Abstract
Non-ampullary duodenal adenocarcinoma (NADC) is extremely rare. Little is known about its clinicopathological and molecular features or its management. Herein we retrospectively analyzed the cases of 32 NADC patients, focusing on microsatellite instability (MSI), genetic mutations, CpG island methylator phenotype (CIMP), and immunostaining including mucin phenotype and PD-L1 expression. The incidence of MSI, KRAS/BRAF/GNAS mutations and CIMP was 51.6%, 34.4%/3.1%/6.5% and 28.1%, respectively. PD-L1 expression was seen in 34.4% of patients. No significant associations between clinicopathological features and KRAS/BRAF/GNAS genetic mutations or CIMP were found. Histologically non-well-differentiated-type NADCs and those in the 1st portion of the duodenum were significantly associated with later stages (stages III–IV) (P = 0.006 and P = 0.003, respectively). Gastric-phenotype NADCs were frequently observed in the 1st portion and in late-stage patients; their cancer cells more frequently expressed PD-L1. Histologically, the non-well-differentiated type was an independent predictor of PD-L1 expression in cancer cells (OR 25.05, P = 0.04) and immune cells (OR 44.14, P = 0.02). Only late-stage disease (HR 12.23, P = 0.01) was a prognostic factor for worse overall survival in a Cox proportional hazards regression model. Our observation of high proportions of MSI and PD-L1 expression may prompt the consideration of immune checkpoint inhibitors as a new treatment option for NADCs.
Similar content being viewed by others
Introduction
Small bowel adenocarcinomas (SBAs) are rare, appearing at a rate of <1.0 per 100,000 people; when age-standardization is applied to the world population, the rate ranges from 0.3 to 2.0 per 100,000 people1. Duodenal adenocarcinoma (DAC), a type of SBA, represents <1% of all gastrointestinal (GI) cancers2, and the incidence of DAC is increasing worldwide3,4. Many studies regarding molecular events in SBAs and small intestinal adenomas have been reported5,6,7,8,9,10,11,12,13,14,15,16,17,18,19, but the associations among clinicopathological features, genetic/epigenetic alterations including CpG island methylation, and the phenotype (CIMP) and immunophenotype in DACs have not been clearly analyzed, unlike in the case of gastric and colorectal cancers (CRCs).
Because SBAs are so rare, little is known about their optimal management, including chemotherapy. No guidelines for the treatment of SBAs have been prepared by the U.S. National Comprehensive Cancer Network20. Clinical practice guidelines for the diagnosis, treatment and follow-up of SBA were recently published in France21. Although there have been four prospective studies on chemotherapy for SBA, no randomized trials of the efficacy of different chemotherapy regimens have been performed, and thus there is no established standard regimen for patients with unresectable or recurrent SBA4.
Le et al. reported that the microsatellite instability (MSI) status — defined as mismatch repair (MMR) deficiency — predicted the clinical benefit of immune checkpoint blockade in mainly CRCs, but they included only two cases of MMR-deficient SBAs in their analysis22. Several later studies investigated the association between the MSI status and the programmed cell death-ligand 1 (PD-L1) expression in GI cancers22,23,24,25,26,27. Immunotherapies using immune checkpoint inhibitors (ICIs) have demonstrated significant clinical benefits and a prolonged duration of response in subsets of patients with GI cancer22,23,24,25,26. Testing for biomarkers, including MSI and PD-L1, may therefore be necessary to broaden the identification of responders to ICI treatment and to achieve a better stratification of patients27,28.
We conducted the present study to retrospectively investigate the associations among the clinicopathological features, immunophenotype (including PD-L1 expression), and genetic or epigenetic alterations in non-ampullary duodenal adenocarcinomas (NADCs). We also evaluated whether those features — i.e., clinicopathological characteristics, immunophenotype, and/or molecular events — impact the survival of NADC patients.
Results
Clinicopathological characteristics in NADCs
The clinicopathological characteristics of the 32 patients with NADC are summarized in Table 1. The median age of the patients was 65.5 years (1st–3rd quartile 53–75 years), with women accounting for only 21.9% of the patients. The tumors were located mostly in the 2nd portion of the duodenum (65.6%), and the histologically well-differentiated-type adenocarcinoma was the most common type (75.0%). The tumor stages were 0–I (n = 18), II (n = 1), III (n = 9), and IV (n = 4).
Mixed gastric (G)-type NADCs were identified in 14 cases (43.8%), comprising 3 G-type and 11 GI-type NADCs. The following expressions were observed: human epidermal growth factor receptor type 2 (HER2) (n = 0, 0%), Das-1 (n = 24, 75.0%), and PD-L1 (n = 11, 34.4%). When we evaluated the PD-L1 expression in cancer cells and immune cells in the stroma separately, the expression rate was 18.8% (6 of 32) in cancer cells and 34.3% (11 of 32) in immune cells. There was no case in which PD-L1 was expressed exclusively in cancer cells. MMR deficiency was seen in 8 of 26 patients (28.6%).
