during these two processes were compared. The effect of H2O2 dose and reaction pH during Fenton-like process was also analyzed.
Materials and methods
Sludge samples
The sludge samples were collected from a local municipal sludge treatment plant in Tianjin, China, and then stored at 4 °C for further use. Prior to the bioleaching experiments, total solid (TS) of the raw sludge was adjusted to 4.0% with deionized water. Total concentrations of the heavy metals in the raw sludge and the control standards of pollutants in sludge for agricultural use of China (National Standard GB 4284-2018) are listed in Table 1.
Enrichment of indigenous iron-oxidizing bacteria
The mixed culture of iron-oxidizing bacteria was used as inoculum for the bioleaching experiments, and the enrichment culture was carried out following the process described in detail in our previous study23. Fresh sludge sample from the thickening tank of a local municipal wastewater treatment plant was used as the seed sludge for enrichment culture of indigenous iron-oxidizing bacteria24.
Bioleaching experiments
The procedures of bioleaching experiments were proceeded according to the process described in our previous study23. 300 mL of sludge sample was mixed with 5% (v/v) inocula and 4.00 g/L Fe2+ (FeSO4) as iron substrate. The conical flasks were placed in water bath shakers set at 150 rpm and 28 °C. The pH and oxidation–reduction potential (ORP) of the leaching sludge were monitored over time. 20 mL of the sludge samples was taken out every 48 h for heavy metals analysis.
Combined bioleaching/Fenton-like experiments
The combination of bioleaching and Fenton-like reaction consisted of two steps. The bioleaching step was carried out following the procedures described in “Bioleaching experiments” section. When the pH of the leaching sludge dropped to 4.5, 4.0 and 3.0, the Fenton-like reaction step was performed by adding H2O2 into the bioleached sludge with dosage of 5.0, 8.0, 11.0, 13.0 and 15.0 g/L.
All experiments were carried out in triplicate.
Analysis
The pH, ORP and TS contents of the sludge were determined according to the standard methods25. Capillary suction time (CST) was measured by CST analyzer (304M, Triton).
The total concentrations of heavy metals were measured using inductively coupled plasma mass spectrometry (ICP-MS, Agilent 7700), and the sample pretreatment procedure was proceeded according to our previous study23. The chemical forms of the heavy metals investigated were analyzed using the improved BCR procedures described by Rauret et al.19.
Results and discussion
Bioleaching process
Variation of pH and ORP during bioleaching process
pH and ORP of the sludge are widely known to be the important parameters influencing heavy metal solubilization during bioleaching process, as well as the activity of iron-oxidizing microorganisms10,26,27. The variation of sludge pH and ORP during the single bioleaching process is presented in Fig. 1.
An appropriate pH could enhance the activities of microbes, affecting the release of metals and the stability of metal ions in the liquid phase5. As shown in Fig. 1, the pH value of sewage sludge quickly decreased from 6.44 to 3.07 in the first 6 days, due to the oxidation of Fe2+ and metal sulfides, the production of sulfuric acid, ferric hydroxide and jarosite from the hydrolysis of Fe3+18. Then the pH gradually decreased to 2.89 on the 10th day. The change of ORP followed an opposite trend. ORP value of the sludge rapidly increased from − 155.6 mV to 480.0 mV in the first 6 days, then to 505.0 mV in the following 4 days, due to the oxidation of Fe2+ to Fe3+ by leaching microorganisms.
Heavy metals solubilization and chemical speciation distribution during bioleaching process
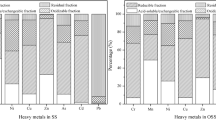
The removal of heavy metals during bioleaching process and the distribution of chemical fractions of heavy metals before and after bioleaching are presented in Figs. 2 and 3, respectively. The single bioleaching led to the removal of Zn, Cu, Cd, Cr, Mn, Ni, As and Pb of 67.28%, 50.78%, 64.86%, 6.32%, 56.15%, 49.83%, 20.78% and 10.52% in 10 days, respectively. The solubilization efficiency was highly related to the evolution of pH and ORP, the chemical fraction distributions and the nature of heavy metals.
Figure 2 illustrated that Zn had the highest solubilization and removal efficiency. It was found that below the threshold pH of 6–6.5, Zn could be dissolved28. Thus, the dissolving out of Zn had started at the beginning of leaching experiment with a removal percentage of 10.15% on the 2nd day. Yet the quick solubilization of Zn was observed from the 4th day (pH 4.01). And until the 6th day (pH 3.00) when the solubilization percentage of Zn was 65.71%, the leaching rate of Zn was slowed down due to the stable pH. In the raw sludge, Zn mainly existed in mobile forms (exchangeable/acid soluble and reducible forms) as shown in Fig. 3. After bioleaching, the solubilization efficiencies of Zn in exchangeable/acid soluble form and reducible form was 58.66% and 87.93%, respectively. Meanwhile, 48.27% of Zn in oxidizable form was also dissolved out due to the oxidation of metal sulfide and loss of sludge organic matter. However, Zn in residual form remained almost unchanged in the bioleached sludge due to its high stability.
