Abstract
Microbial fermentation of plant material alters the composition of volatile and non-volatile plant natural products. We investigated the antioxidant, anticancer, and antiviral properties of extracts of defatted soybean meal fermented with Aspergillus fumigatus F-993 or A. awamori FB-133 using in vitro methods. Gas chromatography–mass spectrometry analysis of soybean meal fermented with A. awamori FB-133 and A. fumigatus F-993 identified 26 compounds with 11,14-octadecadienoic acid and methyl ester (63.63%) and 31 compounds with butylated hydroxytoluene (66.83%) and δ-myrcene (11.43%) as main constituents, respectively. The antioxidant activities of DSM extract were 3.362 ± 0.05 and 2.11 ± 0.02 mmol TE/mL, FDSM treated with A. awamori FB-133 were 4.763 ± 0.05 and 3.795 ± 0.03 mmol TE/mL and FDSM treated with A. fumigatus F-993 were 4.331 ± 0.04 and 3.971 ± 0.02 mmol TE/mL as determined by ABTS and FRAP assays, respectively. Both fermented extracts had better antioxidant activity than the unfermented extract as shown by multiple antioxidant activity assays. The concentration of fermented extracts required for 50% inhibition of cell viability was significantly lower than that of the unfermented extract when tested against the human liver cancer cell line HepG2 as shown by cell viability assays, indicating strong anticancer activity. The IC50 values for DSM, FDSM with A. fumigatusF-993 and FDSM with A. awamori FB-133 were27, 16.88 and 8.60 μg/mL, respectively. The extract of FDSM with A. awamori FB-133 showed the strongest anticancer activity, compared to DSM and FDSM with A. FumigatusF-993 extracts. Fermented extracts also reduced hepatitis A virus titres to a greater extent than unfermented extracts, thus showing strong antiviral property. Hepatitis A virus titres were reduced by 2.66 and 3 log10/0.1 mL by FDSM with A. fumigatusF-993 and FDSM by A.awamori FB-133, respectively, compared to DSM (5.50 log10/0.1 mL). Thus, the fermentation of soybean meal with A. fumigatusF-993 or A. awamori FB-133 improves the therapeutic effect of soybean extracts, which can be used in traditional medicine.
Similar content being viewed by others
Introduction
Fermentation of plant materials causes a significant change in bioactive volatile and non-volatile components1. Moreover, the concentrations of both flavonoids and the unsaturated fatty acid esters, as well, increase due to the fermentation of soybean. Extracts of fermented soybean have shown strong antioxidant, anti-breast cancer and antiviral activities2. A growing major objective interest is to develop special catalytic systems (one-pot tandem), that integrate both the reactivity of a chemical catalyst and a selected active site of an enzyme3. Fungi (as yeasts, moulds), bacteria and several microorganisms are known to produce lipases that digest lipids. Moreover, fungal lipases are used as industrial catalysts to resolve racemic alcohols used for preparing some prostaglandins, steroids, and carboxylic nucleoside analogues. Such lipases also catalyse the hydrolysis of short chain triglycerides, which represent very good substrates for lipases. In addition, proteases and amylases, which catalyse the hydrolysis of proteins and carbohydrates respectively, are widely used in different industries. Recently, the biocatalytic potential of microbial lipases, in both aqueous and non-aqueous media, have encourage many industries to utilize these enzymes in a variety of great important reactions, especially organic synthesis reactions. The region selective nature of lipases are used for different purposes such as, fat modification, butter constituents, biofuels, synthesis of personal care products, the resolution of chiral drugs, flavour enhancers and synthesize of cocoa. Therefore, pharmacists, biophysicists, organic chemists, biochemists, biotechnologists, microbiologists and process engineers are now widely using lipases4. Although, soybeans are commonly rich in phenolics (e.g. flavanols, flavanones, phenolic acids, other phenolic compounds), yet isoflavones are the most abundant. However, such compounds can be modified through fermentation for improved antioxidant activities5. Fermentation of soybean with filamentous fungi has been reported to enhance their phenolic content and radical scavenging activity6. However, the antiviral activity of fermented soybean, for the treatment of viral diseases infecting human intestines, is not yet examined. Soybean fermentation can be done by using a number of microorganisms, for example fungi (as filamentous types) bacteria (as Bacillus sp. and lactic acid bacteria). Many fungal species, such as A. awamori, are commonly used for the production of many in fermented foods and flavours7,8 fermentation of soybean with Aspergillus oryzae increased its antioxidant activity9. Some Lactobacillus spp. and Bifidobacterium spp. have been reported to β-glucosides and enhance the isoflavone bioavailability by fermentation10,11. Several researchers have recommended the use of solid state fermentation method “SSF” for the production many bacterial and fungal metabolites, using different substrates1,6,12.
