Abstract
The intensity and the magnitude of saliva cortisol responses were investigated during the first 48 h following birth in newborn dairy calves which underwent normal (eutocic, EUT, n = 88) and difficult (dystocic, DYS, n = 70) calvings. The effects of parity and body condition of the dam, the duration of parturition, the time spent licking the calf, the sex and birth weight of the calf were also analyzed. Neonatal salivary cortisol concentrations were influenced neither by factors related to the dam (parity, body condition) nor the calf (sex, birth weight). The duration of parturition and the time spent licking the calf also had no effect on salivary cortisol levels. Salivary cortisol concentrations increased rapidly after delivery in both groups to reach their peak levels at 45 and 60 min after delivery in EUT and DYS calves, respectively supporting that the birth process means considerable stress for calves and the immediate postnatal period also appears to be stressful for newborn calves. DYS calves exhibited higher salivary cortisol concentrations compared to EUT ones for 0 (P = 0.022), 15 (P = 0.016), 30 (P = 0.007), 45 (P = 0.003), 60 (P = 0.001) and 120 min (P = 0.001), and for 24 h (P = 0.040), respectively. Peak levels of salivary cortisol and the cortisol release into saliva calculated as AUC were higher in DYS than in EUT calves for the 48-h of the sampling period (P = 0.009 and P = 0.003, respectively). The greater magnitude of saliva cortisol levels in DYS calves compared to EUT ones suggest that difficult parturition means severe stress for bovine neonates and salivary cortisol could be an opportunity for non-invasive assessment of stress during the early neonatal period in cattle.
Similar content being viewed by others
Introduction
Bovine parturition is initiated by rising cortisol levels in the fetus that provoke a cascade of endocrine activity in the dam1. This increase in fetal cortisol is a result of increased adrenocorticotropic hormone production by the maturing fetal pituitary caused by fetal stressors such as hypoxia and hypercapnia. However, the process of parturition may also be a stressful event for the fetus, especially during the stage of expulsion, if difficulties at calving occur2.
Acute responses to stressful stimuli include activation of the hypothalamic–pituitary–adrenal (HPA) axis and the autonomic nervous system. Pain biomarkers related to HPA axis are often measured in biological samples (e.g. blood). Plasma cortisol concentrations have been widely used to evaluate the HPA axis activity in painful procedures in calves3 and in mature cattle4,5. However, the process of taking blood samples is accompanied by additional stress, which can affect the test results6. According to human studies, psychobiological mechanisms, which trigger the HPA axis, can be assessed by salivary cortisol concentrations7,8 that reflect unbound (free) cortisol9,10. Saliva samples can be easily taken at fixed time intervals after an imposed stress11 and it is a minimally invasive6 and appropriate method to assess HPA axis reactivity in cattle. Furthermore, salivary cortisol correlates well with plasma cortisol with 012 or with a 10 min time lag13,14.
Although dystocia is a growing problem on dairy farms15, it is not known how it influences the stress level of calves during the first 48 h of life which is the most critical period in terms of survival16. The present paper attempts to look at the effects of dystocia and some calving-related factors (i.e. parity and body condition of the dam, the duration of parturition, the time spent with licking the calf, and the sex and birth weight of the calf) on salivary cortisol as noninvasive measure of HPA activity in newborn calves.
Methods
Experimental farm and animals
All methods and the applied procedures on the animals were performed in accordance with the relevant guidelines and regulations of the Pest County Government Office, Department of Animal Health (Permit Number: PE/EA/1973-6/2016) that approved the study. A total of 168 calvings were enrolled on a large-scale dairy farm in Hungary consisting of 1,200 lactating Holstein–Friesian cows. The farm was visited for an 8-month period in the spring (between February and May) and autumn (September and December) of 2016 to exclude the possible effects of heat stress on the trial results. Cows calved in the prepartum group pen or, if assistance was required, in a separate maternity pen. Calves were removed from the dams 2 h after birth and received colostrum by nipple bottle and then fed four times a day with 1.65 L of fresh-cow colostrum per feeding during the first 48 h of life. Newborns were housed individually until 60 d of age in 1.65 × 1.20 m plastic calf hutches with a 1.60 m2 exercise pen, both bedded with straw.