It has been pointed out that Cu could be rapidly solubilized below pH of 3.7 or under a high ORP condition29. As shown in Fig. 2, in the first 4 days, the solubilization efficiency of Cu was relatively low (11.44%). The removal rate of Cu increased rapidly to 43.54% on the 6th day due to the increase of ORP (480 mV). The proportion of Cu in exchangeable/acid soluble form increased by 55.16% after bioleaching, probably because the solubilized Cu2+ was re-adsorbed on the EPS of sludge cells30,31. Most of Cu was present in reducible and oxidizable forms in the raw sludge as shown in Fig. 3, because the complexation of copper and organic materials was relatively stable30,32,33. The removal percentages of Cu in reducible and oxidizable forms were 71.11% and 61.83% after bioleaching, respectively, which was the main reason for Cu removal.
Cd could be solubilized rapidly under acidic conditions as shown in Fig. 2, which is consistent with the previous study34. The solubilization of Cd could be finished in 6 days with the removal rate of 64.36%. Cd was mainly present in mobile forms (91.07%) as shown in Fig. 3, which agreed with the findings of Zeng et al.35 Thus, the acid dissolution was the main removal mechanism of Cd34. Due to the low pH of the bioleached sludge, the content of Cd in mobile forms decreased by 62.77% after bioleaching. Furthermore, Cd in immobile forms (oxidizable and residual forms) also reduced significantly.
The previous study found that Cr was relatively stable with the dissolved pH threshold of 2.3–3.028. Although the percentage of Cr present in mobile forms was over 40%, the removal rate of Cr (6.32%) was the lowest among all the heavy metals investigated as shown in Fig. 2, because the lowest pH of the bioleached sludge was about 2.9, which was close to the dissolution threshold limit of Cr.
As shown in Fig. 2, Mn and Ni were solubilized quickly in the first 4 days. The solubilization percentage of Mn and Ni were 56.14% and 49.83% after bioleaching, respectively. Mn and Ni mainly existed in the mobile forms (Mn 82.05%, Ni 76.08%). In the early stage of bioleaching, the removal rates of Mn and Ni were closely related to the variation of pH and displayed obvious acid dissolution mechanism. After bioleaching, the concentrations of Mn in exchangeable/acid soluble, reducible and oxidizable forms were reduced by 34.65%, 78.82% and 90.84%, respectively. As for Ni, the removal rates in such forms were 34.66%, 74.58% and 64.99%, respectively. Thus, the higher extraction efficiency of Mn and Ni arose from mixed bioleaching mechanisms, which contain acid dissolution, oxidation and reduction by Fe2+/Fe3+.
Relatively low removal efficiency of As (20.78%) was observed in this study. One reason, as shown in Fig. 3, was that As was mainly distributed in residual form with high stability. The other reason was that the dissolved As3+ could be oxidized to As5+ (AsO43-) by Fe3+ generated from the metabolism of iron-oxidizing bacteria, and then insoluble FeAsO4 could be produced through the reaction of AsO43- and Fe3+, which resulted in the reprecipitation of As34.
Pb in exchangeable/acid soluble form was not detected in the raw sludge, and mainly existed in reducible (59.20%) and oxidizable (23.19%) forms. The removal rates of Pb in reducible and oxidizable forms were 33.51% and 58.17% after bioleaching, respectively. However, the insoluble compounds such as PbSO4 (Ksp = 1.62 × 10–8) could be generated during the bioleaching process36, which resulted in a significant increase in the concentration of Pb in residual form (from 10.89 to 25.00 mg/kg), and thus led to the low removal ratio of Pb (10.52%).
To summarize, the solubilization efficiencies of Zn, Cu, Cd, Mn and Ni, which mainly existed in mobile forms in the raw sludge, were relatively high due to the instability of these metals, while the removal rates of Cr, As and Pb, which mainly existed in immobile forms, were relatively low. However, the contents of most heavy metals in mobile forms decreased obviously after bioleaching and would lead to the corresponding reduction of the environmental risk of the sludge.
Combined bioleaching/Fenton-like process
Effect of H2O2 dosage on the removal of heavy metals under various pH conditions
Previous studies have shown that the production ability of hydroxyl radical during the Fenton-like reaction process could be enhanced under pH range of 2.5–4.5, and meanwhile, the amount of H2O2 directly influences the production of hydroxyl radical10,18. Therefore, as shown in Fig. 4, the effects of H2O2 dosage on the solubilization efficiencies of heavy metals were investigated at different stages of the bioleaching process, when the pH values of the bioleached sludge were 4.5 (about 3.5th day), 4.0 (4th day) and 3.0 (6th day).
