Abstract
Acute myeloid leukemia (AML) is the most common hematological malignancy in adults. In the last decade, internationally approved AML treatment guidelines, including hematopoietic stem cell transplantation are widely used in Kazakhstan. The categorization of acute myeloid leukemia was done according to the French-American British classification. The prognosis of patients at the time of diagnosis was determined by cytogenetic tests following the guidelines of the European LeukemiaNet. The overall survival and event-free survival were analyzed using the Kaplan–Meier method, and hazard ratios were defined with Cox regression. In total, 398 patients with AML were treated in the National Research Oncology Center between 2010 and 2020. The mean age was 38.3 years. We found a correlation between ethnicity, cytogenetic group, white blood cell count, and treatment approaches with overall and event-free survival. There was a significantly longer OS in a cytogenetic group with a good prognosis compared with intermediate and poor prognosis. The median survival time in the group with a good prognosis was 43 months, 23 months in the intermediate group (p = 0.7), and 12 months in the poor prognosis group (p = 0.016). There was a significantly longer OS for the group of patients who received hematopoietic stem cell transplantation (HSCT), 52 months versus 10 months in the group who received chemotherapy only, p-value < 0.0001. Prognostic factors, such as cytogenetic group, initial WBC count, and treatment approaches are significantly associated with patient survival. Our study data were consistent with the most recent studies, available in the literature adjusted for the population in question.
Similar content being viewed by others
Introduction
According to the Kazakhstan National Cancer Institute, hematological malignancies represent 4.7% of all cancers, 5th place among both sexes in Kazakhstan. Morbidity due to hematological cancer is in 4th place among all cancer-related deaths in Kazakhstan1. Based on to Globocan data, 759 new cases of leukemia were registered in Kazakhstan in 2020, which corresponds to the 14th place among all cancers, the growth rate comprised 2% compared with 20192.
Acute myeloid leukemia (AML) is the most common acute leukemia in adults3. There are no data regarding the estimated number of death due to AML in Kazakhstan. AML is a group of blood cell cancers originating from hematopoietic precursors in the bone marrow and resulting in a conglomeration of poorly differentiated myeloid cells infiltrating bone marrow, peripheral blood, and other organs4.
Blood disorder develops due to the acquisition of chromosomal translocations and multiple genetic mutations by leukemic stem cells5,6,7. Genetic events are usually associated with environmental influence, other blood disorders, tobacco use, and previous chemotherapy8,9. It is well known that AML incidence increases with age. The median age of people being diagnosed is 65 years10. Various factors were proved to influence the AML incidence and clinical outcome. The incidence increases with age with approximately 2 and 20 cases per 100,000 population for those under and over 65 years, respectively11. The male/female ratio is approximately 5:312.
Currently, AML is the most common indication for hematopoietic stem cell transplantation (HSCT)13. Kazakhstan is the only country in Central Asia, where HSCT is available for patients with AML. Therefore, there are little data regarding AML epidemiology, treatment approaches, and clinical outcomes in Central Asia. Russian Federation is the most geographically close country to Kazakhstan. Bondarenko et al. from a single center in Russian Federation reported that 10-year overall survival after HSCT was 50% among patients with AML14. Here we present a single-center experience of AML treatment outcome in Kazakhstan.
Methods
The data were retrospectively analyzed, and all ethical principles of the Helsinki Declaration have been followed. The Ethical Committee of the National Research Oncology Center (permit number №11) approved the study.
Acute myeloid leukemia classification
The further division of AML into its subtypes was done according to the French-American British (FAB) classification. It sorts AML from M0 to M7, depending on the type of cell where leukemia developed and the maturity of these cells15. Moreover, this categorization takes into account the symptoms that show the whole picture of the patient’s condition.
