Abstract
Zinc is a biologically essential element and involved in a wide range of cellular processes. Here, we investigated the associations of zinc levels in hair with brain activity during the n-back working memory task using functional magnetic resonance imaging, fractional anisotropy (FA) of diffusion tensor imaging, and cognitive differences in a study cohort of 924 healthy young adults. Our findings showed that greater hair zinc levels were associated with lower brain activity during working memory in extensive areas in the default mode network (i.e., greater task-induced deactivation) as well as greater FA in white matter areas near the hippocampus and posterior limbs of the internal capsule. These findings advance previous non-neuroimaging findings of zinc’s associations with excitability, excitability-associated disorders, and myelination.
Similar content being viewed by others
Introduction
Zinc is an essential element in both brain and systemic physiology, as it is a component of more than 300 enzymes and involved in a wide range of normal cellular processes1. However, dysregulated greater level of zinc is neurotoxic and can harm neural tissue. Therefore, either high or low zinc can be detrimental to neural tissues2.
One of zinc’s key roles in the brain is modulation of neuronal excitability. Zinc is associated with inhibition of excitatory and inhibitory receptors as well as modulation of glutamate and GABA release, but reduced brain zinc availability leads to excitability, such as epileptiform brain activity, which suggests that the dominant effect of zinc in the normal brain is to reduce excitability3,4. Consistent with this hypothesis, epileptic patients have been shown to have reduced zinc levels5. There is also evidence that greater zinc levels or zinc supplementation are associated with greater sleep quality and quantity; however, the modulation of neural excitability caused by zinc may underlie these findings6. An increased excitability/inhibition ratio is thought to play a key role in the pathology of autism and schizophrenia7,8, and interestingly, children with autism (but not adults were shown to have robustly low hair zinc levels in the huge sample9 and patients with schizophrenia were shown to have low hair zinc levels in multiple studies10,11 and they also show substantially reduced brain zinc levels12. Zinc deficits are also known to lead to socializing deficits13. Animal studies have shown that zinc deprivation leads to poor performance in attention and short-term memory tasks2.
Further, zinc is known to play specific roles in the hippocampus. Zinc is highly concentrated in the hippocampus, and zinc deprivation has been shown to result in deficits in hippocampal activation and abrogate long-term potentiation (LTP), reduce hippocampal neurogenesis, and increase neuronal apoptosis that destroys structural hippocampal plasticity1,14,15. Animal studies have shown that zinc is important for hippocampal memory function via the abovementioned hippocampal activity15.
Previous neuroimaging studies have shown that zinc supplementation leads to reduction in electroencephalogram (EEG) amplitude5. Despite zinc’s wide range of physiological roles and the behavioral deficits and pathologies associated with zinc deprivation, the associations of body zinc level with brain activity measured by functional magnetic resonance imaging (fMRI) and white matter structure remain unknown. The purpose of this study was thus to investigate these issues. To achieve this, we assessed a wide range of cognitive functions, fMRI brain activity during the n-back working memory task, fractional anisotropy (FA) of diffusion tensor imaging (DTI), and hair zinc levels in a large sample of young adults. We also utilized a wide range of cognitive measures to see if zinc’s previously reported effects were seen in our sample and to reveal the nature of cognitive correlates of hair zinc levels.
As summarized in previous studies and mostly reproduced below16,17, hair mineral analysis is often assumed to be the best indicator of mineral levels in the body and has been used in multiple fields18. Hair is a repository of all elements that enter the body, and mineral levels in hair reflect accumulation of mineral composition over several months to years16. And thus, hair mineral levels are not affected substantially by rapid fluctuation in mineral intake and show long-term stability19. These characteristics give hair mineral analysis advantages over other methods to measure mineral levels such as blood and urine analyses. Studies showed positive correlation between concentrations of basic elements in the hair and in the body20,21. However, it was also suggested that hair mineral analysis requires, sampling by trained personnel, with standardized pre-analytical and analytical procedures, using suitable and sensitive equipment were required to obtain comparable results22. And new analytic methods and good practice have improved the precision of hair mineral analysis23. Zinc supplementation robustly increases the hair zinc content compared with controls24. A recent systematic review confirmed that, in healthy individuals, hair zinc is a reliable biomarker of zinc status25. Another review suggested that hair zinc is a biomarker of nutritional value and that zinc deficiency may be the consequence of dietary unavailability, excessive zinc loss, or inherited metabolic disturbances26. Unlike serum zinc, hair zinc levels are more stable and unaffected by diurnal variation, prolonged fasting, meal consumption, and acute infection27. In addition, although the bioavailability of zinc is reduced by phytates in foods of vegetable origin27, studies have shown that diets with high phytate intakes or high phytate:zinc molar ratios are associated with lower hair zinc levels28,29,30,31.
We hypothesized that greater task-induced deactivation (TID) in the default mode network (DMN) during working memory would be associated with greater hair zinc levels. This hypothesis is based on findings that TID in the DMN is associated with brain excitability/inhibition underlain by glutamate and GABA32, which are in turn associated with body zinc levels as described above. Further, TID in the DMN particularly during the externally directed attention demanding tasks has been used as measures to reflect brain excitability or failure to deactivate e.g.,33,34. Based on the specific associations of zinc with hippocampal activity, we predicted that greater hippocampal activity would be associated with greater hair zinc levels. We also hypothesized that greater FA would be associated with greater hair zinc levels based on evidence that zinc is essential for myelination35.
Methods
Subjects
The present study, which is a part of an ongoing project to investigate associations between brain imaging, cognitive function, and aging, included 924 healthy right-handed individuals (563 men and 361 women) from whom the data necessary for whole-brain analyses involving Zn levels were collected. The mean subject age was 20.7 years (standard deviation (SD), 1.8; age range: 18–27 years). Written informed consent was obtained from adult subjects. For nonadult subjects (age < 20 y.o.), written informed consent was obtained from their parents (guardians). This study was approved by the Ethics Committee of Tohoku University. For more detailed subject information, see Supplemental Methods.