With the increasing concentrations of H2O2 (0.0–8.0 g/L), the solubilization efficiency of Zn increased significantly at pH of 4.5 (Fig. 4) due to the oxidation of metal sulfide and organics by hydroxyl radical10. However, the solubilization percentages of Zn barely changed with further increase of H2O2 dosage (from 8.0 to 15.0 g/L). The solubilization percentage of Zn at the H2O2 dosage of 8.0 g/L (pH of 4.5) was significantly higher than when only using single bioleaching (75.31% vs. 67.64%). The enhancement of solubilization efficiency of Zn at a pH of 4.0 and 3.0 was not very noticeable (Fig. 4), because most of the Zn in immobile forms was dissolved out by bioleaching. The highest solubilization percentages of Zn were 74.96% at a pH of 4.0 and 75.53% at a pH of 3.0, which were 7.32% and 7.89% higher than that of the single bioleaching process.
Due to the lower dissolved pH threshold of Cu compared with Zn, the solubilization efficiency of Cu was significantly affected by the dosage of H2O2 at a pH of 4.5 and 4.0 as shown in Fig. 4, while when the reaction pH was 3.0, the subsequent Fenton treatment had a relatively small impact on the removal of Cu. The highest removal rate of Cu (52.17%) was obtained at pH of 3.0 and H2O2 dosage of 13.0 g/L, which was slightly higher than that of the single bioleaching (50.78%). The change in solubilization efficiency of Cd was similar to that of Cu. When the pH values were 4.5 and 4.0, the solubilization percentages of Cd with H2O2 dosage of 15.0 g/L were 4.59% and 1.23% higher than that of the single bioleaching process, respectively. Meanwhile, the highest solubilization percentage of Cd (71.91%) could be reached at a pH of 3.0 and H2O2 dosage of 13.0 g/L, which was higher than that of the single bioleaching process (64.86%).
The addition of H2O2 did not increase the removal rate of Cr significantly as shown in Fig. 4. At a reaction pH of 4.5, the solubilization percentage of Cr was 7.59% with H2O2 dosage of 15.0 g/L, which was a little higher than that of the single bioleaching process (6.32%), while the highest solubilization percentages of Cr could reach 11.63% and 9.18% at pH of 4.0 and 3.0, respectively, with H2O2 dosage of 15.0 g/L.
The solubilization process of Mn and Ni displayed similar trend as shown in Fig. 4. The solubilization percentage of Mn was not significantly improved when the H2O2 dosage was increased from 5.0 to 11.0 g/L at pH of 4.5 and 4.0, but a much faster increase of the removal rate was observed with the H2O2 dosage over 13.0 g/L. It could be due to the enhanced oxidizing ability of Fenton-like reaction with abundant H2O2. However, the solubilization efficiency of Mn under a pH of 3.0 began to increase with H2O2 concentration of 11.0 g/L, which could be attributed to the high efficiency of Fenton action under lower pH15. The highest removal percentage of Mn was 66.29% at pH of 3.0 and H2O2 dosage of 15.0 g/L, while the removal percentage of Mn in the single bioleaching process was 56.14%. The removal behavior of Ni at various pH was consistent with Mn. The highest removal rate of Ni (65.81%) was found at a pH of 3.0 with H2O2 dosage of 15.0 g/L, which was significantly improved, compared with the single bioleaching process (49.83%).
On the contrary, the removal efficiency of As and Pb in the combined process was not promoted compared with the single bioleaching process. Due to the strong oxidizing capacity of Fenton-like process, the yield of SO42− and insoluble FeAsO4 could be improved. Correspondingly, Pb2+ could be transformed into residual form, such as insoluble PbSO410. Therefore, the removal efficiencies of As and Pb decreased in the combined process. The highest removal rates of As and Pb after Fenton-like treatment were 12.46% and 10.20%, respectively.
In the combined process, higher solubilization efficiencies of most heavy metals (Zn, Cu, Cd, Mn, Ni, Cr) could be achieved in 6 days. The removal efficiency of heavy metals (except Cr, As and Pb) of combined process (pH of 3.0, H2O2 dosage of 15 g/L) is higher than that of the single bioleaching process. The removal rate of Zn, Cu, Cd, Mn and Ni increased by 7.89%, 0.38%, 5.56%, 10.15% and 15.35%, respectively. Meanwhile, the total concentrations of heavy metals measured in this study after treatment could meet the control standards of pollutants in sludge for agricultural use of China (National Standard GB 4284-2018). The removal of As and Pb was not improved by the combined process, other methods such as chemical leaching, electrokinetic remediation and phytoremediation could be considered as alternatives. However, their transformation into insoluble forms may also reduce the bioavailability of heavy metals and increase the environmental safety of the treated sludge. For that reason, the chemical speciation distributions of heavy metals in the combined process were further analyzed in detail.