Observation of calvings
Calvings occurring in the group pen were observed with two day/night outdoor network bullet cameras (Vivotek IP8331, VIVOTEK Inc., Taipei, Taiwan), while individual calvings were observed with two portable video cameras (Legria HF M36, CANON Inc., Japan, Tokyo).
Dystocia (DYS, n = 70) was defined as calving difficulty resulting from prolonged spontaneous calving (> 2 h from the appearance of hooves to delivery) or prolonged or severe assisted extraction by one or more people with considerable force with a calving rope or with a calving jack17. Normal calving (eutocia; EUT, n = 98) was regarded as a combination of ‘no assistance’ and ‘slight assistance’ (where assistance was brief, and traction was slight) by one person17.
The condition of the dam was scored using the 5-point BCS system18 following calving. Sex and birth weight of the calves were also recorded immediately after delivery. Since during stage 1 of labor significant stress was found in dairy cows19, and thus, possibly for newborn calves, the duration of parturition was considered as the time lag between the onset of stage 1 (the onset of calving restlessness) and the completion of stage 2 of labor (delivery). The onset of calving restlessness was determined based on accepted behavioral predictors such as lying down frequency, tail raising and walking20 and was observed by two trained experimenters through the above-mentioned camera system. The time spent licking the calf’s head or body was recorded during the first 2 h following calving according to the recommendation of Jensen21.
Salivary cortisol
Using a synthetic swab (Salivette Cortisol, Sarstedt, Nümbrecht-Rommelsdorf, Germany), saliva samples were taken 0, 15, 30, 45, 60, 120 min, 24 and 48 h after delivery. Without retain of the animals, the swabs were placed loosely onto the tongue of the calf until it was well soaked with saliva. This procedure required up to 10 s, and the animals tolerated saliva samplings well. The swabs were then inserted into Salivette polypropylene tubes, which were placed on ice immediately after sampling and stored at 4 °C until centrifugation (within 10 min after sampling) at 1000 g for 10 min. At least 1.5 ml saliva per sample was obtained and frozen at − 20 °C until analysis. After a further dilution step (1:10) with assay buffer, salivary cortisol concentrations were determined in an aliquot (10 µl) with a competitive cortisol enzyme immunoassay (EIA). For the details of the EIA, including cross-reactions and its application in calves refer to Palme and Möstl22 and Wagner et al.23. Interassay coefficients of variation of high and low concentration pool samples from saliva of the calves of this study were 9.2% and 12.8%, respectively. The detection limit of the assay was 0.02 ng/ml.
Statistical analysis
Statistical analyses were performed in the R–3.3.1 statistical environment and language24. All results are expressed as mean plus SEM values.
Multivariable linear regression models were fit to the data25 for each sampling time point to test the effects of independent variables on salivary cortisol concentrations. Independent variables were parity and BCS of the dam, sex and birth weight of the calf, the duration of parturition, calving ease (dystocia or eutocia), and the time spent licking the calf. Salivary cortisol concentrations were inserted into the models as response (dependent) variables. Log-transformation of saliva cortisol concentrations was applied to satisfy the normality and variance homogeneity assumptions of the models.