Hair acquisition and hair mineral analysis
Scalp hair samples (approximately 4 cm length, 0.1 g weight) were collected from each subject and cut as close to the scalp as possible. Hair samples were sent to the La Belle Vie research laboratory and analyzed by established methods, as described previously17. For more details, see Supplemental Methods.
The logarithms of mineral levels in the hair were analyzed for all the measures used because logarithms of hair zinc levels were closer to the normal distribution and could alleviate the effects of outliers. For statistical analysis, zinc levels were converted to logarithms that could be used in the analyses, as reported in previous studies, including those from researchers affiliated with institutions in which mineral levels of our hair samples were measured (Research Laboratory, La Belle Vie Inc.)9,17,36,37,38,39,40.
Psychological measures
Following neuropsychological testing, several questionnaires were administered. These tests were chosen because low body zinc levels are associated with attention deficits, autism, and sleep disturbance as described in “Introduction”. The test descriptions in this subsection were largely reproduced from our previous studies41,42.
[A] RAPM43 is a non-verbal reasoning task and a representative measure of general intelligence. For more details, see our previous study44. [B] Systemizing quotient (SQ) and empathizing quotient (EQ). Japanese versions45 of the SQ and EQ46,47 were administered. EQ score was used as an index of empathizing and SQ score was used as an index of systemizing. [C] The Reverse Stroop and Stroop interference rates of the Stroop task (Hakoda’s version)48 was used to measure response inhibition and impulsivity. Hakoda’s version is a matching-type Stroop task requiring subjects to check whether their chosen answers are correct, unlike the traditional oral naming Stroop task. The test consists of two control tasks (word-color and color-word tasks), a Stroop task, and a reverse Stroop task. The reverse Stroop and Stroop interference rates are calculated from these tests. See our previous study for details49. [D] S-A creativity test. Creativity as divergent thinking was measured using the S-A creativity test50. [E] A computerized digit span task was used to assess working memory for details, see34. [F] Sleep disturbance subscale of the General Heath Questionnaire 3051. [G] The External-Preoccupation Scale52, which was used to measure maintenance of external focus on a specific object.
fMRI task
fMRI was used to map brain activity during cognitive tasks. The descriptions of this task were mostly reproduced from a previous study using the same methods53. The n-back task is a typical fMRI task with conditions of 0-back (simple cognitive process) and 2-back (working memory). Subjects were instructed to judge if a stimulus (one of four Japanese vowels presented visually) appearing “n” positions earlier was the same as the current stimulus by pushing a button. In the 0-back task, subjects were instructed to determine whether a presented letter was the same as the target stimulus by pushing a button. We used a simple block design. For more details, see Supplemental Methods.
The effects of inverted-U shaped activity patterns that are observed between the n-back task’s load and activity patterns (namely, the activity increases as the load increases, but when the tasks are too difficult, the activity drops)54 are not matter of concerns in this study, as in this study, 2-back tasks are apparently easy enough for all participants and the average accuracy rates are almost about 100%.
Image acquisition
The MRI acquisition methods were described in our previous study and reproduced below55. All MRI data acquisition was performed using a 3 T Philips Achieva scanner.
Diffusion-weighted data were acquired using a spin-echo EPI sequence (TR = 10,293 ms, TE = 55 ms, FOV = 22.4 cm, 2 × 2 × 2 mm3 voxels, 60 slices, SENSE reduction factor = 2, number of acquisitions = 1). The diffusion weighting was isotropically distributed along 32 directions (b value = 1,000 s/mm2). In addition, three images with no diffusion weighting (b value = 0 s/mm2) (b = 0 images) were acquired using a spin-echo EPI sequence (TR = 10,293 ms, TE = 55 ms, FOV = 22.4 cm, 2 × 2 × 2 mm3 voxels, 60 slices). FA and MD maps were calculated from the collected images using a commercially available diffusion tensor analysis package on the MR console. For more details, see Supplemental Methods. Descriptions in this subsection were mostly reproduced from a previous study using similar methods56.
Forty-two transaxial gradient-echo images (TR = 2.5 s, TE = 30 ms, flip angle = 90°, slice thickness = 3 mm, FOV = 192 mm, matrix = 64 × 64) covering the entire brain were acquired using an echo planar sequence. For the n-back sessions, 174 functional volumes were obtained.
Thorough instructions and thorough fixation by the pad were given as much as possible to prevent head motion during the scan. We did not exclude any subject from the fMRI analyses based on excessive motion during the scan. The subjects were young adults and the scan did not last for long. Only six subjects’ maximum movement from the original point in one of the directions exceeded 3 mm, and removing these subjects from analyses did not substantially alter the significant results of the present study. Furthermore, frame-wise displacement during fMRI scan did not significantly correlate with hair zinc levels after all the other covariates of the whole brain analyses of n-back tasks were controlled (partial correlation analysis, partial correlation coefficient = 0.006, p = 0.854).