Cancer refers to the development of abnormal cells characterized by autonomous growth and loss of differentiation13. The American Cancer Society estimates the probable numbers of new cancer cases and deaths in the United States of America annually and compiles data on cancer incidence, mortality, and survival14. However, chemotherapy is often accompanied by adverse effects, which prolongs patient recovery13. Recently, there has been a more growing interest in the large-scale industrial utilization of biocatalysts in the production of fine chemicals and medicinal compounds15. In recent years, plant products such as flavonoids, terpenoids, and alkaloids have been studied for their varied pharmacological properties including antioxidant activity against free radicals that can cause cancer, inflammation, and diabetes16. The high concentrations of both polyphenols and volatile constituents in plant extracts account for their higher antioxidant activity17,18,19. Recently, plant extracts have been screened for anticancer activity to identify novel therapeutic strategies against cancer20,21,22.
The major causative pathogens of foodborne illness are viruses, followed by bacteria and parasites23. Several pure and crude plant derivatives and extracts are now being used as novel antimicrobial and/or antiviral against herpes simplex virus type 1 (HSV-1) infections24. In the present times, the anti-HSV-1 potential of plant extracts including phenolic compounds, polyphenols, peptides, isoprenoidal glycosides, pyranocoumarins and benzophenones have been evaluated25. Several fungal species of the genus Aspergillus, e.g. A. niger and A. phoenicus were found to increase b-glucosidase enzyme (BGL) yield, which plays a key role in biomass hydrolysis by producing monomeric sugars from cellulose-based oligosaccharides12. In view of the above facts, the present study evaluates the efficiency of A. fumigatus F-993 and A. awamori FB-133 in bioactive compound production from soybean meal and the antioxidant, anticancer, and antiviral potentials of the ethanolic extracts of defatted soybean meal (DSM) fermented by A. fumigatus F-993 or A. awamori FB-133.
Methods
Raw material and chemicals
Defatted soybean
Egyptian defatted soybean was obtained from the Department of Medicinal and Aromatic Plants, Ministry of Agriculture, Cairo, Egypt.
Microorganisms and culture conditions
Non mycotoxin producing two fungi namely A. fumigates F-993 and A. awamori FB-133 were obtained from Microbial Chemistry Laboratory (National Research Centre, Dokki, Cairo, Egypt) and cultured on potato dextrose agar slants at 30 °C for 72 h. The spore suspensions were prepared by adding 10 mL sterile water to slant cultures and gently scraping the culture with a sterile wire loop.
Fermentation of defatted soybean
The fermentation was carried out under solid state fermentation in 250 mL Erlenmeyer flasks containing 5 g of defatted soybean moistened to 50% with distilled water and inoculated with 1 mL spore suspension (106 spores). The cultures were incubated at 30 °C for 3 days for solid state fermentation24.