Based on the results of the linear models (only calving ease had a significant effect on cortisol levels; see Results section), for reducing the number of statistical comparisons between groups during the 48-h postnatal period, salivary cortisol concentrations of EUT and DYS calves were calculated as area under the curve (AUC) and cortisol responses were compared. The AUC represents both the magnitude and the changes over time of the response26. Response parameters included peak values of salivary cortisol concentrations, baseline (48 h sample), and AUCs that were determined for the first 48 h of life following delivery utilizing a trapezoid method described by Lay et al.27 as follows:
where ‘P’ is salivary cortisol concentration at a given time point, ‘m’ is the time in minutes between the two P values and ‘baseline’ is the mean value of cortisol concentrations in saliva 48 h after delivery. Data were tested for constant variance (Levene’s test) and the Shapiro–Wilk test was used for testing the equality of error variances. Comparisons between EUT and DYS groups for peak salivary cortisol levels and AUCs were made by a Wilcoxon rank-sum test. Significance was set at the level of 0.05 in case of both parameters.
Non-significant variables on salivary cortisol concentrations were compared between EUT and DYS groups with the Welch’s two-sample t test (parity and BCS of the dam, sex and birth weight of the calf, the duration of parturition, and the time spent licking the calf) and with the Pearson’s Chi-squared test (proportions of male and female calves) at the significance level of 0.05 in both cases.
Results
From the 168 calvings, 49, 56 and 63 calves were born to first, second and third parity cows, respectively. Comparison of independent variables between EUT and DYS groups is shown in Table 1. Salivary cortisol concentrations determined within 48 h after delivery were neither influenced by factors related to the dam (parity, body condition score, BCS) nor the calf (sex, birth weight). Although the duration of parturition (range: 1.3–8.2 h) and the time spent linking the calf (5.5–86.5 min) differed significantly between EUT and DYS calves (Table 1) none of these factors influenced salivary cortisol levels.
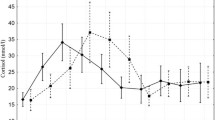
Except for 48 h after delivery, linear models (df = 7; 150) indicated higher salivary cortisol concentrations in DYS calves compared to EUT ones for 0 (P = 0.022), 15 (P = 0.016), 30 (P = 0.007), 45 (P = 0.003), 60 (P = 0.001) and 120 min (P = 0.001), and for 24 h (P = 0.040) after birth, respectively. The evolution of salivary cortisol concentrations after delivery are shown in Fig. 1 for EUT and DYS calves. The HPA response showed a similar pattern in both groups. Salivary cortisol concentrations increased rapidly after delivery in both groups to reach their peak levels at 45 and 60 min after delivery in EUT and DYS calves, respectively. Afterward, a gradual decrease in cortisol concentrations was observed in both groups (Fig. 1). Twenty-four h post-calving, salivary cortisol decreased to 32.9% and 33.7% of the peak levels in EUT and DYS calves, respectively and for the 48-h samples, similar circulating cortisol concentrations were observed in both groups in saliva (P = 0.245).
DYS calves exhibited significantly higher peak levels and AUC of salivary cortisol than EUT calves (with 45.6% and 92.1%, respectively) for the 48-h of the sampling period (Table 2).
Discussion
This is the first study which investigates both the intensity and magnitude of the postnatal HPA response to birth of EUT and DYS dairy calves using AUC analyses based on high sampling frequencies of saliva. The present findings demonstrate that the birth process induces significant elevation in HPA axis activity in newborn calves, even if no difficulties during parturition occur. Differences between cortisol levels measured at 0 and 48 min in EUT (106.4%) and DYS calves (175.2%) found in the present study suggest that calves experienced stress before delivery irrespective of obstetrical conditions. However, calves experiencing DYS births exhibited greater saliva cortisol levels, thus higher stress after calving compared to EUT calves. Our results support earlier findings on serum28, plasma29 and salivary cortisol levels30 of EUT and DYS calves. In general, peak cortisol levels found in saliva in the present study were similar to those observed by Stewart et al.31 in plasma 40 min after administration of adrenocorticotropic hormone (34.5 ng/mL), or after castration without local anesthetic (28.7 ng/mL) in Holstein–Friesian heifer calves32.