Preprocessing of structural data
Preprocessing and analysis of diffusion and functional activation data were performed using SPM8 implemented in MATLAB. The following descriptions were mostly reproduced from our previous study using similar methods53. The methods are summarized below; full details and methodological considerations are provided in the Supplemental Methods. Before analysis, blood-oxygen-level dependent (BOLD) images were realigned and resliced to the mean of the BOLD images, which was then realigned to the mean b = 0 image as previously described 34. Because the mean b = 0 image was aligned with the FA image and MD map, the BOLD image, b = 0 image, FA image, and MD map were all aligned. Next, using a previously validated two-step segmentation algorithm of diffusion images and diffeomorphic anatomical registration through an exponentiated lie algebra (DARTEL)-based registration process that utilizes FA signal distribution for normalization57, all images—including gray matter segments [regional gray matter density (rGMD) map], white matter segments [regional white matter density (rWMD) map], and cerebrospinal fluid (CSF) segments [regional CSF density (rCSFD) map] of the diffusion images were normalized. The voxel size of the normalized FA images and segmented images was 1.5 × 1.5 × 1.5 mm3. The voxel size of the normalized BOLD images was 3 × 3 × 3 mm3.
Next, from the average images of the normalized WM segmentation images from 63 subjects, we created a mask image consisting of voxels with a WM signal intensity > 0.99. We then applied this mask image to the normalized FA images, thereby retaining only areas highly likely to be white matter from the normalized FA images. These images were smoothed (6 mm full-width half-maximum) and carried through to the second-level analyses of FA.
First-level analysis of functional imaging data
The following descriptions were mostly reproduced from our previous study using similar methods53. Individual-level statistical analyses were performed using a general linear model. A design matrix was fitted to each participant with one regressor in each task condition (0- or 2-back in the n-back task) using the standard hemodynamic response function. The cue phases of the n-back task were modeled in the same manner, but were not analyzed further. Six parameters obtained by rigid body corrections for head motion were regressed out by adding these variances to the regressor. The design matrix weighted each raw image according to its overall variability to reduce the impact of movement artifacts58. We removed low-frequency fluctuations using a high-pass filter with a cut-off value of 128 s. After estimation, beta images of contrasts of 2-back > rest and 0-back > rest were smoothed (8 mm full-width half-maximum) and taken to the second-level analyses.
Statistical analyses of non-whole-brain analyses
Behavioral data were analyzed using SPSS 22.0 (SPSS Inc., Chicago, IL). The associations of hair zinc levels with psychological outcome measures were tested using multiple regression analyses. The dependent variables were the 16 cognitive variables presented in Table 2. The independent variables comprised sex, age, self-reported height, and BMI (calculated from self-reported height and weight), and hair zinc levels. Results with a threshold of p < 0.05, corrected for false discovery rate (FDR) using the graphically sharpened method59, were considered statistically significant. Self-reported height, and BMI are included as covariates to exclude confounding effects of these as these variables often correlate with body mineral levels (in the case of zinc, after correcting the effects of age and sex, BMI was significantly negatively correlated with hair zinc levels, p = 0.002, although, height was not). And this is in accordance with our studies17,60.
Whole-brain statistical analysis
We investigated if the imaging measures were associated with individual differences in hair zinc. Whole-brain multiple regression analyses were performed using SPM8.
In the FA analysis, the covariates were sex, age, self-reported height, self-ported weight, BMI, and Zn levels in hair. Analyses were performed within the white matter mask created above.
In the fMRI analyses, the maps of dependent variables were beta estimate images of 2-back > rest contrast and 0-back contrast. Covariates included those used in the FA analysis as well as accuracies and reaction times in the 0-back and 2-back tasks and volume-level mean frame-wise displacement during the scan for the n-back task61.
Correction for multiple comparisons was performed using threshold-free cluster enhancement (TFCE)62 with randomized (5,000 permutations) nonparametric testing using the TFCE toolbox (https://dbm.neuro.uni-jena.de/tfce/). The family-wise error (FWE) threshold was corrected at p < 0.05.
Ethical statement
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Ethical approval
This study was approved by the Ethics Committee of Tohoku University.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Results
Basic data
The mean and standard deviation for age, general intelligence, and logarithm of hair zinc are presented in Table 1. The distribution of the logarithms of zinc levels in the hair of men and women are presented in Fig. 1.
Correlations between hair zinc levels and cognitive differences
After correcting for confounding variables and multiple comparisons, zinc levels in hair were not significantly correlated with any of the psychological variables. However, hair zinc showed a trend (P < 0.1, corrected) toward negative correlation with SQ score, the sleep disturbance subscore of the WHOGHQ30, and reverse Stroop interference. The results of all statistical analyses are presented in Table 2.
Correlations between hair zinc and FA
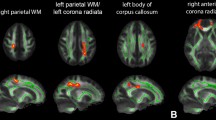
Whole-brain multiple regression analysis showed that zinc levels in hair were significantly and positively correlated with FA in white matter areas, including the left fornix, left internal capsule, left corticospinal tract, left cerebellar peduncle, and the white matter area in the left medical prefrontal cortex (mPFC) (Fig. 2a–d, Table 3).
Positive FA correlates with zinc levels in hair. (a, c) Regions with significant positive correlations between FA and hair zinc levels are overlaid on a “single subject” T1 image from SPM8. Results were obtained using a threshold of threshold-free cluster enhancement (TFCE) of p < 0.05 based on 5,000 permutations. Results were corrected at the whole-brain level. Significant correlations were found in (a) white matter areas close to the left hippocampus and (c) the area of the medial prefrontal cortex. (b, d) Scatter plots of the associations between hair zinc levels and mean FA in the clusters of (a) and (c).
Correlations between hair zinc and brain activity
Whole-brain multiple regression analysis showed that zinc levels in hair were significantly and negatively correlated with brain activity during the 0-back task in an area spreading across the left postcentral gyrus and the left precentral gyrus, which was deactivated during the 0-back task (Fig. 3a–c, Table 4).