Molecular alterations in the NADCs
Table 1 also shows the incidences of molecular events: 51.6% for MSI, 28.1% for CIMP and 34.4% for KRAS mutation. The incidences of BRAF and GNAS mutations were comparatively small. Insufficient amounts of DNA invalidated one MSI test and one GNAS mutation test. In the MSI analysis, a major pattern (as defined in the Methods section) was found in 8 of 31 patients (25.8%). Of the 11 (of 32; 34.4%) NADCs with KRAS mutations, GGT (Gly) changed to both GTT (Val) and GCT (Ala) (n = 1 case), both Val and CGT (Arg) (n = 3), both Ala and GAT (Asp) (n = 1), Asp (n = 2), AGT (Ser) (n = 1), Arg (n = 2), or Val (n = 1). BRAF mutation was detected in V600A in 1 patient: this NADC had MSI but did not have a KRAS mutation. GNAS mutations were detected in 2 cases: 1 case with c.602 G > A, and 1 case with c.602 G > G/A, both in codon 201 (R201H).
Associations among the clinicopathological features and the immunohistochemical and molecular analysis results
The histologically non-well-differentiated-type (i.e., the moderately and poorly differentiated types) and tumors in the 1st portion of the duodenum were more frequently identified in the late stages (stages III–IV) (P = 0.006 and P = 0.003) in association with PD-L1 expression in cancer cells (P < 0.0001 and P = 0.02) and immune cells (P = 0.001 and P = 0.09), respectively. The late tumor stages were significantly associated with mixed G-type (P = 0.09) and PD-L1 expression in immune cells (P = 0.02). Additionally, mixed G-type tended to relate to PD-L1 expression in cancer cells (P = 0.06) (Table 2). However, other factors, i.e., monoclonal antibody (mAb) Das-1 reactivity, MSI, CIMP, and KRAS mutations, were not associated with clinicopathological features (Suppl. Table S3).
PD-L1 expression was associated with histology (the non-well-differentiated type), tumor location (the 1st portion of the duodenum), late tumor stage, and mixed G-type. Although the number of PD-L1-positive cases was small, when we evaluated the expression in cancer cells and immune cells separately, only the non-well-differentiated type was a predictive factor of PD-L1 expression in both cancer cells (odds ratio [OR] 25.05, 95% confidence interval [CI]: 1.22–513.85, P = 0.04) and immune cells (OR 44.14, 95%CI: 1.96–995.97, P = 0.02) (Table 3).
Among the 19 NADCs with MSI, 7 cases (36.8%) stained positive for PD-L1 expression; there was no significant association between MSI and PD-L1 expression (P = 0.70, Suppl. Table S4).
Survival analysis
Seventeen of the 19 patients in early stages (stages 0–II) were alive without disease at the last contact (range 6–110 months; mean 48 months). Of the remaining 2 cases, one patient died suddenly 3 months after the last visit, and the other patient died from the disease. The patients with late-stage disease (stages III–IV, n = 13) were alive at last contact (n = 2) or died of NADC (n = 11).
The Kaplan-Meier survival analysis demonstrated that the NADCs of the histologically non-well-differentiated type (P < 0.0001), in the 1st portion of the duodenum (P = 0.002), at the late stages (P = 0.0001), with MSI (P = 0.09), and with PD-L1 expression in immune cells (P = 0.05) were associated with worse overall survival (OS) by the log-rank test (Fig. 1), while there were no significant associations between other clinicopathological or molecular features and OS (Table 4, Suppl. Fig. S4). In the multivariate Cox proportional hazards regression model, the late tumor stages (HR 12.23, 95%CI: 1.67–134.56, P = 0.01) were independently associated with worse OS (Table 4).
Discussion
Prior studies on molecular events in NADCs have focused on genetic events7,10,13,14,15,16,17,18,28, and there have been few studies evaluating epigenetic alterations6,9,12,16. There have also been no studies of the associations among clinicopathological, immunohistochemical (including PD-L1 expression) and molecular characteristics; our study is the first to explore these associations in NADC, although a single study evaluated the associations in SBA27.
Herein we observed that the NADCs of the histologically moderately and poorly differentiated type (i.e., the non-well-differentiated type) and those in the 1st portion of the duodenum were significantly associated with late tumor stages (stages III–V). Mixed G-type was frequently identified in the late stages. Several studies have shown that duodenal tumors with a G-type component are associated with high histological atypia, location in the 1st portion of the duodenum29,30,31, and reduced disease-free survival29. Therefore, taking into consideration the past and present findings, we speculated that mixed G-type NADCs of histologically non-well-differentiated type in the 1st portion may be more likely to progress. Our analyses also revealed that late tumor stages were independently associated with worse OS, confirming that tumor stage is the most important prognostic factor in SBAs4,7,11,32.