Chemical fraction distributions of heavy metals in the combined process
It can be seen in Fig. 4 that the solubilization efficiency of most heavy metals did not change significantly with H2O2 dosage below 8.0 g/L. Therefore, the chemical speciation changes of heavy metals after Fenton treatment under H2O2 dosage of 11.0, 13.0 and 15.0 g/L, as shown in Fig. 5, were discussed.
Under various pH conditions, the contents of Zn in all of the four forms showed a downward trend along with the increasing H2O2 dosage (Fig. 5). After bioleaching, Zn mainly existed in exchangeable/acid soluble form under the final pH of 4.5 (64.89%), pH of 4.0 (73.33%) and pH of 3.0 (80.82%). The removal of Zn in exchangeable/acid soluble form showed good correlation to the dosage of H2O2, which might be attributed to the destruction of EPS, and the released heavy metals were transferred to the liquid phase. Meanwhile, the improvement of sludge dewaterability could also promote the removal of heavy metals. After Fenton-like reaction at a pH of 4.5, the percentages of Zn in exchangeable/acid soluble forms were reduced by 30.35%, 31.41% and 40.09% at H2O2 dosage of 11.0, 13.0 and 15.0 g/L, respectively, compared with the percentage of Zn in the sludge at the end of the single bioleaching process. However, the percentage of Zn in other forms did not change significantly after Fenton-like treatment. Therefore, the further removal of Zn in exchangeable/acid soluble form and the dewaterability improvement of sludge may be the main reasons for the higher removal efficiency of Zn in the combined process.
Cu was still mainly associated with the oxidizable form after bioleaching ended at pH of 4.5, 4.0 and 3.0 (Fig. 5), which might be attributed to the preference of Cu for organic materials22. The addition of H2O2 at pH 4.5 significantly boosted the solubilization efficiency of Cu in exchangeable/acid soluble form. The percentages of Cu in exchangeable/acid soluble form in the sludge after Fenton treatment at pH 4.5 were 24.69% (11.0 g/L), 29.50% (13.0 g/L) and 38.15% (15.0 g/L), which were lower than that at the end of the single bioleaching process. Meanwhile, the content of Cu in reducible form was reduced by nearly 50% with H2O2 dosage of 13.0 and 15.0 g/L, compared with its content after bioleaching ended at pH 4.5. However, the highest removal rate of Cu in oxidizable form was only 33.20% with H2O2 dosage of 15.0 g/L. The removal efficiency of Cu in exchangeable/acid soluble and reducible forms increased with the increasing H2O2 dosage at pH 4.0 and 3.0, similar to the observation at pH 4.5. Under a reaction pH of 4.0, 47.2% of Cu in oxidizable form was removed after Fenton treatment with H2O2 dosage of 13.0 g/L, while only 28.6% was removed at H2O2 dosage of 15.0 g/L. In addition, the removal rates of Cu in oxidizable form were only 4.9–17.7% at various H2O2 dosage at a Fenton reaction pH of 3.0. The removal efficiency of Cu was reduced in despite of the increasing oxidation capacity of Fenton-like reaction. The macro-molecular organic matters could be degraded into small organic molecules during Fenton treatment process, releasing partial Cu. However, the generated small molecule organic matters had more undissociated carboxyl that would combine with released Cu31, which formed Cu in oxidizable form. Thus, it could explain the low removal efficiency of Cu in oxidizable form under stronger oxidizing condition. However, the highest removal rate of Cu (52.17%) was observed at pH 3.0 and H2O2 dosage of 15.0 g/L, due to the high reduction ratio of Cu in mobile forms at that condition.
Cd mainly existed in mobile forms in the sludge after bioleaching and Fenton treatment, as shown in Fig. 5. The contents of Cd in mobile and oxidizable forms decreased with the increasing H2O2 dosage at pH 4.5. The content of Cd in exchangeable/acid soluble form after Fenton treatment at pH 4.5 and H2O2 dosage of 15.0 g/L was 29.10% lower than that at the end of the single bioleaching process. Meanwhile, the content of Cd in mobile form was decreased by 27.54% (11.0 g/L), 26.56% (13.0 g/L) and 36.72% (15.0 g/L) after Fenton treatment at pH 4.0. The removal of Cd in exchangeable/acid soluble form after Fenton treatment could be largely due to the improvement of sludge dewaterability. However, the reduction of Cd was not obvious after Fenton treatment at pH 3.0, because the solubilization threshold of most of Cd in various forms were reached after the bioleaching process ended at pH 3.0.