Brain activity correlates with zinc levels in hair. Regions with significant correlations between the brain activity during the 0-back task and hair zinc levels are overlaid on a “single subject” T1 image from SPM8. Results were obtained using a threshold of threshold-free cluster enhancement (TFCE) of p < 0.05 based on 5,000 permutations. (a) Significant negative correlations were found in the area of the left precentral and postcentral gyrus. (b) Regions of deactivation during the 0-back task obtained from the 63 subjects from which the template of the diffusion image was created57. Results are overlaid on a “single subject” T1 image from SPM8. Results were thresholded at p < 0.001, uncorrected for visualization purposes. (c) Scatterplots of the associations between hair zinc levels and mean beta estimates in the clusters of (a).
In addition, zinc levels in hair were significantly and positively correlated with brain activity during the 2-back task in peripheral areas of the posterior cingulate gyrus, which are border areas between the areas deactivated during the 2-back task and areas activated during the 2-back task (Fig. 4a–c, Table 4).
Brain activity correlates with zinc levels in hair. (a, d, g, j, m) Regions with significant correlations between the brain activity during the 2-back task and hair zinc levels are overlaid on a “single subject” T1 image from SPM8. Results were obtained using a threshold of threshold-free cluster enhancement (TFCE) of p < 0.05 based on 5,000 permutations. Significant positive correlations were observed in (a) the peripheral areas of the bilateral posterior cingulate gyrus and significant negative correlations were found in (d) the mPFC, (g) the left fusiform gyrus, (j) left superior temporal gyrus and contingent regions, and (m) the left middle cingulate gyrus. (b, e, h, k, n) Regions deactivated during the 2-back task obtained from the 63 subjects from which the template of diffusion image was created57. Results are overlaid on a “single subject” T1 image from SPM8. Results were obtained using a threshold of threshold-free cluster enhancement (TFCE) of p < 0.05 based on 5,000 permutations. (c, f, i, l, o) Scatterplots of the associations between hair zinc levels and mean beta estimates in the clusters of (a), (d), (g), (j), and (m).
Whole-brain multiple regression analysis also showed that zinc levels in hair were significantly and negatively correlated with brain activity during the 2-back task in the extensive areas deactivated during the 2-back task. The areas of significant correlation included the cluster around the mPFC, the cluster around the left superior temporal gyrus, left Rolandic operculum, left Heschl gyrus, left insula, left precentral and postcentral gyrus, the cluster mainly around the right precentral gyrus, postcentral gyrus, and right superior parietal lobule, bilateral clusters mainly around the bilateral fusiform gyrus, bilateral clusters in the bilateral middle cingulate gyrus, the cluster in the right insula, and the cluster in the left orbitofrontal gyrus (Fig. 4d–o, Table 5). We used permutation based corrections for multiple comparisons and effects of existence of outliers are taken care of in corrections of multiple comparisons.
In this study, our focus was TID in the DMN, and TID in the DMN occurs regardless of the whether the task is 2-back or 0-back in mostly similar areas, although the magnitude is different. Furthermore, differences in the brain activity between patients with schizophrenia and control subjects were mostly similarly regardless of whether the task was 0-back task or 2-back task, including in the areas of DMN (i.e., subtracting the activity during the 0-back task from the brain activity during the 2-back task substantially eliminates the group differences)33,63. Therefore, we did not primarily analyze the contrast of (2-back – 0-back) as in case of another study that focused on TID in the DMN33. However, additional analysis involving this contrast revealed that after correcting the same covariates of the whole brain analyses of 2-back and 0-back, the brain activity of the contrast of (2-back – 0-back) showed a significant negative correlation in the area of mPFC [x, y, z = − 12, 63, 3, TFCE score = 691.14, P = 0.012, corrected for FWE (permutation using TFCE), 6,939 mm3] and a significant positive correlation in the area spreading around the posterior cingulate gyrus and the left hippocampus [x, y, z = − 12, − 42, 15, TFCE score = 667.14, P = 0.013, corrected for FWE (permutation using TFCE), 2,970 mm3]. The results indicated that the significant results of the whole brain analyses of the brain activity of the contrast of (2-back – 0-back) are part of the significant results of the correlation between 2-back and hair zinc level (i.e., Fig. 4).
Discussion
The present study revealed brain activity during cognitive tasks and white matter structural properties in a large cohort of young adults in a developed country. Partly consistent with our hypothesis, greater hair zinc level was associated with lower brain activity during a working memory task in extensive areas in the DMN. Since these areas are deactivated during the task, the results suggest that greater hair zinc is associated with greater TID in the DMN. Although negative correlations were found in many areas of the DMN, there were few significant correlations in the hippocampus. Greater hair zinc was also associated with lower brain activity in the left postcentral gyrus and the left precentral gyrus, which was deactivated during the simple cognitive task (greater TID in this area). Further, partly consistent with our hypothesis, greater hair zinc was positively correlated with FA in white matter areas, mainly the left fornix, left internal capsule, left corticospinal tract, left cerebellar peduncle, and white matter area in the left mPFC. Finally, partly consistent with our hypothesis, greater hair zinc was tended to be associated with lower systemizing which is one of the key traits of autistic spectrum disorder, lower sleep disturbance, and lower reverse Stroop interference scores.
