Abstract
Due to fresh water scarcity, farmers are using polluted water for irrigation. This research was conducted to study the bioaccumulation of Pb in wheat (Cv. Shafaq-2006). The experiment was comprised of seven treatments of lead i.e. 0–1,000 mg Pb/kg. The results revealed that lead severely reduces germination (− 30%), seedling fresh weight (− 74%), seedling dry weight (− 77%), vigor index (− 89%), tolerance index (− 84%), plant height (− 33%), number of leaves (− 41%), root fresh weight (− 50%), shoot fresh weight (− 62%), root dry weight (− 63%), shoot dry weight (− 71%), and root length (− 45%). The physiological parameters also respond negatively like stomatal conductance (− 82%), transpiration rate (− 72%) and photosynthetic rate (− 74%). Similarly, biochemical parameters also showed negative impacts, like carotenoids (− 41), total chlorophyll (− 43), chlorophyll a (− 42) and chlorophyll b (− 53). Yield parameters like the number of seed/plant, seed weight/plant, 1,000 seed weight and harvest index were reduced by 90%, 88%, 44% and 61%, respectively in T6. In addition, protein contents (− 81%), phosphorous (− 60%) and potassium (− 55%) were highly effected in the highest lead concentration (T6). Lead accumulation was extremely higher in seeds (119%) as compared to control plants. Lead bio-accumulation above threshold concentrations in crop parts is a serious human health concern.
Similar content being viewed by others
Introduction
Fresh water scarcity is a major issue for agriculture in developing countries including Pakistan. To meet water requirements, farmers are now using industrial wastewater1. These industrial wastewaters have the high amount of toxic heavy metals (HM) like Pb, Ni, Cd, Zn, Fe, Mn, etc. Heavy metals tend to bio-accumulate in crops and are creating serious health threats to human and ecosystem2. Other sources of metal entry in the ecosystem include mining activities, industrial effluents, agronomic practices and use of sludge as manure3,4,5. Worldwide environmental and human health problems are increasing sharply due to heavy metal contamination. Cultivation of crops near these contaminated sites result heavy metal bio-accumulation in crops and poor growth. These accumulated metals pose high risk to humans as well as to livestock health, in case of ingestion6.
Zajac et al., investigated 195 TSIP (Toxic Site Identification Program) sites in 33 LMICs (low- and middle-income countries) and reported the 820,000 womoen of childbearing age are at the risk for lead exposure7. Lead is a major pollutant in the ecosystem, being persistent/high retention time (150–1,500 years) and shows negative impacts on human8,9. Lead can accumulate in plants from various routes including water, air and soil. Prominent harmful effects of Pb include interference with nutrient uptake, germination reduction, reduced photosynthesis, delay in plant growth, disturbed respiration, metabolism alteration, changes in enzymatic activities, changes in root morphology and inhibition of mitosis (in tip)10,11,12,13, enlarged vacuoles, deformed nucleolus, increased plasmolysis and damaged thylakoid14. Heavy metals are also responsible for ROS5,15 (reactive oxygen species), MDA (malondialdehyde) formation of excessive amount, mainly in chloroplast and mitochondria of leaves, root and shoot16. Rafaqat at el, reported the damage in root ultra-structures in Brassica napus L. due to chromium toxicity. They also alter the natural antioxidant enzymes of plants17. These ROS have the tendency to attack biomolecules and may cause cell death. Thus, lead contamination serves as a severe problem for agriculture for agriculture18.
Rice, wheat, cotton and sugarcane are most important cash crops in Pakistan and account 75% of total output. Wheat is Pakistan's main food crop. Due to the shortage of irrigation water farmers are using industrial wastewater for irrigation. The presence of lead in the food chain can cause chronic health problems19,20. The application of plant growth regulator like 5-aminolevulinic acid (ALA) improves metal tolerance in crops15. Therefore, this study was designed and executed to investigate the effect of Pb on germination, seedling growth, physiological, biochemical, mineral contents and yield.