Defatted soybean extraction
Volatile compounds were extracted from fermented defatted soy bean DSM samples according to previously reported protocols with some modifications25,26. The DSM and fermented DSM (FDSM) samples were ground individually to a powder using a coffee bean blender. Five grams of the ground powder was transferred into a 25 mL test tube and extracted with 15 mL ethanol. The supernatant was separated from the residue by centrifuging at 2000 × g for 10 min in a centrifuge and transferred to a clean test tube. The residue was re-extracted with 15 mL ethanol; the separated ethanol layers were combined, and dried using a vacuum evaporator at less than 50 °C. The dried soybean extract was weighed and stored at −20 °C.
Gas chromatography–mass spectrometry (GC–MS) analysis
About 2 μl of each extract was used for GC–MS analysis using a HP 5890 GC coupled to HP5970 MS. The MS was set to an ionization voltage of 70 eV and the mass range of m/z 39–400 amu was scanned. The GC oven temperature was maintained initially at 50 °C for 5 min and then programmed to rise to 250 °C at a rate of 4 °C/min. Helium was used as the carrier gas at a flow rate of 1.1 mL/min. The injector and detector temperatures were set to 220 and 250 °C, respectively. The isolated peaks were identified by matching mass spectra with data in the National Institute of Standard and Technology library. Quantitation was carried out by peak area integration.
Determination of antioxidant activity
2,2-Diphenyl-1-picrylhydrazyl (DPPH) assay
The DPPH assay was performed according to a previously reported protocol27. The antioxidant activity was determined by a calibration curve prepared with ascorbic acid and expressed as mg of ascorbic acid equivalent (AAE)/mL of sample28.
2,2′-Azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) assay
The ABTS assay was performed as described previously29 and expressed in mMof Trolox equivalents (TE)/mL of extract. When the measured ABTS value was over the linear range of the standard curve, values of additional dilutions were recorded30.
Ferric reducing antioxidant power (FRAP) assay
The FRAP assay was performed according to the Benzie and Strain method, and expressed in mM TE/mL extract. Values of additional dilutions were measured when the FRAP value was over the linear range of the standard curve9.
Anticancer activity
Cell culture and treatments
Human liver cancer cell lineHepG2 was obtained from the American Type Culture Collection (Rockville, MD, USA). Cells were grown in RPMI-1640 medium supplemented with 10% foetal bovine serum, 1% MEM non-essential amino acid solution, and 1% penicillin streptomycin solution (10,000 units of penicillin and 10 mg of streptomycin in 0.9% (NaCl) in a humidified atmosphere having 5% CO2 at 35 °C. The passage number range was maintained between 20 and 25. The cells were cultured in 75 cm2 cell culture flasks. For experimental purposes, the cells were cultured in 96-well plates (0.2 mL of cell solution/well). The optimum cell concentration, as determined by the cell line growth profile, was 2 × 105 cells/mL. The cells were allowed to attach for 24 h prior to treatment with the extracts. The stock solution of each extract was filtered with 0.22 µm Minisart Filters (Sigma-Aldrich) before applying to cells. To test toxicity, 11 dilutions of each extract were tested by addition to cell monolayers washed with phosphate buffer saline (PBS)10.
3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay
The MTT assay was performed using the protocol described previously13. Briefly, the cells were incubated for 4 h with 0.8 mg/mL of MTT dissolved in serum-free medium. After washing with 1 mL PBS, 1 mL DMSO was added and gently shaken for 10 min for complete dissolution. Aliquots (200 μL) of the resulting solution were transferred into 96-well plates and absorbance was recorded at 560 nm using the SpectraMax® 190 Microplate Reader (Molecular Devices). The concentration required for 50% inhibition of cell viability (IC50) was calculated as described3.
Antiviral assay
Cells and viruses
Vero and BHK-21 cells were grown in Eagle’s minimal essential medium (MEM, GIBCO) containing 5% inactivated calf serum and 50 mg/mL gentamicin. Maintenance medium at pH 7.5 consisted MEM supplemented with 1.5% inactivated calf serum and gentamicin. The vesicular stomatitis virus (VSV) was propagated in Vero cells. Virus stocks were plaque-assayed on Vero cells as described31.