Previous work has reported that greater hair zinc is associated with greater TID64 in extensive areas of the DMN. Although we can only speculate on the micro-level mechanisms from our macro-level MRI observations, the results are congruent with the idea that zinc inhibits both excitatory and inhibitory receptors, that the dominant role of zinc in the normal brain is to reduce excitability3,4, and that TID in the DMN is associated with brain excitability/inhibition underlain by glutamate and GABA32. It is possible that greater zinc availability affects glutamate and GABA receptors and reduces excitability/inhibition, resulting in enhancement of the TID in the DMN. In addition,proteins of the ProSAP/Shank family act as major organizing scaffolding elements within the postsynaptic density of excitatory synapses65, and mutations in the genes of these proteins are associated with autism and Phelan-McDermid syndrome, which is characterized by the occurrence of seizures and autism-like symptoms for summary, see ref66. Zinc ion, which is released from the presynaptic vesicles and influxed into the postsynaptic compartment3, interplays with proteins of the ProSAP/Shank family and also plays a key role in their functions12. An animal model study has shown that acute and prenatal zinc deficiencies lead to the loss of proteins of the ProSAP/Shank family and autism spectrum disorder-related symptoms66. The observed associations are congruent with findings of low hair zinc in autistic children and schizophrenia patients9 and the notion that an increased excitability/inhibition ratio plays a key role in the pathology of these conditions7,8. Further, zinc level was associated with lower systemizing in the present study, which is a characteristic of autistic spectrum disorders. Thus, our psychological finding may be congruent with previous clinical findings. Finally, greater zinc levels were associated with reduced sleep disturbance in the present study, consistent with previous findings that zinc is important for sleep quality, possibly due to its ability to suppress brain excitability6. Interestingly, primary insomnia is also characterized by reduced TID in the DMN67 and better Stroop performance is associated with greater GABA levels in the brain68. Therefore, hair zinc’s association with low characteristics of autism and sleep disturbance may be parallel to the reduced TID in the DMN and underlain by reduced neural excitability. These speculations may be congruent with the suggestion that zinc deficiency leads to autism-like symptoms, and zinc supplementation may help in improving this problem through animal model studies66,69, through animal model studies66,69, a large sample study of reduced hair zinc levels in children with autism spectrum disorders9. However, the results revealed by intervention studies of zinc are mixed69 and future studies should confirm these notions.
Although extensive areas in several brain regions deactivated during the working memory task showed negative correlations between fMRI activity and hair zinc, such associations were mostly lacking in the hippocampus, which also showed deactivation during the working memory task. As described above, the hippocampus was a region of particular interest in this study. Although it is difficult to make inferences from negative findings in whole-brain imaging analyses using stringent thresholds, we can speculate about the lack of results for the hippocampus. Increased availability of zinc largely works to reduce excitability in the brain; however, zinc deprivation produces deficits in hippocampal activation and abrogates LTP, which are important activities for memory formation (and may also be important for working memory)15. Therefore, the role of zinc in deactivation during the working memory task may have distinct characteristics. Future studies are needed to investigate the effects of hair zinc on brain activity during a task that recruits the hippocampus more so than during rest.
Partly consistent with our hypothesis, greater hair zinc was associated with greater FA in the white matter area of the left fornix, left internal capsule, left corticospinal tract, left cerebellar peduncle, and white matter area in the left medical prefrontal cortex. This is congruent with previous studies suggesting the importance of zinc in myelination35, as increased myelination is thought to lead to greater FA in DTI70. In addition, it has been well established that myelination continues even in adulthood71,72,73. Moreover, human studies involving DTI have shown experience-dependent white matter plasticity measured via FA, even in adulthood74,75. Although the importance of zinc in myelination has been discussed in many reviews4,76, its exact mechanism remains unclear. Thus, we can only speculate. Among the regions showing significant correlations, the fornix acts as a major output tract of the hippocampus. In mice, induction of myelination by neural activity has been demonstrated both in vivo and in vitro (Deremens et al., 1996). As stated earlier, zinc is important for facilitation of activity critical to memory formation in the hippocampus1,14,15. Thus, one intriguing possibility is that facilitated activity involving the hippocampus enhances myelination and leads to FA in relevant areas, such as the fornix. Consistently, partial correlation analysis revealed that after controlling for hair zinc levels and all covariates of whole brain analyses of n-back tasks and the FA analysis, mean beta estimate of the significant positive cluster of the area close to the hippocampus/posterior cingulate gyrus (Fig. 4a) and the mean FA value of the significant cluster of the area close to the left hippocampus (Fig. 2a) were significantly and positively correlated (partial correlation coefficient = 0.151, P < 0.001). However, this is pure speculation. Other studies have shown that zinc binds to CNS myelin basic protein in the presence of phosphate, which results in aggregation of this protein and may affect myelination77. Ultimately, we cannot determine micro-level mechanisms from macro-level neuroimaging data, and so future studies are needed to investigate such issues. As such, the effects of zinc on white matter microstructural properties are assumed to be independent from the effects of zinc on greater deactivation, and this is not particularly strange given the diverse mechanisms each nutrient affects.
There is at least one limitation of this study. As it is a cross-sectional macro-level neuroimaging study, we cannot determine causal relationships or the underlying micro-level neural mechanisms behind the observed associations involving hair zinc levels. Future studies using other methods, such as interventions involving zinc supplementation or animal experiments, could complement the present findings.
In conclusion, we investigated the associations of zinc levels in hair with cognitive domains, brain activity, and FA in a relatively large cohort of young adults. Greater zinc levels were associated with lower TID in the DMN and greater FA in white matter areas near the hippocampus and areas like the posterior limbs of the internal capsule. Greater hair zinc showed a tendency of association with lower systemizing which is a key characteristic of autistic spectrum disorders, lower sleep disturbance, and lower reverse Stroop interference. These findings advance previous non-neuroimaging findings of zinc’s associations with excitability, disorders associated with excitability, and myelination.
Data availability
All the experimental data obtained in the experiment of this study will be available for the studies that were admitted in the ethics committee of Tohoku University’s medical faculty. All the data sharing should be first admitted by the ethics committee of Tohoku University’s medical faculty.
References
Suh, S. W. et al. Decreased brain zinc availability reduces hippocampal neurogenesis in mice and rats. J. Cereb. Blood Flow Metab. 29, 1579–1588 (2009).