An earlier study found lower peak levels of salivary cortisol (14.8 ng/mL) in newborn calves after induced parturitions33, whereas others reported 6 ng/mL concentrations in calves born from assisted deliveries30; however, authors collected saliva once within 24 h of birth, therefore they were not able to determine peak levels.
Gradually increasing cortisol levels after delivery in both groups support that the birth process means considerable stress for calves34 and the immediate postnatal period also appears to be stressful for the newborn calf. In the present study, peak cortisol levels at 45 and 60 min after delivery in EUT and DYS calves, respectively, may be a delayed increase due to cortisol transfer from the serum to the saliva13,14 or even reflect additional stress experienced by the calves during transition from the fetal to the extrauterine life.
Similar to our findings, Nagel et al.33 reported peak saliva cortisol levels at 60 min after birth. Others found peak levels immediately after birth in serum28, and 3 h after delivery in plasma35. Salivary cortisol peak levels found in the present study in DYS calves was 12.2% of those observed by the latter authors in plasma35, which support field12 and laboratory observations on cattle36 indicating that salivary cortisol levels yield around 10% of plasma cortisol levels.
Although Hoyer et al.37 found that reversal of stress occurs rapidly during the first hours of neonatal life, the results presented here suggest that newborn calves appear to adapt to the extrauterine environment by 24 h of age. In line with our results, cortisol concentrations measured from saliva33 and plasma35,38,39 decreased gradually for 24 and 48 h after parturition.
As calving means significant stress also for the dam even from the onset of stage 1 of labor19, it came into question if naturally occurring cortisol in cows before delivery had a significant influence on the amount of cortisol levels of the neonatal calf. It has been shown in goats40 and ewes41 that cortisol can partly cross the placenta, from the mother to the fetus and may lead to hypercortisolism in situations of prolonged stress experienced by the dam during parturition42. However, results of Wooley35 indicate that maternal cortisol concentrations in plasma do not influence calf cortisol concentration in cattle.
It should be noted that within the early neonatal life, other factors might also affect HPA axis functioning. Similar to recent observations33, birth weight and duration of parturition had no effect on neonatal salivary cortisol concentrations. As calves were removed from the dams only 2 h after delivery to receive colostrum by farmhand, the only factor would have been the dam-offspring contact. According to our earlier findings, the duration of licking the calf is a prominent factor in the thermal and metabolic adaptation of newborn calves to extrauterine life43. Although the time spent licking the calf had no effect on salivary cortisol concentrations in this study, it can be assumed that maternal grooming might have caused a positive stress for the calves by increasing cortisol levels between 15 and 60 min after birth, irrespectively for calving ease.
As a progressive maturation and activation of the fetal HPA axis during late gestation results in a considerably increased cortisol release from the fetal adrenals starting between 7 and 3 days before parturition44,45 it is thus questionable whether this initial fetal cortisol would affect cortisol levels measured from saliva after delivery. According to our assumption it could not have been significantly present in the saliva of newborns, as fetal cortisol is proven to be absorbed by the maternal unit causing initiation of the preparation stage of labor46 and the gradual prepartum rise in fetal plasma cortisol during the last week of gestation was found to be much less marked even in spontaneously born calves than the abrupt increase immediately after birth47.
Glucocorticoids can have a significant influence on the amount of immunoglobulins in colostrum and also on the amount of immunoglobulins absorbed by the neonate. Decreased cortisol concentrations may reduce the ability or time available for the calf to absorb colostral immunoglobulins, whereas increased serum cortisol concentrations increase IgG concentrations48. However, it has been proposed that the increased susceptibility to bacterial infection in calves may be enhanced by high plasma cortisol concentrations at birth and their effects upon the lymphocytes49. This is a limitation of the present study that we did not measure immune parameters or followed-up calves to examine the longer-term effects of dystocia-related stress either on growth, behavior, or overall welfare.