Molecular genetic stratification of patients by risk groups ELN 2017
Depending on disease pathogenesis, European LeukemiaNet defined three genetic groups for the classification of AML: poor cytogenetic prognosis, intermediate prognosis, and favorable prognosis16. The categorization was based on genetic abnormalities correlated with clinical characteristics. The poor prognosis included translocations t(6;9), t(9;22), inversions inv(3), deletions, mutations of RUNX1, ASXL1, and TP53. As intermediate ones were categorized t(9;11), mutations of NPM1 and FLT3-ITDhight, and anomalies not classified within poor or favorable prognosis. Translocations t(8;21), t(16;16), inversion of chromosome 16, RUNX1-RUNX1T1, and some mutations of NPM1 were categorized as favorable prognosis17,18,19.
Induction therapy
Chemotherapy is the main treatment of AML to reach remission and complete response. The induction therapy was chosen according to the clinical protocol of diagnostics and treatment of AML approved by the Ministry of Health of the Republic of Kazakhstan20. Common procedures include cytarabine for 7 days, which is followed by 3 days of anti-tumor antibiotics. Mostly used anti-tumor antibiotics were daunorubicin (DNR), idarubicin (IDA), and doxorubicin (Doxo). There were other antibiotics used in the frame of chemotherapy, and they were united in one category “others”.
There were different complications after chemotherapy: multiple organ failure syndrome, disseminated intravascular coagulation, anal fissures, sepsis, paraproctitis, pneumonia, Hand-Foot Syndrome, candidiasis, polysinusitis, and others. Most common were febrile neutropenia, sepsis, and aspergillosis.
Indications for HSCT and conditioning regimens
The clear indication for the HSCT was the first remission in the patients who had poor and intermediate cytogenetic prognosis groups, or the second remission in the patients who had intermediate and favorable prognosis according to cytogenetic analysis.
Reduced-intensity conditioning (RIC) regimens were carried out before HSCT according to the Fludarabine + Busulfan scheme (Fludarabin 30 mg/m2/day D-7 to D-2; Busulfan 10 mg/kg D-4, D-3). To carry out allogeneic HSCT, bone marrow, peripheral HSCs, or their combination was used as a source. The average number of HSC-CD 34 cells was 4.5 mln/kg.
Statistical analysis
All data were analyzed with statistical software STATA 14.0. For bivariate analysis, the outcome was taken as the dependent variable, and association with predictors was checked with t-tests and chi-square tests. Kaplan–Meier curves were used to show the overall survival (OS) and event-free survival (EFS) of patients. A Long-rank test was used to access the equality of survival functions, and Cox regression was applied to define the Hazard Ratios. The significance level was set at 0.05.
Ethical approval
The Ethical Committee of the National Research Oncology Center (permit number №11) approved the study.
Results
In total, 398 patients with AML were treated in the National Research Oncology Center between 2010 and 2020. The final study included 371 patients, 27 patients were excluded from the study, patients were excluded if they were lost from follow-up or a history of prior treatment. Patients were predominantly younger than 60 years old (95.1%). The mean age of the study group was 38.3 (± 13) years old. There was no significant difference in the gender of the patients. Clinical-demographical characteristics are shown in Table 1.
There was a statistically significant difference in overall survival (OS) and event-free survival (EFS) related to the patient's age. Median OS (Fig. 1A) of the 17 months for patients younger than 60 years old versus 4 months for the group of patients older than 60 years (p-value 0.0001; HR = 2.6). The median EFS (Fig. 1B) was 14 months for the younger group versus 7 months for the group older than 60 years (p-value 0.0010; HR-2.24).
Kazakhstan is a multinational country, in our cohort study we also compared OS and EFS related to race (Fig. 2). Our data showed that there was a significant difference in OS between the Asian and Caucasians group of patients, 19 and 10 months respectively (p = 0.0001).Our data showed that gender had no significance on the overall survival (Fig. 3A). The event-free survival was significantly related to gender (Fig. 3B). The median EFS in females was 18 months and 14 months in males.