Mocchegiani, E., Bertoni-Freddari, C., Marcellini, F. & Malavolta, M. Brain, aging and neurodegeneration: Role of zinc ion availability. Prog. Neurobiol. 75, 367–390 (2005).
Frederickson, C. J., Koh, J.-Y. & Bush, A. I. The neurobiology of zinc in health and disease. Nat. Rev. Neurosci. 6, 449–462 (2005).
Bhatnagar, S. & Taneja, S. Zinc and cognitive development. Br. J. Nutr. 85, S139–S145 (2001).
Penland, J. G. Behavioral data and methodology issues in studies of zinc nutrition in humans. J. Nutr. 130, 361S-364S (2000).
Ji, X. & Liu, J. Associations between blood zinc concentrations and sleep quality in childhood: A cohort study. Nutrients 7, 5684–5696 (2015).
Rubenstein, J. & Merzenich, M. M. Model of autism: Increased ratio of excitation/inhibition in key neural systems. Genes Brain Behav. 2, 255–267 (2003).
Lewis, D. A., Curley, A. A., Glausier, J. R. & Volk, D. W. Cortical parvalbumin interneurons and cognitive dysfunction in schizophrenia. Trends Neurosci. 35, 57–67 (2012).
Yasuda, H., Yoshida, K., Yasuda, Y. & Tsutsui, T. Infantile zinc deficiency: Association with autism spectrum disorders. Sci. Rep. 1, 129 (2011).
Rahman, M. A. et al. Zinc, manganese, calcium, copper, and cadmium level in scalp hair samples of schizophrenic patients. Biol. Trace Elem. Res. 127, 102–108 (2009).
Ghanem, A.-E.A. et al. Copper and zinc levels in hair of both schizophrenic and depressed patients. J. For. Med. Clin. Toxicol. 17, 89–102 (2009).
M Grabrucker, A., Rowan, M. & C Garner, C. Brain-delivery of zinc-ions as potential treatment for neurological diseases: Mini review. Drug Deliv. Lett. 1, 13–23 (2011).
Prasad, A. S. Discovery of human zinc deficiency: Its impact on human health and disease. Adv. Nutr. 4, 176–190 (2013).
Gao, H.-L. et al. Zinc deficiency reduces neurogenesis accompanied by neuronal apoptosis through caspase-dependent and-independent signaling pathways. Neurotox. Res. 16, 416 (2009).
Swardfager, W. et al. Potential roles of zinc in the pathophysiology and treatment of major depressive disorder. Neurosci. Biobehav. Rev. 37, 911–929 (2013).
Priya, M. D. L. & Geetha, A. Level of trace elements (copper, zinc, magnesium and selenium) and toxic elements (lead and mercury) in the hair and nail of children with autism. Biol. Trace Elem. Res. 142, 148–158 (2011).
Takeuchi, H. et al. Association of hair iron levels with creativity and psychological variables related to creativity. Front. Hum. Neurosci. 7, article 875, 1–9 (2013).
Lech, T. Lead, copper, zinc, and magnesium content in hair of children and young people with some neurological diseases. Biol. Trace Elem. Res. 85, 111–126 (2002).
Ayodele, J. & Bayero, A. Lead and zinc concentrations in hair and nail of some Kano inhabitants. Afr. J. Environ. Sci. Technol. 3, 164–170 (2009).
Chłopicka, J. et al. Lead and cadmium in the hair and blood of children from a highly industrial area in Poland. Biol. Trace Elem. Res. 62, 229–234 (1998).
Kedzierska, E. Concentrations of selected bioelements and toxic metals and their influence on health status of children and youth residing in Szczecin. Ann. Acad. Med. Stetin. 49, 131–143 (2003).
Kosanovic, M. & Jokanovic, M. Quantitative analysis of toxic and essential elements in human hair. Clinical validity of results. Environ. Monit. Assess. 174, 635–643 (2011).
Bass, D. A., Hickok, D., Quig, D. & Urek, K. Trace element analysis in hair: factors determining accuracy, precision, and reliability. Altern. Med. Rev. 6, 472–481 (2001).
Kim, J. E., Yoo, S. R., Jeong, M. G., Ko, J. Y. & Ro, Y. S. Hair zinc levels and the efficacy of oral zinc supplementation in children with atopic dermatitis. Acta Derm. Venereol. 94, 558–562 (2014).
Lowe, N. M., Fekete, K. & Decsi, T. Methods of assessment of zinc status in humans: A systematic review. Am. J. Clin. Nutr. 89, 2040S-2051S (2009).
Shamberger, R. Validity of hair mineral testing. Biol. Trace Elem. Res. 87, 1–28 (2002).
Hotz, C. & Brown, K. H. Assessment of the risk of zinc deficiency in populations and options for its control. Food Nutr. Bull. 25, S91–S204 (2004).
Gibson, R. et al. Are young women with low iron stores at risk of zinc as well as iron deficiency? In Trace Elements in Man and Animals 10 edn. 323–328 (Springer, New York, 2002).
Gibson, R. S., Vanderkooy, P. D. S. & Thompson, L. Dietary phytate× calcium/zinc millimolar ratios and zinc nutriture in some Ontario preschool children. Biol. Trace Elem. Res. 30, 87–94 (1991).
Ferguson, E. L., Gibson, R. S., Thompson, L. U. & Ounpuu, S. Dietary calcium, phytate, and zinc intakes and the calcium, phytate, and zinc molar ratios of the diets of a selected group of East African children. Am. J. Clin. Nutr. 50, 1450–1456 (1989).
Gibson, R. S. & Huddle, J.-M. Suboptimal zinc status in pregnant Malawian women: Its association with low intakes of poorly available zinc, frequent reproductive cycling, and malaria. Am. J. Clin. Nutr. 67, 702–709 (1998).