The greater magnitude of saliva cortisol responses in DYS calves compared to EUT ones suggest that difficult calving is more stressful for bovine neonates than a normal birth due to prolonged parturition and/or forced extraction, and salivary cortisol could be an opportunity for non-invasive assessment of stress during the early neonatal period in cattle. The findings of the present study should be integrated in further investigations with data from behavioral observations, production, and pathology records in a comprehensive approach of bovine neonatal well-being.
Data availability
All materials, data that support the findings of this study and associated protocols are available from the corresponding author upon reasonable request.
References
Hunter, J. T., Fairclough, R. J., Peterson, A. J. & Welch, R. A. Foetal and maternal hormonal changes preceding normal bovine parturition. Acta Endocrinol. 84, 653–662 (1977).
Kovács, L., Kézér, F. L. & Szenci, O. Effect of calving process on the outcomes of delivery and postpartum health of dairy cows with unassisted and assisted calvings. J. Dairy Sci. 99, 7568–7573 (2016).
Stafford, K. J. & Mellor, D. J. Addressing the pain associated with disbudding and dehorning in cattle. Appl. Anim. Behav. Sci. 135, 226–231 (2011).
Lay, D. C. Jr. et al. Behavioral and physiological effects of freeze or hot-iron branding on crossbred cattle. J. Anim. Sci. 70, 330–336 (1992).
Fidan, A., Pamuk, F. K., Ozdemir, A., Saritas, Z. K. & Tarakci, U. Effects of dehorning by amputation on oxidant-antioxidant status in mature cattle. Rev. Med. Vet. 161, 502–508 (2010).
Cook, N. J. Review: Minimally invasive sampling media and the measurement of corticosteroids as biomarkers of stress in animals. Can. J. Anim. Sci. 92, 227–259 (2012).
Hellhammer, D. H., Wüst, S. & Kudielka, B. M. Salivary cortisol as a biomarker in stress research. Psychoneuroendocrinology 34, 163–171 (2009).
Kudielka, B. M., Hellhammer, D. H. & Wüst, S. Why do we respond so differently? Reviewing determinants of human salivary cortisol responses to challenge. Psychoneuroendocrinology 34, 2–18 (2009).
Kirschbaum, C. Salivary cortisol. In Encyclopedia of Stress (ed. Fink, G.) 379–383 (Academic Press, San Diego, 2000).
Möstl, E. & Palme, R. Hormones as indicators of stress. Domest. Anim. Endocrinol. 23, 67–74 (2002).
Mormède, P. et al. Exploration of the hypothalamic-pituitary-adrenal function as a tool to evaluate animal welfare. Physiol. Behav. 92, 317–339 (2007).
Kovács, L. et al. Hypothalamic–pituitary–adrenal and cardiac autonomic responses to transrectal examination differ with behavioral reactivity in dairy cows. J. Dairy Sci. 99, 7444–7457 (2016).
Negrao, J. A., Porcionato, M. A., de Passille, A. M. & Rushen, J. Cortisol in saliva and plasma of cattle after ACTH administration and milking. J. Dairy Sci. 87, 1713–1718 (2004).
Hernandez, C. E. et al. Time lag between peak concentrations of plasma and salivary cortisol following a stressful procedure in dairy cattle. Acta Vet. Scand. 56, 61 (2014).
Mee, J. F. Newborn dairy calf management. Vet. Clin. N. Am. Food Anim. Pract. 24, 1–17 (2008).
Schuijt, G. Iatrogenic fractures of ribs and vertebrae during delivery in perinataly dying calves: 235 cases (1978–1988). J. Am. Vet. Med. Assoc. 197, 1196–1202 (1990).
Mee, J. F., Berry, D. P. & Cromie, A. R. Risk factors for calving assistance and dystocia in pasture-based Holstein-Friesian heifers and cows in Ireland. Vet. J. 187, 189–194 (2011).
Hady, P. J., Domecq, J. J. & Kaneene, J. B. Frequency and precision of body condition scoring in dairy cattle. J. Dairy Sci. 77, 1543–1547 (1994).