PD-L1 expression in NADCs has not been described other than in two studies of ampulla of Vater carcinoma and SBA19,27; according to the findings of those studies, PD-L1 was expressed in 26.9–44% of duodenal cancers (an incidence that is similar to our present result). Many studies of PD-L1 evaluated its expression in both neoplastic cells and immune cells19,27,33,34,35, revealing that PD-L1 is more frequently expressed in immune cells than in neoplastic cells. Our present findings showed that there was no positivity of PD-L1 in cancer cells without positivity in immune cells, as in previous reports27,33,34.
The MSI rate in our study was higher (51.6%) than the reported rates in SBAs (7.6–33.3%)5,7,8,11,13,14,18,19. One of the explanations for this discrepancy may be differences in the methods of MSI analysis—i.e., differences in the immunohistochemistry for MMR proteins, the method of analysis (either polyacrylamide/urea gel electrophoresis following silver staining or next-generation sequencing), and the number and location of MSI markers evaluated. We identified MMR deficiency in 28.6% of the patients in our series, which is similar to the incidence of the major pattern (25.8%, 8 of 31) in our MSI analysis (as described in the Methods section below). The high-resolution fluorescent microsatellite analysis (HRFMA) assay used in the present study allows for a more accurate assessment of phenomena such as the allele shift pattern (minor pattern) compared to previous formats36,37. It might therefore be impossible to detect the minor pattern in MSI by using other methods36,37. In the future, it will be necessary to establish the definition of MSI in the HRFMA assay and other procedures. This lack of an established definition may be another cause of the discrepancy in the incidence between MSI and MMR deficiency in our study, although the incidence obtained by these two tests is considered to be similar in GI cancers7,38.
In general, the MSI tumor microenvironment strongly expresses several immune checkpoint ligands (including PD-L1), indicating that the active immune microenvironment of the tumors is counterbalanced by immune inhibitory signals that resist tumor elimination39. There are several reports regarding the association between MSI and PD-L1 expression in SBAs and other GI cancers19,22,27,28,33,35,39,40; some of these reports found a positive correlation between MSI and PD-L1 expression19,22,28,39 in most of GI cancers examined, except in SBA in one study28.
The lack of a positive association between MSI and PD-L1 expression in NADC in our present report (Suppl. Table S3) is thus in agreement with a recent report on some GI cancers including SBAs28. Generally, MSI cancers are associated with a higher mutational burden and tumor neoantigen load; these tumors provoke an antitumor immune response by dense immune cell infiltration, and thereby exhibit heightened sensitivity to ICIs22,39. Therefore, taking into account both these studies and our present findings, ICIs could be a new treatment option for NADCs that have MSI and/or are positive for PD-L1, as is the case in other cancers22,23,24.
In regard to the CIMP in SBAs, including DACs, there have been only a few studies8,9,16. Incidences of 26.6–29.7% were seen in those studies; our current result (28.1%) is within that range. However, CIMP was associated with MSI in those studies, contrary to our present finding. According to the studies analyzing duodenal adenomas and DACs9,12, CIMP is an early event in tumorigenesis and may be lost in later stages of tumor progression; alternatively, advanced-stage DACs may arise from different mechanisms.
It has also been reported that patients with CIMP-positive DACs showed worse OS than CIMP-negative cases9. We did not observe this association in the present study. The discrepancy may be due in part to differences in the analysis methods of CIMP and MSI mentioned above and in our previous investigations33,34,35,37.
The KRAS mutation rate we observed among 32 NADCs (34.4%) was in line with those in previous reports9,10,11,13,15,16 except for one study (11%)14. Fu et al. reported that the KRAS G > A mutation correlated significantly with late-stage disease and poor tumor differentiation10. In our present investigation however, there were only two cases with KRAS G > A mutation: the well-differentiated type and stage 0 in one case and the moderately differentiated type and stage III in the other. In addition, KRAS mutation was not related to the mucin phenotype as reported by Matsubara et al.15.
There have been only a few reports of BRAF and GNAS mutations in NADCs, and BRAF and GNAS mutations were as rare in our patients as in those studies13,14,15,17. Warth et al. showed that BRAF mutations were associated with SBAs with CIMP, but they did not separately analyze NADC data8. Another interesting study reported that GNAS mutations are more common in DACs with the G-type phenotype15. We did not observe this association in our present analyses; further studies with larger sample sizes are needed to clarify the reason for this lack of association.