Material and methods
Germination experiment
Wheat cultivar (Shafaq-2006) was selected as test variety. The Germination experiment was setup in petri dishes (90 mm) in the growth room of The Botany Department, GC University Lahore, Pakistan. Petri dishes were washed, sterilized and blotting paper was placed in each petri dish. Twenty five, seeds were surface sterilized and placed in each petri dish. For irrigation purpose, seven treatments of lead were prepared using lead nitrate.
Crop specie | Sees per petri dish | Lead treatments | Replicates |
|---|---|---|---|
1 | 25 | 7 | 4 |
Treatments were named To, T1, T2, T3, T4, T5, T6 having 0, 100, 200, 400, 600, 800 and 1,000 mg of Pb/L. All the petri dishes were arranged in completely randomized design (CRD). 2 ml of each treatment was added in respective petridish on daily bases. Germination was noted with the emergence of radical. Data was collected for germination %, seedling length, plumule length, radical length and fresh biomass. Seedling vigor index and tolerance index was estimated by multiplying seedling length and germination percentage21. The Metals Tolerance Index is a diagnostic metric used to identify samples with a high degree of organisms tolerant of metals.
Pot experiment
The pot experiment was set up in Botanical Garden (located at The Mall road) of Government College University in ambient condition. No additional instrument was used to control any abiotic factor (light, temperature, humidity, etc.). Each pot (earthen, 12 inch diameter) was washed, dried, lined with polythene bag and filled with 5.5 kg soil (1:6, humus: garden soil) mix. Lead nitrate was added in the soil in different concentration. Treatments were named To, T1, T2, T3, T4, T5, T6 having 0, 100, 200, 400, 600, 800 and 1,000 mg of Pb kg−1. Pot’s positions were changed on a weekly basis to keep climatic condition uniform. Data of weather conditions and other meteorological parameters is given in Table S1. All other agronomic practices were kept same for all treatments. All parameters were measured at harvest. After harvesting, plants were washed, dried and stored in polythene zipper bags. Biomass (fresh and dry) of root/shoot and length were measured at harvest using electric balance and measuring rod. For measuring dry weight, crop samples were dried (at 70 °C) for 24 h. Yield attributes were calculated by the protocols of Zadoks22.
Physiological and biochemical attributes
IRGA, ‘Infra-Red Gas Analyzer’ (LCA4 Model) was used to measure stomatal conductance, transpiration and photosynthesis rate. Three leaves from each pot were selected and readings were noted at 75th and 130th day23. Chlorophyll contents were determined by following the protocols of Arnon24. Carotenoid extraction was carried out in dim light where 1 g of randomly selected fresh leaves was crushed in acetone. Homogenate was filtered and more acetone was added to make the final volume up to 50 ml. Carotenoids were estimated with the help of spectrophotometer at the wavelength of 440.5 nm25.
where: “V” is the volume used for analysis; “383” is the extinction coefficient of carotenoids; “As” is the absorbance of the sample; “Ab” is the cuvette error; “W” is the weight of the sample in gram.
Mineral contents and quality attributes
Potassium was estimated through flame photometer (PF Pt JENWWAY, England)26. For phosphorous quantification, 1 g of plant sample was dried, crushed, and placed at 650 °C in the furnace for 3 h27. Burned samples was added in 10 ml of H2SO4 (0.7 N), stayed for about 1 h and later filtered (through Whatman-No-42) and the final volume was raised to 50 ml.
Over dried potassium phosphate (0.43 g) was dissolved in one liter of distilled water to make stoke solution. Different standards (ranging from 2 to 50 ppm) were prepared from this stock solution. Ammonium vanadomolybdate (10 ml) and the respective standard (5 ml) were mixed and stayed for 10 min. Absorbance was noted by spectrophotometer at 410 nm. The same protocol was used for samples. The standard graph was plotted using absorbance value of the standard and absorbance value of phosphorous.
Nitrogen was determined by using Kjeldahl method using the following formula:
where ‘A’ is the HCl used, B is HCl used for blank; 14.01 is atomic mass of nitrogen.
While protein was estimated by multiplying the nitrogen contents (%) with 6.25.