The removal efficiency of Cr was not improved obviously by Fenton treatment in this study, as shown in Fig. 5. It was also reported that Cr was difficult to be removed by bioleaching or combined process due to its relatively high stability10. However, the content of Cr in oxidizable form after Fenton treatment at pH 4.5 was 4.76% (11.0 g/L), 9.20% (13.0 g/L) and 9.84% (15.0 g/L) lower than that at the end of the single bioleaching process, due to the strong oxidizing capacity of hydroxyl radical. And the lowest content of Cr in oxidizable form was observed after Fenton treatment at pH 4.0 and H2O2 dosages of 13.0 g/L, which was 39.4% lower than that in the bioleached sludge. Meanwhile, the highest Cr removal rate was also obtained at this condition after Fenton-like treatment. Thus, the improvement of Cr removal in combined process was mainly due to the release of Cr in oxidizable form. Furthermore, the released metals could be absorbed on the surface of oxides31, thus inevitably caused the increase of Cr in reducible form as shown in Fig. 5. The chemical speciation change of Cr after Fenton treatment at pH 3.0 was similar to that at pH 4.0.
The removal efficiency and chemical speciation distribution of Mn varied obviously after Fenton treatment with different dosages of H2O2. The removal rate of Mn was improved with the increasing dosage of H2O2 at various pH values. Because most of the Mn in reducible form (over 80%) was removed by bioleaching process, the reduction of Mn in exchangeable/acid soluble form should account for the removal of a substantial part of Mn after Fenton treatment. The highest removal rate of Mn in exchangeable/acid soluble form under different pH conditions was 26.27% (pH 4.5), 25.06% (pH 4.0) and 42.18% (pH 3.0), all with H2O2 dosage of 15.0 g/L. Although nearly 30% of Mn in reducible and oxidizable forms was also removed after Fenton treatment with H2O2 dosage of 15.0 g/L at various pH values, it contributed little to the removal of Mn considering the low concentration of Mn in reducible and oxidizable forms in the raw sludge. Furthermore, the changes of Mn in residual form were not obvious under different pH.
The chemical speciation change of Ni was similar to that of Mn after Fenton treatment. The contents of Ni in mobile and oxidizable forms decreased along with the increasing dosage of H2O2, as shown in Fig. 5. Meanwhile, the reduction of Ni in exchangeable/acid soluble form after the addition of H2O2 was the prime reason for the higher removal efficiency of Ni after the combined process than that after the single bioleaching process. The highest removal rate of Ni in exchangeable/acid soluble form was found with H2O2 dosage of 15.0 g/L at pH 4.0, which was 34.47% lower than that in the sludge after the signal bioleaching process. However, the highest removal efficiency of Ni (65.19%) was reached when the reaction pH was 3.0 with H2O2 dosages of 15.0 g/L due to the simultaneous reduction of Ni in reducible and oxidizable forms. The contents of Ni in reducible and oxidizable forms were reduced by 50.30% and 52.83% under this reaction condition, respectively, compared with that at the end of the single bioleaching process.
As and Pb were mainly present in residual form before Fenton treatment as shown in Fig. 5. The content of As in exchangeable/acid soluble form decreased significantly due to the degradation of EPS at various pH values with the addition of H2O2. However, the content of As in residual form gradually rose with the increasing dosage of H2O2, probably because As3+ could be oxidized to As5+ by hydroxyl radical and/or Fe3+ with the formation of insoluble FeAsO434. The content of Pb in reducible form showed a trend of increase after Fenton treatment. SO42− was generated due to the oxidation of sulfur elements and/or sulfide in sludge by hydroxyl radicals with the production of insoluble PbSO410, and thus the content of Pb in residual form also increased after further Fenton treatment. Although the Fenton treatment had a negative impact on the removal of As and Pb as shown in Fig. 5, because of the formation of insoluble compounds under strong oxidizing condition, the environmental risk of these two heavy metals decreased to some extent under an appropriate condition, due to the increased proportion of immobile fractions, especially residual form. compared with the bioleached sludge.
The content and proportion of most heavy metals (Zn, Cu, Cd, Mn, Ni, As) in mobile forms were lower in the treated sludge after the combined bioleaching and Fenton-like process, compared with the single bioleaching process, which was also the main reason for the high removal efficiency of these metals. Their bioavailability and toxicity were also reduced. However, Fenton treatment was found to have a negative impact on the removal of As, but the increased proportion of As in residual form also lowered its bioavailability and mobility in the environment. The increase in the content of Pb in both mobile forms (mainly in reducible form) and immobile forms (mainly in residual form) was observed under different conditions, so special attention should be paid to the chemical speciation distributions of Pb during sludge treatment process.