Hu, Y., Chen, X., Gu, H. & Yang, Y. Resting-state glutamate and GABA concentrations predict task-induced deactivation in the default mode network. J. Neurosci. 33, 18566–18573 (2013).
Whitfield-Gabrieli, S. et al. Hyperactivity and hyperconnectivity of the default network in schizophrenia and in first-degree relatives of persons with schizophrenia. Proc. Natl. Acad. Sci. USA. 106, 1279–1284 (2009).
Takeuchi, H. et al. Failing to deactivate: The association between brain activity during a working memory task and creativity. Neuroimage 55, 681–687 (2011).
Liu, H., Oteiza, P. I., Gershwin, M. E., Golub, M. S. & Keen, C. L. Effects of maternal marginal zinc deficiency on myelin protein profiles in the suckling rat and infant rhesus monkey. Biol. Trace Elem. Res. 34, 55–66 (1992).
Yasuda, H., Yonashiro, T., Yoshida, K., Ishii, T. & Tsutsui, T. Mineral imbalance in children with autistic disorders. Biomed. Res. Trace Elem. 16, 285–292 (2005).
Yasuda, H. et al. Metallomics study using hair mineral analysis and multiple logistic regression analysis: Relationship between cancer and minerals. Environ. Health Prevent. Med. 14, 261–266 (2009).
Munakata, M. et al. Reduced exposure to mercury in patients receiving enteral nutrition. Tohoku J. Exp. Med. 210, 209–212 (2006).
Yasuda, H., Yoshida, K., Yasuda, Y. & Tsuts, T. Two age-related accumulation profiles of toxic metals. Curr. Aging Sci. 5, 105–111 (2012).
Yasuda, H. et al. High accumulation of aluminum in hairs of infants and children. Biomed. Res. Trace Elem. 19, 57–62 (2008).
Takeuchi, H. et al. Brain structures in the sciences and humanities. Brain Struct. Funct. 220, 3295–3305 (2015).
Takeuchi, H. et al. Effects of working memory-training on functional connectivity and cerebral blood flow during rest. Cortex 49, 2106–2125 (2013).
Raven, J. Manual for Raven’s progressive matrices and vocabulary scales (Oxford Psychologists Press, Oxford, 1998).
Takeuchi, H. et al. Regional gray matter volume of dopaminergic system associate with creativity: Evidence from voxel-based morphometry. Neuroimage 51, 578–585 (2010).
Wakabayashi, A. et al. Empathizing and systemizing in adults with and without autism spectrum conditions: Cross-cultural stability. J. Autism Dev. Disord. 37, 1823–1832 (2007).
Baron-Cohen, S., Richler, J., Bisarya, D., Gurunathan, N. & Wheelwright, S. The systemizing quotient: an investigation of adults with Asperger syndrome or high-functioning autism, and normal sex differences. Philos. Trans. R. Soc. Lond. Ser. Biol. Sci. 358, 361–374 (2003).
Baron-Cohen, S. & Wheelwright, S. The empathy quotient: An investigation of adults with Asperger syndrome or high functioning autism, and normal sex differences. J. Autism Dev. Disord. 34, 163–175 (2004).
Hakoda, Y. & Sasaki, M. Group version of the Stroop and reverse-Stroop test : The effects of reaction mode, order and practice. Kyoikushinrigakukenkyu (Educ. Psychol. Res.) 38, 389–394 (1990).
Takeuchi, H. et al. Degree centrality and fractional amplitude of low-frequency oscillations associated with Stroop interference. Neuroimage 119, 197–209 (2015).
Society_For_Creative_Minds Manual of S-A Creativity Test. (Tokyo shinri Corporation, Tokyo, Japan, 1969).
Iwata, N., Uno, B. & Suzuki, T. Psychometric properties of the 30-item version general health questionnaire in Japanese. Psychiatry Clin. Neurosci. 48, 547–556 (1994).
Sakamoto, S. The preoccupation scale: Its development and relationship with depression scales. J. Clin. Psychol. 54, 645–654 (1998).
Takeuchi, H. et al. Cognitive and neural correlates of the 5-repeat allele of the dopamine D4 receptor gene in a population lacking the 7-repeat allele. Neuroimage 110, 124–135 (2015).
Callicott, J. H. et al. Complexity of prefrontal cortical dysfunction in schizophrenia: more than up or down. Am. J. Psychiatry 160, 2209–2215 (2003).
Takeuchi, H. et al. The association between resting functional connectivity and creativity. Cereb. Cortex 22, 2921–2929 (2012).
Takeuchi, H. et al. Impact of videogame play on the brain’s microstructural properties: Cross-sectional and longitudinal analyses. Mol. Psychiatry 21, 1781–1789 (2016).
Takeuchi, H. et al. White matter structures associated with empathizing and systemizing in young adults. Neuroimage 77, 222–236 (2013).
Diedrichsen, J. & Shadmehr, R. Detecting and adjusting for artifacts in fMRI time series data. Neuroimage 27, 624–634 (2005).
Benjamini, Y. & Hochberg, Y. On the adaptive control of the false discovery rate in multiple testing with independent statistics. J. Educ. Behav. Stat. 25, 60–83 (2000).
Takeuchi, H. et al. Association of copper levels in the hair with gray matter volume, mean diffusivity, and cognitive functions. Brain Struct. Funct. 224, 1203–1217 (2019).
Power, J. D., Barnes, K. A., Snyder, A. Z., Schlaggar, B. L. & Petersen, S. E. Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. Neuroimage 59, 2142–2154 (2012).
Smith, S. M. & Nichols, T. E. Threshold-free cluster enhancement: addressing problems of smoothing, threshold dependence and localisation in cluster inference. NeuroImage 44, 83–98 (2009).