Kovács, L. et al. Heart rate and heart rate variability in multiparous dairy cows with unassisted calvings in the periparturient period. Physiol. Behav. 139, 281–289 (2015).
Miedema, H. M., Cockram, M. S., Dwyer, C. M. & Macrae, A. I. Behavioural predictors of the start of normal and dystocic calving in dairy cows and heifers. Appl. Anim. Behav. Sci. 132, 14–19 (2012).
Jensen, M. B. Behaviour around the time of calving in dairy cows. Appl. Anim. Behav. Sci. 139, 195–202 (2012).
Palme, R. & Möstl, E. Measurement of cortisol metabolites in faeces of sheep as a parameter of cortisol concentration in blood. Z. Saugetierkd. Int. J. Mammal. Biol. 62(Suppl. 2), 192–197 (1997).
Wagner, K. et al. Mother rearing of dairy calves: Reactions to isolation and to confrontation with an unfamiliar conspecific in a new environment. Appl. Anim. Behav. Sci. 147, 43–54 (2013).
R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna. http://www.r-project.org/ (2017).
Pinheiro, J. C. & Bates, D. M. Mixed-Effects Models in S and S-PLUS 1st edn. (Springer, 2000).
Fekedulegn, D. B. et al. Area under the curve and other summary indicators of repeated waking cortisol measurements. Psychosom. Med. 69, 651–659 (2017).
Lay, D. C. Jr. et al. Adrenocorticotropic hormone dose response and some physiological effects of transportation on pregnant Brahman cattle. J. Anim. Sci. 74, 1806–1811 (1996).
Vannucchi, C. I. et al. Association between birth conditions and glucose and cortisol profiles of periparturient dairy cows and neonatal calves. Vet. Rec. 176, 358 (2015).
Civelek, T., Celik, H. A., Avci, G. & Cingi, C. C. Effects of dystocia on plasma cortisol and cholesterol levels in holstein heifers and their newborn calves. Bull. Vet. Inst. Pulawy 52, 649–654 (2008).
Barrier, A. C. et al. The impact of dystocia on dairy calf health, welfare, performance and survival. Vet. J. 195, 86–90 (2013).
Stewart, M., Stafford, K. J., Dowling, S. K., Schaefer, A. L. & Webster, J. R. Eye temperature and heart rate variability of calves disbudded with or without local anaesthetic. Physiol. Behav. 93, 789–797 (2008).
Stewart, M., Verkerk, G. A., Stafford, K. J., Schaefer, A. L. & Webster, J. R. Noninvasive assessment of autonomic activity for evaluation of pain in calves, using surgical castration as a model. J. Dairy Sci. 93, 3602–3609 (2010).
Nagel, C. et al. Stress response and cardiac activity of term and preterm calves in the perinatal period. Theriogenology 86, 1498–1505 (2016).
Aurich, J. E. et al. Influence of labor and neonatal hypoxia on sympathoadrenal activation and methionine enkephalin release in calves. Am. J. Vet. Res. 54, 1333–1338 (1993).
Wooley, D. N. Prepartum maternal cortisol concentrations on postnatal cortisol concentration and immunoglobulin absorption in neonatal dairy calves. LSU Master's Theses. 4124. https://digitalcommons.lsu.edu/gradschool_theses/4124 (2010).
Chacón, G., Laita, S.G.-B., del Portal, J. C. I. & Liesa, J. P. Validation of an EIA technique for the determination of salivary cortisol in cattle. Spanish J. Agric. Res. 2, 45–52 (2004).
Hoyer, C., Grunert, E. & Jochle, W. Plasma glucocorticoid concentrations in calves as an indicator of stress during parturition. Am. J. Vet. Res. 51, 1882–1884 (1990).
Hadorn, U., Hammon, H., Bruckmaier, R. M. & Blum, J. W. Delaying colostrum intake by one day has important effects on metabolic traits and on gastrointestinal and metabolic hormones in neonatal calves. J. Nutr. 127, 2011–2023 (1997).