Lead accumulation
Lead accumulation in roots, shoots and leaves were determined by following protocol of Chandra28. Crop samples were 1st washed with double distilled water and later with calcium chloride (10 mM solution). Samples were dried and turned into ashes at 450 °C. Digestion of ash was carried out in HNO3 (2%), which was later filtered with glass fiber filter29. Lead was quantified by “inductively coupled plasma spectrophotometer (Thermo Electron; Model IRIS Intrepid II XDL, USA)”. Detection limit ranges from 8 to 80 ng L−1.
Data analysis
Results were evaluated through ANOVA and DMR using Costat version 3.0330.
Result and discussion
This study has given significant results regarding the Pb toxicity in the germination, growth, physiological, biochemical and yield attributes of wheat. Data regarding germination attributes revealed that Pb treatments significantly affected the germination percentage, fresh weight, dry weight, plumule length, radical length and seedling length of the wheat (Table 1 and Fig. 1). Increase Pb concentration results in negative impacts in wheat. The highest decline recorded was in germination i.e. 30%. Radical length was decreased by 45%, fresh weight of seedling by 74%, dry weight of seedlings by 77%), seedling vigor index by 89% and tolerance index by 74% in T6 (1,000 mg Pb/L) compared to the control (Table 1 and Fig. 1). Chun31 observed that at 0–0.5 mg Pb/kg non-significant adverse impact of lead on germination index and energy required during the process of germination. Likewise, growth rate of roots and shoots also remain unaffected at low level contamination of lead32. However, concentration above 4.5 mg/kg of soil is toxic to plant health, which decreases germination33. Roots of plants are more affected by lead toxicity as compared to other plant parts12,34. Excessive Pb contamination (including Pb) results in the formation of ‘reactive oxygen species’ (ROS) in mitochondria, chloroplast and cellular compartments14,35. The results of (seedling growth) present study are in line with that of Yadav36 who stated that growth attributes were significantly affected by increasing the Pb concentration. Co-cropping of T. minuta and soybean enhanced the Pb accumulation in T. minuta without posing any health risk from grain consumption of soybean9.
Data regarding the growth attributes revealed that Pb significantly (p ≤ 0.01; 0.05) affect the growth attributes (Table 2). Minimum plant height (58.2 ± 0.32 cm), root length (13.5 ± 0.24 cm), number of leaves (21.67 ± 0.05) were noticed in T6. Dry weight and fresh weight was also affected negatively at higher lead concentration. Minimum fresh weight of roots (0.66 ± 0.05 g), shoot (2.53 ± 0.04 g), and dry weight of roots (1.61 ± 0.02 g), shoot (0.34 ± 0.05 g) were noticed in T6 (Table 2). Gopal and Rizvi37 also observed that due to increase in lead concentration length and weight of root and shoot decreases. Adverse effects of lead were not apparent at early stage of the plant’s life but detrimental effects increased with the increase in maturity level of plant. Metal uptake, translocation and bio-accumulation are age dependent. Antoniadis38 also reported that root development in excessive Pb concentration was poor. The major reason behind the reduction in biomass of plants is reduction in the rate of photosynthesis and nitrogen metabolism39. Osma40 reported that specie and Pb concentration significantly effect the fresh weight in brassica. Decrease in fresh weight of shoots among different species was observed in following order, B. juncea. < B. oleracea I < B. carinata. Plants may develop resistance and as a result may survive in lead contaminated soils41. Gill et al., reported the damage in leaf mesophyll, root tips and disruption of golgbodies, diffused cell wall due to chromium toxicity at 400 µM14.