Jansma, J., Ramsey, N., Van Der Wee, N. & Kahn, R. Working memory capacity in schizophrenia: A parametric fMRI study. Schizophr. Res. 68, 159–171 (2004).
McKiernan, K. A., Kaufman, J. N., Kucera-Thompson, J. & Binder, J. R. A parametric manipulation of factors affecting task-induced deactivation in functional neuroimaging. J. Cogn. Neurosci. 15, 394–408 (2003).
Boeckers, T. M., Bockmann, J., Kreutz, M. R. & Gundelfinger, E. D. ProSAP/Shank proteins—A family of higher order organizing molecules of the postsynaptic density with an emerging role in human neurological disease. J. Neurochem. 81, 903–910 (2002).
Grabrucker, S. et al. Zinc deficiency dysregulates the synaptic ProSAP/Shank scaffold and might contribute to autism spectrum disorders. Brain 137, 137–152 (2014).
Drummond, S. P. et al. Neural correlates of working memory performance in primary insomnia. Sleep 36, 1307–1316 (2013).
Silveri, M. M. et al. Frontal lobe γ-aminobutyric acid levels during adolescence: Associations with impulsivity and response inhibition. Biol. Psychiatry 74, 296–304 (2013).
Hagmeyer, S., Sauer, A. K. & Grabrucker, A. M. Prospects of zinc supplementation in autism spectrum disorders and shankopathies such as Phelan McDermid syndrome. Front. Synaptic Neurosci. 10, 11 (2018).
Demerens, C. et al. Induction of myelination in the central nervous system by electrical activity. Proc. Natl. Acad. Sci. USA. 93, 9887–9892 (1996).
Benes, F. M., Turtle, M., Khan, Y. & Farol, P. Myelination of a key relay zone in the hippocampal formation occurs in the human brain during childhood, adolescence, and adulthood. Arch. Gen. Psychiatry 51, 477–484 (1994).
Nunez, J. L., Nelson, J., Pych, J. C., Kim, J. H. Y. & Juraska, J. M. Myelination in the splenium of the corpus callosum in adult male and female rats. Dev. Brain Res. 120, 87–90 (2000).
Yakovlev, P.I. & Lecours, A.R. The myelogenetic cycles of regional maturation of the brain. Region. Dev. Brain Early Life 3–70 (1967).
Bengtsson, S. L. et al. Extensive piano practicing has regionally specific effects on white matter development. Nat. Neurosci. 8, 1148–1150 (2005).
Takeuchi, H. et al. Training of working memory impacts structural connectivity. J. Neurosci. 30, 3297–3303 (2010).
Ciubotariu, D. & Nechifor, M. Zinc involvements in the brain. Rev. Med. Chir. Soc. Med. Nat. Iasi 111, 981–985 (2007).
Riccio, P. et al. Specificity of zinc binding to myelin basic protein. Neurochem. Res. 20, 1107–1113 (1995).
Takeuchi, H. et al. Shorter sleep duration and better sleep quality are associated with greater tissue density in the brain. Sci. Rep. 8, 5833 (2018).
Pike, N. Using false discovery rates for multiple comparisons in ecology and evolution. Methods Ecol. Evol. 2, 278–282 (2011).
Vul, E., Harris, C., Winkielman, P. & Pashler, H. Reply to comments on “puzzlingly high correlations in fMRI studies of emotion, personality, and social cognition”. Perspect. Psychol. Sci. 4, 319–324 (2009).
Maldjian, J. A., Laurienti, P. J. & Burdette, J. H. Precentral gyrus discrepancy in electronic versions of the Talairach atlas. Neuroimage 21, 450–455 (2004).
Maldjian, J. A., Laurienti, P. J., Kraft, R. A. & Burdette, J. H. An automated method for neuroanatomic and cytoarchitectonic atlas-based interrogation of fMRI data sets. Neuroimage 19, 1233–1239 (2003).
Tzourio-Mazoyer, N. et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 15, 273–289 (2002).
Acknowledgements
We respectfully thank Yuki Yamada for operating the MRI scanner, and Haruka Nouchi for being an examiner of psychological tests. We also thank study participants, the other examiners of psychological tests, and all of our colleagues in Institute of Development, Aging and Cancer and in Tohoku University for their support. This study was supported by a Grant-in-Aid for Young Scientists (B) (KAKENHI 23700306) and a Grant-in-Aid for Young Scientists (A) (KAKENHI 25700012) from the Ministry of Education, Culture, Sports, Science, and Technology. The authors would like to thank Enago (https://www.enago.jp) for the English language review. We would like to thank La Belle Vie Inc. and its employees for the hair mineral level analyses as well as Dr. Yasuda and Dr. Sonobe for their technical advice regarding the analyses. This study was supported by JST/RISTEX, JST/CREST, a Grant-in-Aid for Young Scientists (B) (KAKENHI 23700306) and a Grant-in-Aid for Young Scientists (A) (KAKENHI 25700012) from the Ministry of Education, Culture, Sports, Science, and Technology.
Author information
Authors and Affiliations
Contributions
H.T., Y.T. and R.K. designed the study. H.T., R.N., R.Y., Y.K., S.N., A.S., K.I., S.H., T.A., C.M.M., K.S., T.N., S.I., S.Y., D.M. and Y.S. collected the data. H.T. analyzed the data and prepared the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Takeuchi, H., Taki, Y., Nouchi, R. et al. Succeeding in deactivating: associations of hair zinc levels with functional and structural neural mechanisms. Sci Rep 10, 12364 (2020). https://doi.org/10.1038/s41598-020-69277-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-69277-4
- Springer Nature Limited
This article is cited by
-
Lead exposure is associated with functional and microstructural changes in the healthy human brain
Communications Biology (2021)