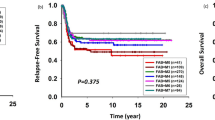
There was a significantly longer OS in a cytogenetic group with a good prognosis compared with intermediate and poor prognosis (Fig. 4). The median survival time in the group with a good prognosis was 43 months, 23 months in the intermediate group (p = 0.7), and 12 months in the poor prognosis group (p = 0.016). A similar significance was observed for EFS (Fig. 5), median survival in the favorable prognosis group was 24 months versus 11 months in the poor prognosis group (p = 0.036).
There was a significantly longer OS for the group of patients who received hematopoietic stem cell transplantation (HSCT), 52 months versus 10 months in the group who received chemotherapy only, p-value < 0.0001 (Fig. 6).
Our analysis showed that OS and EFS in a group of patients with white blood cell (WBC) lower than 30 × 109/L was significantly higher compared with the group with a WBC count of more than 30 × 109/L at the time of diagnosis, 20 months vs 13 months and 17 months vs 10 months respectively (Fig. 7). The median survival time for those, who underwent allogeneic stem cell transplantation was 43 months, while for haploidentical stem cell transplantation patients it was 39 months, and the difference was not statistically significant, p = 0.28 (Fig. 8).
We found that the development of febrile neutropenia after chemotherapy was significantly associated with a negative outcome. In this study, 63.9% of patients died; 67% died due to the disease progression, the second most common cause of death was an infection.
Discussion
The current study reports a 10-year single-center experience of AML treatment in Kazakhstan. The National Research Oncology Center is one of the largest centers in Central Asia. In our study, we analyzed the incidence, death, and other epidemiological data of AML, based on the medical record database of our center during the past 10 years. There is scarce epidemiological data on AML in Kazakhstan.
Although population data says that acute myeloid leukemia occurs mostly in adults21, the mean age of our patients was 38 years. It can be explained by the fact that initially there was a selection bias to perform hematopoietic stem cell transplantation. AML can be cured at younger ages more successfully than for those older than 60 years22. There is no clear cut-off point in literature for age to predict prognosis; however, the separation as younger than 60 and older showed significant results in both overall survival and event-free survival. Older individuals had for 160% more risk of death due to disease (Fig. 1A), and this result is consistent with other research outcomes23.
The data showed significantly better event-free survival for females (HR = 1.38). The trend of higher survival rates among women diagnosed with AML also was previously reported24. However, the cause of this phenomenon is still to be discovered. Moreover, according to our findings, ethnicity was also significantly associated with outcome, where Asians showed approximately two times longer median overall survival time compared to Caucasians. The impact of ethnicity on survival matches with the results of other countries, and such tendency could be explained with different genetic alterations among races25.
Similar to the literature, cytogenetic prognosis and white blood cell count were significantly related to the outcome. Both of these factors are key points in the diagnosis and treatment of AML. Consistent with the literature, the data of the present study shows that poor cytogenetic prognosis according to ELN classification at the time of diagnosis leads to higher mortality rates compared to intermediate and favorable ones26. For WBC count, a level at 30 × 109/L was taken as a cut-off point and showed a significant association between high white blood cell count at the diagnosis time and lower overall and event-free survival of patients. It is consistent with the results of other researchers27,28. The possible explanation is that a high WBC count is associated with the possible progression of leukocytosis, which affects the prognosis of patients29.
The primary induction therapy with IDA showed a significant association with a favorable outcome compared to daunorubicin (DNR) and doxorubicin (Doxo), 52% vs 37% and 31% respectively. Hanyu Wang and colleagues performed a meta-analysis comparing the effect of different therapies on AML patient survival30. This could be a turning point to change the Kazakhstani protocol of AML treatment, where DNR and IDA are equally suggested for induction therapy.