Maximum reduction in all biochemical and physiological attributes was noticed in those pots whose soil was spiked with 1,000 mg Pb/kg. all the treatments differ significantly (p ≤ 0.01; 0.05). A linear decrease in the physiological and growth attributes was noted with increase in Pb concentration. Regarding the physiological attributes maximum reduction in photosynthetic rate (44%), stomatal conductance (82%) and transpiration rate (72%) was noticed in T6 (Table 3). Maximum reduction in chlorophyll a (44%), chlorophyll b (53%), carotenoids contents (42%) and total chlorophyll contents (43%) was noticed in those pots whose soil was spiked with the 1,000 mg Pb/kg (Fig. 2). Similar results were observed by Zeng42 that performed an experimental analysis by applying lead acetate at six different levels (0–900 mg/kg). This may be due to non-toxic effect of lead at low concentration. All the physiological and biochemical attributes of the wheat was reduced by increasing the Pb concentration that might be due to lead toxicity as it damages the chloroplast structure, decreases chlorophyll synthesis, restricted enzymatic and carotenoids activities. Lead is also responsible for obstructions in electron transport chain, deficiency of carbon dioxide (due to closure of stomata) and altered thylokoid membrane. Qadri43 reported that metal presence reduce the chlorophyllase and results in low chlorophyll content. Studies have indicated that chlorophyll b contents is more disturbed then chlorophyll a39. Rafaqat et al., reported the decline in photosynthetic rate in Brassica napus L. due to the elevated stress of chromium5.
All the Pb treatments significantly (p ≤ 0.01; 0.05) decrease the mineral contents and the quality attributes of the wheat (Table 4). Phosphorous content decreased from 11 to 58%, potassium contents from 14 to 34%, nitrogen contents from 38 to 82% and proteins contents from 18 to 81% in the T6 as compared to the control. Range of all the yield attributes was as follows, phosphorous contents (18.85–7.51 ppm), potassium contents (409.20–269.57 ppm), nitrogen contents (1.54–0.28%) and protein contents (9.63–1.75%).
Result of Pot experiment were significantly changed by Pb treatments (Table 5). At higher concentration the yield decrease. Minimum number of seeds per plant (16.33 ± 0.58), seed weight per plant (0.85 ± 0.09 g), 1,000 seed’s weight (30.03 ± 0.63 g), straw weight per plant (2.62 ± 0.12 g), and harvest index (263.39 ± 11.52%) was noticed in T6 (Table 5). In order to feed the growing population, farmers demand those varieties of crops which give more yields all the yield attributes were reduced by the increasing Pb concentrations. Scientists are also focused on developing new and improved varieties (with genetic engineering), to produce more yield. The current research investigate severe negative impacts of Pb on wheat yield. Xiong44 reported that cadmium, lead and zinc accumulate in cabbage and spinach which were grown near smelter. Lead interferes with active sites of enzymes and results in low yield. Zheljazkov and Nielsen45 revealed that a 400 m away source of lead (through air) affected corn mint yield by 16%. Similarly, 14% decrease in yield of essential oils was observed due to air pollution46. Gill et al ., reported increased size and number of starch grains due to chromium in oilseed rape cultivars24. The presence of heavy metals revealed elite molecular transporters (MTs) genes that were responsible for water transmembrane transporter activity5.
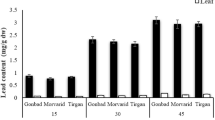
The results of the Pb accumulation in wheat plant including root, shoot and seed were depicted in the (Table 6). Significant variation in the treatment means was revealed. The extent of increase in lead concentration was quite alarming compared to control treatments. For instance, roots showed 4,600% enhance in Pb quantity in T6. Likewise, shoots and seeds showed an increase of 9,800% and 118% in Pb concentration, respectively. Lead concentration decreases in parts of the wheat in the following order root > shoot > seeds. The considerable increase in lead concentration was noticed in T6 as compared to the control (Table 6). Lead uptake by roots is directly proportional to the lead concentration. Other factors which contribute towards Pb adsorption include: type of fertilizer, amount of fertilizer, microbial activity, soil pH, precipitates of carbonate/phosphate and concentration of extractable lead in soil47. Antoniadis38 reported lead translocation decreases significantly as the metal moves away from root, which implies that roots accumulate more lead then seeds and shoots. Different studies revealed high accumulation rate of lead in roots; Kenaf root may retain upto 85% of total Pb present in plant body48, Thlaspi praecox can accumulate up to 80% Pb49 and Indian mustard (Brassica junce) has the tendency to accumulate upto 95% of total Pb50. Zhang51 conclude that rice cultivars different variable in metal uptake and translocation. Lead absorption and translocation to leaves depends on number of factors and these factors and variable among different crops. Shoot- accumulators store more lead in shoots of the plant while root-accumulator tends to store large amount of lead in roots and allow very small concentration of lead to be transported to above ground parts of crop. Another study revealed that uptake of lead by plants depends upon the surface area of roots52. Heavy metal effects root tip cells more as enlarged vacuole, disrupted cell membrane, plasmolysis and damaged mitochondrial thylakoid is more prominent in root cells17. Presence of different forms of lead in plants is also responsible for difference in its translocation rate. Sharma53 concluded that ions and lower molecular complexes are more mobile then other forms. Roots may absorb more lead in such plants but high molecular weight and complexes restrict its translocation and distribution to aerial crop part54. In an experiment conducted by Basharat et al., the lead induce reduction in Brassica napus L. biomass, reduced macronutrients in shoot, increased ROS and MDA6. Ali et al., reported the use of plant growth regulators (5-aminolevulinic acid, ALA) can cpounter the injurious effect of heavy metals in crops15.