The effect of H2O2 dosage on sludge dewaterability at different pH values
The changes of CST of treated sludge under various conditions are presented in Fig. 6. The CST of the raw sludge (98.7 s) was dramatically reduced by bioleaching and Fenton oxidation treatments. After bioleaching ended on the 10th day (pH 2.89), the 6th day (pH 3.0), the 4th day (4.0) and the 3.5th day (pH 4.5), CST values of 20.3 s, 24.2 s, 30.7 s and 35.0 s were observed. The decreased pH after bioleaching process could destroy the EPS and neutralize the negative charge of the sludge flocs, resulting in the release of bound water37. Moreover, sludge dewatering could also be improved by the coagulation effect of Fe2+ 10. Furthermore, hydroxyl radicals were essential to improve sludge dewatering performance by destroying EPS and porous structure during the Fenton treatment process35. Therefore, the CST value of treated sludge was reduced to 20.6 s after Fenton treatment with H2O2 dosage of 15 g/L at pH 4.5, which was comparable to the CST value at the end of the single bioleaching process. The CST values were further reduced along with the decreasing reaction pH (4.0 and 3.0) and the increasing H2O2 dosage. The lowest CST value of 12.4 s was observed at Fenton reaction pH 3.0 and H2O2 dosage of 15.0 g/L, which meant a reduction from the initial CST of 87.44%. Therefore, the combined process could lead to an obvious improvement of the sludge dewaterability and significantly reduced the treatment period.
Conclusion
Zn, Cu, Cd, Cr, Mn, Ni, As and Pb could be leached and removed to some extent after a 10-day bioleaching process. The contents of heavy metals in oxidizable form were all decreased. And the proportions of most heavy metals (except As) in mobile forms were also reduced after bioleaching. The pH value of the bioleached sludge could satisfy the requirement of Fenton-like process. The removal efficiencies of most heavy metals investigated (except As and Pb) in the combined process were improved under appropriate pH and H2O2 dosage, compared with that of the single bioleaching, due to the further removal of these heavy metals in mobile forms and oxidizable form after Fenton-like treatment. The combined bioleaching Fenton-like process could also significantly reduce the treatment time. However, the transformation of As and Pb into residual forms during Fenton treatment process had negative effects on their removal after the combined process. The reaction pH of Fenton and H2O2 dosage had obvious influence on the removal and chemical form transformation of the heavy metals. Meanwhile, the combined process also led to a significant improvement in the dewaterability of the treated sludge.
References
Youssef, N. H., Al-Huqail, A. A., Ali, H. M., Abdelsalam, N. R. & Sabra, M. A. The role of Serendipita indica and Lactobacilli mixtures on mitigating mycotoxins and heavy metals’ risks of contaminated sewage sludge and its composts. Sci. Rep-UK 10(1), 15159. https://doi.org/10.1038/s41598-020-71917-8 (2020).
Qi, G. X. et al. Leaching behavior and potential ecological risk of heavy metals in Southwestern China soils applied with sewage sludge compost under acid precipitation based on lysimeter trials. Chemosphere 126, 126212. https://doi.org/10.1016/j.chemosphere.2020.126212 (2020).
Sharma, B., Sarkar, A., Singh, P. & Singh, R. P. Agricultural utilization of biosolids: A review on potential effects on soil and plant grown. Waste Manag. 64, 117–132. https://doi.org/10.1016/j.wasman.2017.03.002 (2017).
Chen, B. W. et al. A unique Pb-binding flagellin as an effective remediation tool for Pb contamination in aquatic environment. J. Hazard. Mater. 363, 34–40. https://doi.org/10.1016/j.jhazmat.2018.10.004 (2019).
Gu, T. Y., Rastegar, S. O., Mousavi, S. M., Li, M. & Zhou, M. H. Advances in bioleaching for recovery of metals and bioremediation of fuel ash and sewage sludge. Bioresour. Technol. 261, 428–440. https://doi.org/10.1016/j.biortech.2018.04.033 (2018).
Du, F. Z. et al. Nitrite addition to acidified sludge significantly improves digestibility, toxic metal removal, dewaterability and pathogen reduction. Sci. Rep-UK 6, 39795. https://doi.org/10.1038/srep39795 (2016).
Yesil, H. & Tugtas, A. E. Removal of heavy metals from leaching effluents of sewage sludge via supported liquid membranes. Sci. Total Environ. 693, 133608. https://doi.org/10.1016/j.scitotenv.2019.133608 (2019).