Although the survival depending on the type of stem cell transplantation did not give statistical significance, it has medical importance for doctors and patients. The lack of donors for haploidentical stem cell transplantation is a big issue in Kazakhstan. Moreover, it should be noticed that the HSCT was initiated in our country only in 2010. This study gives an overview of the first 10-year experience’s results, implementation of acute myeloid leukemia treatment guidelines in Kazakhstan. The main limitation is a bias by age: the data is scarce of adults older than 60 years. It can be improved in further research, which can include all ages and be a prospective study.
Conclusion
There is no doubt that allogeneic HSCT is one of the most effective methods for patients with AML. Significant progress in AML treatment has been achieved with the introduction of intensive chemotherapy with subsequent allogeneic HSCT. Until 2010 HSCT was not available in Kazakhstan. Our study revealed that HSCT for patients with AML is a preferable treatment method in comparison with chemotherapy alone.
This study has several limitations. First, it is a retrospective cohort study, with a small sample size. The lost to follow-up patients or missed data could be an issue. Further research with a prospective design of study could be done. In order to increase the generalizability of the findings, the data from several organizations that carry out the HSCT could be combined for analysis.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- AML:
-
Acute myeloid leukemia
- DNR:
-
Daunorubicin
- Doxo:
-
Doxorubicin
- EFS:
-
Event-free survival
- FAB:
-
French-American British classification
- HSCT:
-
Hematopoietic stem cell transplantation
- IDA:
-
Idarubicin
- OS:
-
Overall survival
- WBC:
-
White blood cell
References
Kazakh Research Institute of Oncology and Radiology. Pokazateli oncologicheskoi sluzhby-Respubliki- Kazakhstan-za-2019-g. (2020) https://onco.kz/o-rake/ponimanie-raka/statistika-raka/.
World Health Organization. International Agency for Research on Cancer. Kazakhstan. Source: Globocan 2020 (2020) https://gco.iarc.fr/today/data/factsheets/populations/398-kazakhstan-fact-sheets.pdf.
Yamamoto, J. F. & Goodman, M. T. Patterns of leukemia incidence in the United States by subtype and demographic characteristics, 1997–2002. Cancer Causes Control. 19, 379–390 (2008).
Grove, C. & Vassiliou, G. Acute myeloid leukaemia: A paradigm for the clonal evolution of cancer?. Dis. Model. Mech. 7(8), 941–951 (2014).
Gilliland, D. G. Molecular genetics of human leukemia. Leukemia 12(Suppl 1), S7-12 (1998).
Dash, A. & Gilliland, D. G. Molecular genetics of acute myeloid leukaemia. Best Pract. Res. Clin. Haematol. 14(1), 49–64 (2001).
Gilliland, D. G. & Tallman, M. S. Focus on acute leukemias. Cancer Cell 1(5), 417–420 (2002).
Patel, J. P. et al. Prognostic relevance of integrated genetic profiling in acute myeloid leukemia. N. Engl. J. Med. 366, 1079–1089 (2012).
Cancer Genome Atlas Research Network et al. Genomic and epigenomic landscapes of adult de novo acute myeloid leukemia. N. Engl. J. Med. 368, 2059–2074 (2013).
Appelbaum, F. R. et al. Age and acute myeloid leukemia. Blood 107(9), 3481–3485 (2006).
Forman, D. et al. Cancer prevalence in the UK: Results from the EUROPREVAL study. Ann Oncol. 14, 648–654 (2003).
Parkin, D. M., Whelan, S. L., Ferlay, J., Teppo, L., & Thomas, D. B. E. Cancer incidence in five continents volume VIII. IARC Sci. Publications. 155 (2002).
Takami, A. Hematopoietic stem cell transplantation for acute myeloid leukemia. Int. J. Hematol. 107(5), 513–518 (2018).
Bondarenko, S.N. The role of allogeneic hemopoietic stem cell transplantation in programmed therapy of acute myeloid leukemia in adults. First Saint Petersburg State Medical University (Saint Petersburg State Medical University I.P. Pavlov of the Ministry of Health of the Russian Federation, Ph.D. in hematology. 2020.