Conclusion
The present study revealed that lead imparts number of negative effects on germination, physiological, biochemical, yield, mineral, and growth and quality attributes of wheat crop. It was observed that all adverse impacts of lead were due to reduced photosynthesis rate and related phenomenon like decreased rate of transpiration and stomatal conductance. Accumulation rate of lead in wheat parts (shoots, seeds and roots) was also above threshold level. Lead contamination in wheat is alarming as it is the staple food in Pakistan. Slow bioaccumulation of lead ultimately leads towards serious disorders and illness. And certain remedial techniques should be adopted to restrict the entry of lead from contaminated soils to wheat. Maintaining supply of lead free wheat is the need of an hour.
References
Rezapour, S., Atashpaz, B., Moghaddam, S. S. & Damalas, C. A. Heavy metal bioavailability and accumulation in winter wheat (Triticum aestivum L.) irrigated with treated wastewater in calcareous soils. Sci. Total Environ. 656, 261–269. https://doi.org/10.1016/j.scitotenv.2018.11.288 (2019).
Wang, S., Wu, W., Liu, F., Liao, R. & Hu, Y. Accumulation of heavy metals in soil–crop systems: a review for wheat and corn. Environ. Sci. Pollut. Res. 24, 15209–15225. https://doi.org/10.1007/s11356-017-8909-5 (2017).
Rezapour, S., Kouhinezhad, P., Samadi, A. & Rezapour, M. Level, pattern, and risk assessment of the selected soil trace metals in the calcareous cultivated Vertisols. Chem. Ecol. 8, 692–706. https://doi.org/10.1080/02757540.2013.810728 (2015).
Zhang, Y. et al. Heavy metal accumulation and health risk assessment in soil-wheat system under different nitrogen levels. Sci. Total Environ. 622–623, 1499–1508. https://doi.org/10.1016/j.scitotenv.2017.09.317 (2018).
Gill, R. A. et al. Reduced glutathione mediates pheno-ultrastructure kinome and transportome in chromium-induced Brassica napus L.. Front. Plant Sci. 8, 2037. https://doi.org/10.3389/fpls.2017.02037 (2017).
Khan, M. U., Malik, R. N. & Muhammad, S. Human health risk from heavy metal via food crops consumption with wastewater irrigation practices in Pakistan. Chemosphere 93, 2230–2238. https://doi.org/10.1016/j.chemosphere.2013.07.067 (2013).
Zajac, L. et al. Probabilistic estimates of prenatal lead exposure at toxic hotspots in low- and middle-income countries. Environ. Res. 183, 109251. https://doi.org/10.1016/j.envres.2020.109251 (2020).
Odongo, A. O., Moturi, W. N. & Mbuthia, E. K. Heavy metals and parasitic geo helminths toxicity among geophagous pregnant women: a case study of Nakuru Municipality, Kenya. Environ. Geochem. Health 38, 123–131. https://doi.org/10.1007/s10653-015-9690-3 (2015).
Vergara, C., María, C., Judith, L. P. & Rodriguez, H. Effects of co-cropping on soybean growth and stress response in lead-polluted soils. Chemosphere 246, 125833. https://doi.org/10.1016/j.chemosphere.2020.125833 (2020).