Fonti, V., Dell’Anno, A. & Beolchini, F. Influence of biogeochemical interactions on metal bioleaching performance in contaminated marine sediment. Water Res. 47(14), 5139–5152. https://doi.org/10.1016/j.watres.2013.05.052 (2013).
Mehrotra, A., Kundu, K. & Sreekrishnan, T. R. Decontamination of heavy metal laden sewage sludge with simultaneous solids reduction using thermophilic sulfur and ferrous oxidizing species. J. Environ. Manag. 167, 228–235. https://doi.org/10.1016/j.jenvman.2015.11.004 (2016).
Fontmorin, J. M. & Sillanpaa, M. Bioleaching and combined bioleaching/Fenton-like processes for the treatment of urban anaerobically digested sludge: Removal of heavy metals and improvement of the sludge dewaterability. Sep. Purif. Technol. 156, 655–664. https://doi.org/10.1016/j.seppur.2015.10.061 (2015).
Akcil, A., Erust, C., Ozdemiroglu, S., Fonti, V. & Beolchini, F. A review of approaches and techniques used in aquatic contaminated sediments: Metal removal and stabilization by chemical and biotechnological processes. J. Clean. Prod. 86, 24–36. https://doi.org/10.1016/j.jclepro.2014.08.009 (2015).
Pathak, A., Morrison, L. & Healy, M. G. Catalytic potential of selected metal ions for bioleaching, and potential techno-economic and environmental issues: A critical review. Bioresour. Technol. 229, 211–221. https://doi.org/10.1016/j.biortech.2017.01.001 (2017).
Neyens, E., Baeyens, J., Dewil, R. & De heyder, B. Advanced sludge treatment affects extracellular polymeric substances to improve activated sludge dewatering. J. Hazard. Mater. 106(2-3), 83–92. https://doi.org/10.1016/j.jhazmat.2003.11.014 (2004).
Pilli, S., Yan, S., Tyagi, R. D. & Surampalli, R. Y. Overview of Fenton pre-treatment of sludge aiming to enhance anaerobic digestion. Rev. Environ. Sci. Biol. 14(3), 453–472. https://doi.org/10.1007/s11157-015-9368-4 (2015).
Babuponnusami, A. & Muthukumar, K. A review on Fenton and improvements to the Fenton process for wastewater treatment. J. Environ. Chem. Eng. 2(1), 557–572. https://doi.org/10.1016/j.jece.2013.10.011 (2014).
Fontmorin, J. M. & Sillanpaa, M. Dewatering and removal of metals from urban anaerobically digested sludge by Fenton’s oxidation. Environ. Technol. 38(4), 495–505. https://doi.org/10.1080/09593330.2016.1199598 (2016).
Pathak, A., Dastidar, M. G. & Sreekrishnan, T. R. Bioleaching of heavy metals from sewage sludge by indigenous iron-oxidizing microorganisms using ammonium ferrous sulfate and ferrous sulfate as energy sources: A comparative study. J. Hazard. Mater. 171, 273–278. https://doi.org/10.1016/j.jhazmat.2009.05.139 (2009).
Zhu, Y. et al. Feasibility of bioleaching combined with Fenton-like reaction to remove heavy metals from sewage sludge. Bioresour. Technol. 142, 530–534. https://doi.org/10.1016/j.biortech.2013.05.070 (2013).
Rauret, G. et al. Improvement of the BCR three step sequential extraction procedure prior to the certification of new sediment and soil reference materials. J. Environ. Monit. 1, 57–61. https://doi.org/10.1039/a807854h (1999).
Hsieh, C. H., Lo, S. L., Chiueh, P. T., Kuan, W. H. & Chen, C. L. Microwave enhanced stabilization of heavy metal sludge. J. Hazard. Mater. 139(1), 160–166. https://doi.org/10.1016/j.jhazmat.2006.06.019 (2007).
Yuan, C. G., He, B., Gao, E. L., Lu, J. X. & Jiang, G. B. Evaluation of extraction methods for arsenic speciation in polluted soil and rotten ore by HPLC-HG-AFS analysis. Microchim Acta 159, 175–182. https://doi.org/10.1007/s00604-006-0709-4 (2007).
Zhang, J. et al. Distribution and risk assessment of heavy metals in sewage sludge after ozonation. Environ. Sci. Pollut. Res. 24, 5118–5125. https://doi.org/10.1007/s11356-016-6313-1 (2017).
Qiu, C. S. et al. Effect of ozonation treatment on the chemical speciation distributions of heavy metals in sewage sludge and subsequent bioleaching process. Environ. Sci. Pollut. Res. 27(16), 19946–19954. https://doi.org/10.1007/s11356-020-08539-0 (2020).
Wang, J. W., Bai, J. F., Xu, J. Q. & Liang, B. Bioleaching of metals from printed wire boards by Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans and their mixture. J. Hazard. Mater. 172(2–3), 1100–1105. https://doi.org/10.1016/j.jhazmat.2009.07.102 (2009).