Hammon, H. M. & Blum, J. W. Metabolic and endocrine traits of neonatal calves are influenced by feeding colostrum for different durations or only milk replacer. J. Nutr. 128, 624–632 (1998).
Thorburn, G. D., Nicol, D. H., Bassett, J. M., Shutt, D. A. & Cox, R. I. Parturition in the goat and sheep: changes in corticosteroids, progesterone, oestrogens and prostaglandin F. J. Reprod. Fert. 16(suppl), 61–84 (1972).
Dixon, R. et al. Feto-maternal transfer and production of cortisol in the sheep. Steroids 16, 771–789 (1970).
Jones, C. T., Robinson, R. O., Luther, E., Ritchie, J. W. K. & Worthington, D. Control of adrenocorticotrophin secretion by catecholamines in the pregnant and foetal sheep. J. Endocr. 73, 11–20 (1977).
Kovács, L., Kézér, F. L., Albert, E., Ruff, F. & Szenci, O. Seasonal and maternal effects on acid-base, lactate, electrolyte and hematological status of 205 dairy calves born to eutocic dams. J. Dairy Sci. 100, 7534–7543 (2017).
Schuler, G., Fürbass, R. & Klisch, K. Placental hormones in gestation and parturition. Anim. Reprod. 15(Suppl. 1), 822–842 (2018).
Taverne, M. A. M., Bevers, M. M., van der Weyden, G. C., Dieleman, S. J. & Fontijne, P. Concentration of growth hormone, prolactin, and cortisol in fetal and maternal blood and amniotic fluid during late pregnancy and parturition in cows with cannulated fetuses. Anim. Reprod. Sci. 17, 51–59 (1988).
Hoffmann, B., Wagner, W. C., Rattenberger, E. & Schmidt, J. Endocrine relationships during late gestation and parturition in the cow. Ciba Found. Symp. 47, 107–125 (1977).
Comline, R. S., Hall, L. W., Lavelle, R. B., Nathanielsz, P. W. & Silver, M. Parturition in the cow: endocrine changes in animals with chronically implanted catheters in the foetal and maternal circulations. J. Endocrinol. 63, 451–472 (1974).
Johnston, N. E. & Oxender, W. D. Effect of altered serum glucocorticoid concentrations on the ability of the newborn calf to absorb colostral immunoglobulin. Am. J. Vet. Res. 40, 32–34 (1979).
Eberhart, R. J. & Patt, J. A. Plasma cortisol concentrations in newborn calves. Am. J. Vet. Res. 32, 1921–1927 (1971).
Acknowledgements
The authors would like to thank Ferenc Bodó, farm owner, and Ágoston Bodó, farm manager for supporting the study, and the farm staff of Prograg Agrárcentrum Ltd. at Ráckeresztúr, Lászlópuszta for taking care of the animals during the experiment. Levente Kovács was supported by the OTKA Research Scholarship of the National Research, Development and Innovation Office (Budapest, Hungary; K-134204) and by the NTP-NFTÖ-20 project by the Human Capacities Grant Management Office and the Hungarian Ministry of Human Capacities, Budapest, Hungary (NTP-NFTÖ-20-B-0030). Thanks to Edith Klobetz-Rassam for cortisol analysis.
Author information
Authors and Affiliations
Contributions
Plan of the experiment: L.K., O.S.Z. Performing the experiment: L.K., F.L.K. Salivary cortisol assessment: R.P. Manuscript writing: L.K. Statistical analysis: F.R. Supervision: S.Z.B., O.S.Z.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kovács, L., Kézér, F.L., Bodó, S. et al. Salivary cortisol as a non-invasive approach to assess stress in dystocic dairy calves. Sci Rep 11, 6200 (2021). https://doi.org/10.1038/s41598-021-85666-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-85666-9
- Springer Nature Limited