Shekar, C. C., Sammaiah, D., Shasthree, T. & Reddy, K. J. Effect of mercury on tomato growth and yield attributes. Int. J. Pharm. Biol. Sci. 2, B358–B364. https://doi.org/10.1007/s11356-018-1498-0 (2011).
Tiwari, K., Singh, N. K. & Rai, U. N. Chromium phytotoxicity in radish (Raphanus sativus): effects on metabolism and nutrient uptake. Bull. Environ. Contam. Toxicol. 91, 339–344. https://doi.org/10.1007/s00128-013-1047-y (2013).
Bergqvist, C., Herbert, R., Persson, I. & Greger, M. Plants influence on arsenic availability and speciation in the rhizosphere, roots and shoots of three different vegetables. Environ. Pollut. 184, 540–546. https://doi.org/10.1016/j.envpol.2013.10.003 (2014).
Rizwan, M. et al. A critical review on effects, tolerance mechanisms and management of cadmium in vegetables. Chemosphere 182, 90–105. https://doi.org/10.1016/j.chemosphere.2017.05.013 (2017).
Rafaqat, A. G. et al. Chromium-induced physio-chemical and ultrastructural changes in four cultivars of Brassica napus L.. Chemosphere 120, 154–164. https://doi.org/10.1016/j.chemosphere.2014.06.029 (2015).
Ali, B. et al. Regulation of cadmium-induced proteomic and metabolic changes by 5-aminolevulinic acid in leaves of Brassica napus L.. PLoS ONE 10(4), e0123328. https://doi.org/10.1371/journal.pone.0123328 (2015) (eCollection).
Basharat, A. et al. Promotive role of 5-aminolevulinic acid on mineral nutrients and antioxidative defense system under lead toxicity in Brassica napus. Ind. Crops Prod. 52, 617–626. https://doi.org/10.1016/j.indcrop.2013.11.033 (2014).
Gill, R. A. et al. Genotypic variation of the responses to chromium toxicity in four oilseed rape cultivars. Biol. Plant. 58, 539–550. https://doi.org/10.1007/s10535-014-0430-9 (2014).
Tandon, V., Gupta, B. M. & Tandon, R. Free radicals/reactive oxygen species. JK Pract. Nurs. Res. Pract. 12, 143–148. https://doi.org/10.1155/2011/260482 (2005).
Yang, Y., Liu, H., Xiang, X. H. & Liu, F. Y. Outline of occupational chromium poisoning in China. Bull Environ. Contam. Toxicol. 90, 742–749. https://doi.org/10.1007/s00128-013-0998-3 (2013).
Vaiserman, A. M. Aging-modulating treatments: from reductionism to a system oriented perspective. Front. Genet. 5, 1–3. https://doi.org/10.3389/fgene.2014.00446 (2016).
Bewley, J.D. & Black, M. Biochemistry of germination and growth. In: Physiology and Biochemistry of seeds in relation to germination. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-66668-1_5 (1978).
Zadoks, J. C., Chang, T. T. & Konzak, C. T. A decimal code for the growth stages of cereals. Weed Res. 14, 415–421. https://doi.org/10.1111/j.1365-3180.1974.tb01084.x (1974).
Vernay, P. et al. Effect of chromium species on phytochemical and physiological parameters in Datura innoxia. Chemosphere 72, 763–771. https://doi.org/10.1016/j.chemosphere.2008.03.018 (2008).
Arnon, D. T. Copper enzymes in isolated chloroplasts: polyphenol oxidase in Beta vulgaris. J. Plant Physiol. 24, 1–15. https://doi.org/10.1104/pp.24.1.1 (1949).
Zofia, L., Kmiecik, W. & Korus, A. Content of vitamin C, carotinoids, chlorophylls and polyphenols in green parts of dill (Anethum graveolens L.) depending on plant height. J. Food Compos. Anal. 19, 134–140. https://doi.org/10.1016/j.jfca.2005.04.009 (2006).
Ryan, J., Estfan, G. & Rashid, A. Soil and Plant Analysis Laboratory Manual. 2nd ed., pp. 87–89. ISBN 9788172337650 (2001).