Cytotoxicity assay
Vero cells grown in 96-well tissue culture plates were incubated with 12 concentrations (μg/mL) of each extract for 24 h at 37 °C. Cell viability was measured with the MTT assay. The cytotoxicity of each extract was expressed as the 50% cytotoxic concentration (CC50), which is the concentration required to reduce cell viability to 50% of the control32.
Statistical analysis
The results are reported as mean ± standard deviation (SD) for at least three experiments. Statistical differences were analysed by the one-way ANOVA test.
Results
GC-MS analysis of FDSM
GC-MS analysis of extract of DSM fermented with A. fumigatus F-993(FDSM1) identified31 compounds, of which butylated hydroxytoluene (66.83%) and δ-myrcene (11.43%) were the main constituents (Table 1). In the GC-MS analysis of extract of DSM fermented with A. awamori FB-133 (FDSM2), 26 compounds were identified, of which 11,14-octadecadienoic acid and methyl ester (63.63%) was the main constituents (Table 2).
Antioxidant activity
The mean values of the antioxidant activity measured for DSM, FDSM1, and FDSM2 are presented in Table 3. The antioxidant activity of DSM extract was 3.362 ± 0.05 and 2.11 ± 0.02 mmol TE/mL as determined by ABTS and FRAPS assays, respectively, and 0.511 ± 0.01 mg AAE/mL with the DPPH assay. The antioxidant activity of FDSM2 extract was 4.763 ± 0.05 and 3.795 ± 0.03 mmolTE/mL with ABTS and FRAPS assays, respectively, and 0.625 ± 0.02 mg AAE/mL by DPPH assay. The antioxidant activity of FDSM1 extract was 4.331 ± 0.04 and 3.971 ± 0.02 mmolTE/mL by ABTS and FRAP assays, respectively, and 0.692 ± 0.01 mg AAE/mL with DPPH assay. Therefore, both FDSM extracts showed stronger antioxidant activity than the DSM extract.
Anticancer activity
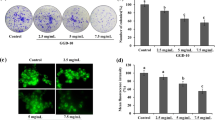
Colorimetric MTT assay was used to determine the extracts’ anticancer activity, utilizing human hepatic cancerous cell line “HepG2”. The cytotoxic effects of DSM, FDSM2, and FDSM1 extracts, expressed asIC50, are summarized in f 4. The IC50 values for FDSM2, FDSM1, and DSM extracts were 8.6, 16.88, and 27 μg/mL, respectively, indicating that the FDSM extracts were more cytotoxic than the DSM extract.
Antiviral activity
Table 4 also represents the titres of hepatitis A virus expressed as log10/0.1 mL. Extracts of FDSM1 showed stronger antiviral activity (2.66 log10/0.1 ml) compared to that of the DSM and FDSM2 extracts (5.5 and 3 log10/0.1 ml, respectively). The results of the present study revealed no significant differences in hepatitis A virus titres for FDSM1 and DSM extracts.
Discussion
Plant extracts contain many important phytoestrogens compounds, the most relevant to human health include: phytoestrogens genistein and daidzein (from soybean), formononetin (from clover), biochanin-A (from chickpeas) and coumestans and lignans (from flaxseed). However, the majority of such compounds are naturally found, in plants, in the glycosylated form. Moreover, the bioavailability of such glycoconjugates varies from their analogous unsubstituted aglycones. In mammals, it has been documented that phytoestrogens induce oestrogenic and anti-oestrogenic effects via their weak binding to the nuclear α and β oestrogen receptors (ER). In general, the relative binding affinity of phytoestrogens to β oestrogen receptors (ERβ) is higher than that to α oestrogen receptors (ERα). It has been reported that such phytoestrogens are associated with a lower rate of steroid-hormone-dependent cancers, such as colon, prostate and breast cancer.