Neame, P.B., Soamboonsrup, P., Browman, G.P., Meyer, R.M., Benger, A. et.al. Classifying acute leukemia by immunophenotyping: a combined FAB-immunologic classification of AML. (1986).
Döhner, H. et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 129(4), 424–447 (2017).
Voso, M. T. et al. MRD in AML: The role of new techniques. Front. Oncol. 9, 655 (2019).
Phikulsod, P. et al. The development of personalized medicine: Acute myeloid leukemia as a model. Siriraj Med. J. 71(5), 414–425 (2019).
Röllig, C. et al. Long-term prognosis of acute myeloid leukemia according to the new genetic risk classification of the European LeukemiaNet recommendations: Evaluation of the proposed reporting system. J. Clin. Oncol. 29(20), 2758–2765 (2011).
Rakhimbekova, G.A. & Pivovarova, I.A. Clinical protocol for diagnosis and treatment of acute myeloblastic leukemia in adults. Republican Center for Health Development of the Ministry of Health and Social Development of the Republic of Kazakhstan. Protocol #51 from January 11, 2019 (2019).
De Kouchkovsky, I. & Abdul-Hay, M. Acute myeloid leukemia: A comprehensive review and 2016 update. Blood Cancer J. 6(7), e441–e441 (2016).
Saultz, J. N. & Garzon, R. Acute myeloid leukemia: A concise review. J. Clin. Med. 5(3), 33 (2016).
Appelbaum, F. R. et al. Age, and acute myeloid leukemia. Blood 107(9), 3481–3485 (2006).
Acharya, U. H. et al. Impact of region of diagnosis, ethnicity, age, and gender on survival in acute myeloid leukemia (AML). J. Drug Assess. 7(1), 51–53 (2018).
Wei, H. et al. Distinct genetic alteration profiles of acute myeloid leukemia between Caucasian and Eastern Asian population. J. Hematol. Oncol. 11(1), 1–4 (2018).
Mrózek, K. et al. Prognostic significance of the European LeukemiaNet standardized system for reporting cytogenetic and molecular alterations in adults with acute myeloid leukemia. J. Clin. Oncol. 30(36), 4515 (2012).
Nguyen, S. et al. A white blood cell index as the main prognostic factor in t (8; 21) acute myeloid leukemia (AML): A survey of 161 cases from the French AML Intergroup. Blood J. Am. Soc. Hematol. 99(10), 3517–3523 (2002).
Padilha, S. L., Souza, E. J. D. S., Matos, M. C. C. & Domino, N. R. Acute myeloid leukemia: Survival analysis of patients at a university hospital of Paraná. Rev. Bras. Hematol. Hemoter. 37, 21–27 (2015).
Abramson, N. & Melton, B. Leukocytosis: Basics of clinical assessment. Am. Fam. Physician 62(9), 2053–2060 (2000).
Wang, H., Xiao, X., Xiao, Q., Lu, Y. & Wu, Y. The efficacy and safety of daunorubicin versus idarubicin combined with cytarabine for induction therapy in acute myeloid leukemia: A meta-analysis of randomized clinical trials. Medicine. 99(24) (2020).
Acknowledgements
We would like to thank the transplant team of the National Research Oncology Center for their great work throughout 10 years.
Author information
Authors and Affiliations
Contributions
G.K., J.S.: research design; data analysis and writing the paper. V.K.: research design, writing the paper. A.K. research design; data collection writing the paper. G.Z.: data analysis, writing the paper; Z.K., A.A., S.D.: data collection.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kulkayeva, G.U., Kemaykin, V.M., Kuttymuratov, A.M. et al. First report from a single center retrospective study in Kazakhstan on acute myeloid leukemia treatment outcomes. Sci Rep 11, 24001 (2021). https://doi.org/10.1038/s41598-021-03559-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-03559-3
- Springer Nature Limited