Panichev, N., Mandiwana, K., Kataeva, M. & Siebert, S. Determination of Cr (VI) in plants by electrothermal atomic absorption spectrometry after leaching with sodium carbonate. Spectrochim. Acta Part B 60, 699–703. https://doi.org/10.1016/j.sab.2005.02.018 (2005).
Chandra, R., Kumar, P. K. & Singh, J. Impact of an aerobically treated and untreated (raw) distillery effluent irrigation on soil micro flora, growth, total chlorophyll and protein contents of Phaseolus aureus L.. J. Environ. Biol. 25, 381–385 (2004).
Velthof, G., Van-Beusichem, M. & Raijmakers, W. Relationship between availability indices and plant uptake of nitrogen and phosphorus from organic products. Plant Soil 200, 215. https://doi.org/10.1023/A:1004336903214 (1998).
Steel, R. G. D. & Torrie, J. H. Principles and Procedures of Statistics 172–177 (McGraw Hill Book Crop., Inc., Singapore, 1984).
Chun, X. L. et al. Effects of arsenic on seed germination and physiological activities of wheat seedlings. J. Environ. Sci. 19, 725–732. https://doi.org/10.1016/S1001-0742(07)60121-1 (2007).
Alghobar, M. A. & Suresha, A. Evaluation of metal accumulation in soil and tomatoes irrigated with sewage water from Mysore city, Karnataka India. J. Saudi Soc. Agric. Sci. 16, 49–59. https://doi.org/10.1016/j.jssas.2015.02.002 (2017).
Yourtchi, M. S. & Bayat, H. Y. Effect of cadmium toxicity on growth, cadmium accumulation and macronutrient content of durum wheat (Dena CV). Int. J. Agric. Crop Sci. 6, 1099–1103 (2013).
Barberon, M. & Geldner, N. Radial transport of nutrients: the plant root as a polarized epithelium. Plant Physiol. 166, 528–537. https://doi.org/10.1104/pp.114.246124 (2014).
Shahid, M. et al. Heavy-metal-induced reactive oxygen species: phytotoxicity and physicochemical changes in plants. Rev. Environ. Contam. Toxicol. 232, 1–44. https://doi.org/10.1007/978-3-319-06746-9_1 (2014).
Yadav, K. K. et al. Mechanistic understanding and holistic approach of phytoremediation: a review on application and future prospects. Ecol. Eng. 120, 274–298. https://doi.org/10.1016/j.ecoleng.2018.05.039 (2018).
Gopal, R. & Rizvi, A. H. Excess lead alters growth, metabolism and translocation of certain nutrients in radish. Chemosphere 70, 1539–1544. https://doi.org/10.1016/j.chemosphere.2007.08.043 (2008).
Antoniadis, V. et al. Trace elements in the soil-plant interface: phytoavailability, translocation, and phytoremediation—a review. Earth Sci. Rev. 172, 621–645. https://doi.org/10.1016/j.earscirev.2017.06.005 (2017).
Hamid, N., Bukhari, N. & Jawaid, F. Physiological responses of phaseolus vulgaris to different lead concentrations. Pak. J. Bot. 42, 239–246 (2010).
Osma, M., Serin, Z. & Leblebici, A. Heavy metals accumulation in some vegetables and soils in Istanbul. Ekoloji. 21, 1–8. https://doi.org/10.5053/ekoloji.2011.821 (2012).
Singh, S., Parihar, P., Singh, R., Singh, V. P. & Prasad, S. M. Heavy metal tolerance in plants: role of transcriptomics, proteomics, metabolomics, and ionomics. Front. Plant Sci. 6, 1143–1148. https://doi.org/10.3389/fpls.2015.01143 (2015).
Zeng, L. S., Liao, M., Chen, C. L. & Huang, C. Y. Effects of lead contamination on soil microbial activity and physiological indices in soil-Pb-rice (Oryza sativa L.) system. Chemosphere 65, 567–574. https://doi.org/10.1016/j.chemosphere.2006.02.039 (2006).
Qadir, S., Qureshi, M. I., Javed, S. & Abdin, M. Z. Genotypic variation in phytoremediation potential of Brassica juncea cultivars exposed to Cd stress. Plant Sci. 167, 1171–1181. https://doi.org/10.1016/j.plantsci.2004.06.018 (2004).