It has been revealed that the role played by of polyphenols against flue virus, as antiviral mechanism, can be attributed to the inhibition of its adsorption to Madin-Darby canine kidney cells33, as well as the interference in viral membrane fusion28. It is a well-known fact that mechanism of oxidative stress, commonly associated viral infections including HSV-1, plays a major role in viral replication inside the viral-infected cells. Moreover, viral replication can be inhibited through restoring the intracellular redox conditions by the help of special antioxidants (e.g., glutathione “GSH” or n-butanoyl derivative “GSH-C4”) which inhibit HSV-1 replication34,35. In addition, Ramadan et al. 2014 have concluded that the main ingredients of the volatile compounds in DSM extracts are 23.8% (2,3-dicyano-7,7-dimethyl-5,6-benzodiene) and 19.1% (9-ditertbutyl-1-oxaspiro-4,5-deca-6,9-diene-2,8-dione)1.
It has been concluded that the major volatile antioxidant compounds, in FDSM, were butylated hydroxytoluene and 11,14-octadecadienoic acid, methyl ester. Octadecadienoic acid has been reported to be present in Michelia champaca Linn extracts and impart antioxidant and anticancer activities to the plant extract36. Specific food additives prevent lipid oxidation and rancidity, for example Butylated hydroxyl toluene “BHT”, which is the most commonly used synthetic antioxidant37. Studies have revealed that “BHT” has anticancer activities and tumour promotion effects. Moreover, its effect on other carcinogens depends on a number of factors including, the carcinogen, target organ, exposure parameters, as well as the animal being tested. The toxicity of butylated hydroxytoluene is suggested to be the result of it being an electrophilic metabolite38,39,40,41,42,43. Numerous studies have revealed that Butylated hydroxyl toluene “BHT” acts as a potent antioxidant that significantly inhibits cytokine-induced inflammatory responses in both human and mouse cells44.
Although, it has been concluded that the potency of modified ester of epigallocatechin gallate against HSV-1 infections, in vitro, is greater than that of epigallocatechin gallate, yet further studies are still recommended to fully explain the exact mechanism of action taking place in humans. Though, the use of the appropriate natural products could improve the health status of several patients via offering much better life. Murakami et al. 2003 have reported that bis-butylated hydroxyanisole inhibits the activation of activator protein-1, and hence it could be used in the chemoprevention of oral diseases, e.g. leukoplakia and destructive chronic periodontitis45.
The transformation of isoflavone glycosides into aglycones has been suggested to be promoted by soybean fermentation, hence fermented soybean is used as an ingredient in the formulation of foods with functional properties for health benefits44. By comparing with the results of earlier studies, it was clearly seen that these major compounds have major neuroprotective, antioxidant, anti-inflammatory, anticancer, hepatoprotective, and antimicrobial effects46,47,48,49,50. In conclusion, our results indicate that FDSMs possess antioxidant, anticancer and antiviral properties.
References
Ramadan, M. M., Elbandy, M. A., Fade, M. & Ghanem, K. Z. Biotechnological production of volatile and non-volatile antioxidant compounds from fermented soy bean meal with Trichodermasp. Res. J. Pharm. Biol. Chem. Sci. 5, 537–547 (2014).
Ghanem, Z. K., Ramadan, M. M., Ghanem, H. Z. & Fadel, M. Improving production of unsaturated fatty acid esters and flavonoids from date palm pollen and their effects as anti-breast cancer and anti-viral: In-vitro study. J. Arab Soc. Med. Res. 10, 46–55 (2015).
Yajie, W. & Huimin. Tandem Reactions Combining Biocatalysts and Chemical Catalysts for Asymmetric Synthesis. Catalysts. 6(12), 194 (2016).
Sivakumar, T. & Gajalakshmi, D. Phytochemical screening and GC-MS analysis of root extract from Asparagus racemosus L. Int. J. Pharmaceut. Sci. Res. 5, 1000–1005 (2014).