APHA. Standard Methods for the Examination of Water and Wastewater, 22nd. (2012).
Liu, Y. G. et al. Bioleaching of heavy metals from mine tailings by indigenous sulfur-oxidizing bacteria: Effects of substrate concentration. Bioresour. Technol. 99(10), 4124–4129. https://doi.org/10.1016/j.biortech.2007.08.064 (2008).
Liu, F. W., Zhou, L. X., Zhou, J., Song, X. W. & Wang, D. Z. Improvement of sludge dewaterability and removal of sludge-borne metals by bioleaching at optimum pH. J. Hazard. Mater. 221–222, 170–177. https://doi.org/10.1016/j.jhazmat.2012.04.028 (2012).
Villar, L. D. & Garcia, O. Solubilization profiles of metal ions from bioleaching of sewage sludge as a function of pH. Biotechnol. Lett. 24(8), 611–614. https://doi.org/10.1023/A:1015010417315 (2002).
Chan, L. C., Gu, X. Y. & Wong, J. W. C. Comparison of bioleaching of heavy metals from sewage sludge using iron- and sulfur-oxidizing bacteria. Adv. Environ. Res. 7(3), 603–607. https://doi.org/10.1016/S1093-0191(02)00050-3 (2003).
Chou, J. D., Wey, M. Y. & Chang, S. H. Evaluation of the distribution patterns of Pb, Cu and Cd from MSWI fly ash during thermal treatment by sequential extraction procedure. J. Hazard. Mater. 162(2–3), 1000–1006. https://doi.org/10.1016/j.jhazmat.2008.05.155 (2009).
Wu, H. M., Li, M., Zhang, L. & Sheng, C. Research on the stability of heavy metals (Cu, Zn) in excess sludge with the pretreatment of thermal hydrolysis. Water Sci. Technol. 73(4), 890–898. https://doi.org/10.2166/wst.2015.537 (2016).
Li, J. H., Zhang, M., Ye, Z. Y. & Yang, C. M. Effect of manganese oxide-modified biochar addition on methane production and heavy metal speciation during the anaerobic digestion of sewage sludge. J. Environ. Sci. 76, 267–277. https://doi.org/10.1016/j.jes.2018.05.009 (2019).
Xu, Q. Y., Wang, H. D., Wang, Q. D., Zhang, W. J. & Wang, D. S. Characterization of changes in extracellular polymeric substances and heavy metal speciation of waste activated sludge during typical oxidation solubilization processes. J. Environ. Sci. 80, 146–158. https://doi.org/10.1016/j.jes.2018.12.005 (2019).
Wang, J. et al. Bioleaching mechanism of Zn, Pb, In, Ag, Cd and As from Pb/Zn smelting slag by autotrophic bacteria. J. Environ. Manag. 159, 11–17. https://doi.org/10.1016/j.jenvman.2015.05.013 (2015).
Zeng, X. F. et al. Removal of trace metals and improvement of dredged sediment dewaterability by bioleaching combined with Fenton-like reaction. J. Hazard. Mater. 288, 51–59. https://doi.org/10.1016/j.jhazmat.2015.02.017 (2015).
Kim, H. A., Lee, K. Y., Lee, B. T., Kim, S. O. & Kim, K. W. Comparative study of simultaneous removal of As, Cu, and Pb using different combinations of electrokinetics with bioleaching by Acidithiobacillus ferrooxidans. Water Res. 46(17), 5591–5599. https://doi.org/10.1016/j.watres.2012.07.044 (2012).
Wong, J. W. C., Zhou, J., Kurade, M. B. & Murugesan, K. Influence of ferrous ions on extracellular polymeric substances content and sludge dewaterability during bioleaching. Bioresour. Technol. 179, 78–83. https://doi.org/10.1016/j.biortech.2014.10.099 (2015).
Acknowledgements
This work was supported by the Major Science and Technology Program for Water Pollution Control and Treatment of China (2017ZX07106001).
Author information
Authors and Affiliations
Contributions
C.S.Q. designed the experiment, wrote the manuscript and contributed to data analysis. S.Y.X. and K.Q.M. performed the experiment. N.N.L. contributed to the conception of the study and editing of the maunscript. C.C.W., D.W. and S.P.W. contributed to data analysis and manuscript preparation. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Qiu, C., Xie, S., Liu, N. et al. Removal behavior and chemical speciation distributions of heavy metals in sewage sludge during bioleaching and combined bioleaching/Fenton-like processes. Sci Rep 11, 14879 (2021). https://doi.org/10.1038/s41598-021-94216-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-94216-2
- Springer Nature Limited