Xiong, T. T. et al. Foliar uptake and metal(loid) bioaccessibility in vegetables exposed to particulate matter. Environ. Geochem. Health 36, 897–909. https://doi.org/10.1007/s10653-014-9607-6 (2014).
Zheljazkov, V. D. & Nielsen, N. E. Effect of heavy metals on peppermint and cornmint. Plant Soil 178, 59–66. https://doi.org/10.1007/BF00011163 (1996).
Lavado, R. S., Porcelli, C. A. & Alvarez, R. Nutrient and heavy metal concentration and distribution in corn, soybean and wheat as affected by different tillage systems in Argentine Pampas. Soil Tillage Res. 62, 55–60. https://doi.org/10.1016/S0167-1987(01)00216-1 (2001).
Gupta, N. et al. Trace elements in soil-vegetables interface: translocation, bioaccumulation, toxicity and amelioration—a review. Sci. Total Environ. 651, 2927–2942. https://doi.org/10.1016/j.scitotenv.2018.10.047 (2019).
Ho, W. M., Ang, L. H. & Lee, D. K. Assessment of Pb uptake, translocation in Kenaf (Hibiscus cannabinus L.) for phytoremediation of sand tailings. J. Evniron. Sci. 20, 1341–47. https://doi.org/10.1016/S1001-0742(08)62231-7 (2008).
Vogel-Mikus, K., Drobne, D. & Regvar, M. Zn, Cd and Pb accumulation and arbuscular mycorrhizal colonization of pennycress Thlaspi praecox Wulf (Brassicaceae) from the vicinity of a lead mine and smelter in Slovenia. Environ. Pollut. 133, 233–242. https://doi.org/10.1016/j.envpol.2004.06.021 (2005).
Liu, J. G., Li, K. Q., Xu, J. K. & Zhang, Z. J. Lead toxicity, uptake and translocation in different rice cultivars. Plant Sci. 165, 793–802. https://doi.org/10.1016/S0168-9452(03)00273-5 (2003).
Zhang, M. K., Liu, Z. Y. & Wang, H. Use of single extraction methods to predict bioavailability of heavy metals in polluted soils to rice. Commun. Soil Sci. Plant Anal. 41, 820–831. https://doi.org/10.1080/00103621003592341 (2010).
Shahid, M. et al. Foliar heavy metal uptake, toxicity and detoxification in plants: a comparison of foliar and root metal uptake. J. Hazard. Mater. 325, 36–58. https://doi.org/10.1016/j.jhazmat.2016.11.063 (2016).
Sharma, R. K., Agrawal, M., Bhushan, S. & Agrawal, S. B. Physiological and biochemical responses resulting from cadmium and zinc accumulation in carrot plants. J. Plant Nutr. 33, 1066–1079. https://doi.org/10.1080/01904161003729774 (2010).
McBride, M. B., Shayler, H. A., Russell-Anelli, J. M., Spliethoff, H. M. & Marquez, L. G. Arsenic and lead uptake by vegetable crops grown on an old Orchard site amended with compost. Water Air Soil Pollut. 226, 265–272. https://doi.org/10.1007/s11270-015-2529-9 (2015).
Acknowledgements
We are thankful to field/research staff of SDSC and Botanic Garden, Government College University, Lahore. We are also grateful to PCSIR Labs, Lahore for their technical assistance.
Author information
Authors and Affiliations
Contributions
A.K. and M.F. designed the study. U.H. and A.K. set up the experiment. G.Z. helped in IRGA analysis. F.S. help in heavy metal analysis. G.Z. and F.S. designed tabled and graphs. L.S. and M.F. conducted statistical analysis. Research write up was done by A.K., M.F., F.S. all authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors declare no competing interests
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kanwal, A., Farhan, M., Sharif, F. et al. Effect of industrial wastewater on wheat germination, growth, yield, nutrients and bioaccumulation of lead. Sci Rep 10, 11361 (2020). https://doi.org/10.1038/s41598-020-68208-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-68208-7
- Springer Nature Limited