Zakir, M. & Freitas, I. Benefits to human health in consumption of isoflavones present in soybean products. Journal of bioenergy and food science 2(3), 107–116 (2015).
Kim, E. H. et al. Analysis of phenolic compounds and isoflavones in soybean seeds [Glycine max (L.) Merill] and sprouts grown under different conditions. Eur. Food Res. Technol. 222, 201–208 (2006).
Takemoto, K. et al. Effect of Aspergillus awamori-Fermented Burdock Root on Mouse Diabetes Induced by Alloxan—Prevention of Cell Apoptosis. Food and Nutrition Sciences 05(16), 1554–1560 (2014).
Kaminski, E., Stawicki, S. & Wasowicz. Volatile Flavor Compounds Produced by Molds of Aspergillus, Penicillium, and Fungi. imperfectiApplMicrobiol. 27(6), 1001–1004 (1974).
Wardhani, D. H., Vázquez, J. A. & Pandiella, S. S. Mathematical modelling of the development of antioxidant activity in soybeans fermented with Aspergillus oryzae and Aspergillus awamori in the solid state. J. Agric. Food Chem. 57, 540–544 (2009).
Chun, J. Y. et al. Conversion of isoflavone glucosides to aglycones in soymilk by fermentation with lactic acid bacteria. J. Food Sci. 72, M39–44 (2007).
Lin, C. H., Wei, Y. T. & Chou, C. C. Enhanced antioxidative activity of soybean koji prepared with various filamentous fungi. Food Microbiol. 23, 628–633 (2006).
Oh, D. et al. Systematic review of pediatric health outcomes associated with childhood adversity. BMC Pediatr. 18, 83 (2018).
Muruganantham, N., Solomon, S. M. & Senthamilselvi, M. Anti-cancer activity of Plumeriarubra (flowers) against human liver cancer. Int. J. Pharmacogn. Phytochem. Res. 6, 1007–1009 (2014).
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer Statistics, 2019. Ca Cancer J. Clin. 69, 7–34 (2019).
Wang, Y. & Zhao, H. Tandem Reactions Combining Biocatalysts and Chemical Catalysts for Asymmetric Synthesis. Catalysts. 6, 194 (2016).
Shad, A. et al. Phytochemical and Biological Activities of Four Wild Medicinal Plants. The Scientific World Journal 2014, 7 (2014).
Nersesyan, H. & Slavin, K. V. Current approach to cancer pain management: Availability and implications of different treatment options. Ther. Clin. Risk Manag. 3, 381–400 (2007).
DeFeudis, F. V., Papadopoulos, V. & Drieu, K. Ginkgo biloba extracts and cancer: a research area in its infancy. Fundam. Clin. Pharmacol. 17, 405–417 (2003).
Abd-Algader, N. N., El-Kamali, H. H., Ramadan, M. M., Ghanem, K. Z. & Farrag, A. R. H. Xylopiaaethiopica volatile compounds protect against panadol-induced hepatic and renal toxicity in male rats. World Appl. Sci. J. 27, 10–22 (2013).
Ramadan, M. M., Abd-Algader, N. N., El-kamali, H. H., Ghanem, K. Z. & Farrag, A. R. H. Chemopreventive effect of Coriandrumsativum fruits on hepatic toxicity in male rats. World J. Med. Sci. 8, 322–333 (2013).
Ramadan, M. M., Abd-Algader, N. N., El-kamali, H. H., Ghanem, K. Z. & Farrag, A. R. H. Volatile compounds and antioxidant activity of the aromatic herb Anethumgraveolens. J. Arab Soc. Med. Res. 8, 79–88 (2013).
Ramadan, M. M., Ali, M. M., Ghanem, K. Z. & El-Ghorab, A. H. Essential oils from Egyptian aromatic plants as anti-oxidant and novel agents in human cancer cell lines. GrasasAceites 66, e080, https://doi.org/10.3989/gya.0955142 (2015).
Bintsis, T. Foodborne pathogens. AIMS microbiology 3(3), 529–563 (2017).
Prabhakar, K. R., Srinivasan, K. K. & Rao, P. G. M. Chemical investigation, anti-inflammatory and wound healing properties of Coronopusdidymus. Pharmaceutical Biology 40 (7) 49–493 (2002).
Mesquita, J. R. & Nascimento, M. S. A foodborne outbreak of norovirus gastroenteritis associated with a Christmas dinner in Porto Portugal, December 2008. Euro Surveill. 14, 19355, https://doi.org/10.2807/ese.14.41.19355-en (2009).
Naga Vamsi Krishna, A., Venkata Raman, B., Kasetti Ramesh, B. & Chippada, A. Antioxidant activity and GC-MS analysis of Phragmytesvallatoria leaf ethanolicextract. Int. Res. J. Pharm. 3, 252–254 (2012).
Thaipong, K., Boonprakoba, U., Crosby, K., Cisneros, L. & Byrnec, D. H. Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J. Food Compos.Anal. 19, 669–675 (2006).
Imanishi, N. et al. Additional inhibitory effect of tea extract on the growth of influenza A and B viruses in MDCK cells. Microbiol. Immunol. 46, 491–494 (2002).
Arnao, M. B., Cano, A. & Acosta, M. The hydrophilic and lipophilic contribution to total antioxidant activity. Food Chem. 73, 239–244 (2001).
Malencic, D., Maksimovic, Z., Popovic, M. & Niladinovic, J. Polyphenol content and antioxidant activity of soybean seed extracts. Bioresour. Technol. 99, 6688–6691 (2008).
Mansouri, A., Embarek, G., Kokkalou, E. & Kefalas, P. Phenolic profile and antioxidant activity of the Algerian ripe date palm fruit (Phoenix dactylifera). Food Chem. 89, 121–127 (2005).
El-Ghorab, H. A., Ramadan, M. M. & Abd El-Moez, S. I. Essential oil, antioxidant, antimicrobial and anticancer activities of Egyptian Plucheadioscoridis extract. Res. J. Pharm. Biol. Chem. Sci. 6, 1255–1265 (2015).
Zhoua, Z. et al. Protocatechuic aldehyde inhibits hepatitis B virus replication both in vitro and in vivo. Antiviral Res. 74, 59–64 (2007).
De Chiara, G. et al. APP Processing Induced by Herpes Simplex Virus Type 1 (HSV-1) Yields Several APP Fragments in Human and Rat Neuronal Cell. PLOS ONE 5(11), e13989 (2010).
Mathew, O. P., Ranganna, K. & Yatsu, F. M. Butyrate, an HDAC inhibitor, stimulates interplay between different posttranslational modifications of histone H3 and differently alters G1-specific cell cycle proteins in vascular smooth muscle cells. Biomedicine & Pharmacotherapy. 64(10), 733–740 (2010).
Song, J. M., Lee, K. H. & Seong, B. L. Antiviral effect of catechins in green tea on influenza virus. Antiviral Res. 68, 66–74 (2005).
Hossain, M. M. et al. Antioxidant, analgesic and cytotoxic activity ofMicheliachampaca Linn. Leaf. Stamford J. Pharm. Sci. 2, 1–7 (2009).
Wanita, A. & Lorenz, K. Antioxidant potential of 5-N-pentadecylresorcinol. J. Food Process. Preserv. 20, 417–429 (1996).
Chetverikova, L. K. et al. Factors of antiviral resistance in the pathogenesis of influenza in mice. [Article in Russian] Vestn. Akad.Med.Nauk SSSR 11, 63–68 (1989).
Chetverikova, L. K. & Inozemtseva, L. I. Role of lipid peroxidation in the pathogenesis of influenza and search for antiviral protective agents. [Article in Russian] Vestn. Akad. Med. Nauk SSSR 3, 37–40 (1996).
Forni, C et al. Beneficial Role of Phytochemicals on Oxidative Stress and Age-Related Diseases. Biomed Research International. (2019).
Murakami, Y. et al. Preventive effect of ortho dimer of butylated hydroxyanisole on activator protein-1 activation and cyclooxygenase-2 expression in macrophages stimulated by fimbriae of Porphyromonasgingivalis, an oral anaerobe. Anticancer Research. 26(4B), 2915–2920 (2006).
Keith, A. D., Arruda, D., Snipes, W. & Frost, P. The antiviral effectiveness of butylated hydroxytoluene on herpes cutaneous infections in hairless mice. Proc. Soc. Exp. Biol. Med. 170, 237–244 (1982).
Murakami, Y., Ito, S., Atsumi, T. & Fujisawa, S. Theoretical prediction of the relationship between phenol function and COX-2/AP-1 inhibition for ferulic acid-related compounds. In Vivo 19, 1039–1044 (2005).
Murakami, Y., Shoji, M., Hanazawa, S., Tanaka, S. & Fujisawa, S. Preventive effect of bis-eugenol, a eugenol ortho dimer, on lipopolysaccharide-stimulated nuclear factor kappa B activation and inflammatory cytokine expression in macrophages. Biochem.Pharmacol. 66, 1061–1066 (2003).
Ramadan, M. M., El-Ghorab, A. H. & Ghanem, K. Z. Volatile compounds, antioxidants and anticancer activities of Cape gooseberry fruit (Physalisperuviana L.): In vitro study. J. Arab Soc. Med. Res. 10, 56–64 (2015).
Wadl, M. et al. Food-borne norovirus-outbreak at a military base, Germany, 2009. BMC Infect. Dis. 10, 30, https://doi.org/10.1186/1471-2334-10-30 (2010).
Zomer, T. P. et al. A foodborne norovirus outbreak at a manufacturing company. Epidemiol.Infect. 138, 501–506 (2010).
Bourinbaiar, A. S. & Jirathitikal, V. Low-cost anti-HIV compounds: potential application for AIDS therapy in developing countries.Curr.Pharm. Des. 9, 1419–1431 (2003).
Murakami, Y. et al. Dehydrodiisoeugenol, an isoeugenol dimer, inhibits lipopolysaccharide-stimulated nuclear factor kappa B activation and cyclooxygenase-2 expression in macrophages. Arch. Biochem. Biophys. 434, 326–332 (2005).
Acknowledgements
The authors really express their deep thanks and gratitude to the Scientific Research Deanship of King Saud University, Riyadh, Kingdom Saudi Arabia for their fruitful support through funding the present study.
Author information
Authors and Affiliations
Contributions
K.G., M.R., M.Z.M. and H.G. carried out the antioxidant analysis, GC-MS analysis, fermentation experiments, results preparation and participation in the manuscript drafting. M.F. carried out the anticancer and antiviral Analysis. K.G., M.R. and M.H.M. put the study design, performed the statistical analysis, and participated the manuscript drafting.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflicts of interest, state that the manuscript has not been published or submitted elsewhere, state that the work complies with the Ethical Policies of the Journal and state that the work has been conducted under internationally accepted ethical standards after relevant ethical review.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ghanem, K.Z., Mahran, M.Z., Ramadan, M.M. et al. A comparative study on flavour components and therapeutic properties of unfermented and fermented defatted soybean meal extract. Sci Rep 10, 5998 (2020). https://doi.org/10.1038/s41598-020-62907-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-62907-x
- Springer Nature Limited
This article is cited by
-
Bioactive metabolites in functional and fermented foods and their role as immunity booster and anti-viral innate mechanisms
Journal of Food Science and Technology (2023)