We previously reported that all SBAs (8 of 8) reacted highly with the mAb Das-141, which is in agreement with our present findings for NADCs. Das-1 specifically reacts with the colonic epithelium and not with enterocytes (including goblet cells) from the jejunum or ileum42. The high reactivity of Das-1, a unique epitope related to colonic metaplasia, may therefore indicate that there is a phenotypic change of small intestinal enterocytes to colonocytes in NADCs. We detected no HER2 expression, as was the case in previous studies4,11. However, two studies that evaluated a large number of SBCs identified HER2 genomic alteration in 8.4–9.5% of their patients13,18.
It has generally been considered that in gastric cancer and CRC, tumors with MSI have better survival than those without43,44, but our present findings contradict that assumption. The association between MSI and overall survival in DAC remains uncertain. Overman et al. provided evidence of an improved prognosis in a subset of patients with a deficient expression of MMR proteins, which indicates MSI7, whereas Aparicio et al. reported that DAC with MMR deficiency was associated with longer OS11. Future investigations with larger sample sizes are needed to more clearly elucidate the relationship between MSI and survival in NADC patients.
The present study has some limitations. First, this was a retrospective analysis of a relatively small number of NADC patients, particularly considering that several different cellular phenotypes and molecular markers were evaluated. Second, we did not evaluate the CD8+ T-cell density in intratumoral or immune stroma. It was recently reported that increasing CD8+ densities in both tumors and immune stroma were associated with increasing percentages of tumor and stromal PD-L1 expression, indicating adaptive immune resistance45. An additional study is needed to evaluate the integration of biomarkers such as MSI and PD-L1 with CD8+ tumor-infiltrating lymphocytes.
In conclusion, although the number of NADC cases investigated was small (n = 32), our results suggest that in NADCs, genetic (KRAS/BRAF/GNAS mutations) and epigenetic alterations (CIMP) are not involved in the clinicopathological characteristics. However, MSI was more frequently observed and was significantly associated with clinical behavior, and the histology and tumor location were involved in the late tumor stage. Notably, histologically non-well-differentiated-type NADCs were an independent predictor of PD-L1 expression in both cancer and immune cells, but MSI was not. Since NADCs have molecular alterations that are different from those in gastric cancer and CRC, other therapeutic strategies may be necessary. Our results may thus indicate that ICIs could be a promising novel treatment for NADCs.
Patients, Materials, and Methods
Patients
Thirty-two consecutive patients with NADC treated at Hyogo College of Medicine Hospital or Aichi Cancer Center Hospital between April 2009 and March 2016 were enrolled in this study. There were no patients with a family history of cancer. Formalin-fixed, paraffin-embedded (FFPE) tissue blocks from patients who had been biopsied or resected by endoscopy or surgery for stage 0–IV NADC were obtained. The tissue sections from all cases in this study were reviewed by expert GI pathologists (S.H. and Y.Y.).
Consent and institutional review board approval
The study was approved by the Institutional Review Boards of Hyogo College of Medicine (No. Rin-Hi 315) and Aichi Cancer Center Hospital (No. 2016-1-090). This trial was registered with the UMIN Clinical Trials Registry (No. UMIN000023622). The informed consent of each patient was obtained by the opt-out procedure or as written informed consent, according to the procedure described in the study protocol (Rin-Hi 315 and 2016-1-090). The study was performed in accordance with the Declaration of Helsinki.
Clinicopathological and tumor evaluations
We evaluated the patients’ clinical data for both demographics (age and sex) and tumor characteristics (histology and tumor location). Their NADCs were staged using the combined American Joint Committee on Cancer/International Union Against Cancer (AJCC/UICC) TNM staging system46.
Immunohistochemistry
FFPE tissue blocks were cut into 4-µm-thick tissue sections and subjected to both hematoxylin and eosin staining and immunohistochemical staining. For immunostaining, the avidin-biotin peroxidase complex method was used to detect MUC5AC (CLH2; Agilent Technologies, Santa Clara, CA), MUC6 (CLH5, 1:50; Leica Biosystems, Newcastle, UK), MUC2 (clone Ccp58; Agilent Technologies), and CD10 (clone 56C6; Agilent Technologies). Immunohistochemical staining for gastric phenotype markers (MUC5AC and MUC6) and intestinal phenotype markers (MUC2 and CD10) was considered positive when distinct staining was observed in >10% of the cancer cells, as described previously29.
On the basis of the mucin histochemistry, the NADCs were classified into three subtypes: (1) gastric phenotype (G-type), (2) gastric and intestinal phenotype (GI-type), and (3) intestinal phenotype (I-type)29. NADCs that showed immunoreactivity for either the G-type or the G-I type were defined as mixed G-type.
HER2 testing was performed for selected cases using the HER2 monoclonal antibody (mAb) (4B5; Ventana Medical Systems, Tucson, AZ) with an automated slide stainer (BenchMark XT; Ventana Medical Systems) per the manufacturer’s protocol. The staining for HER2 was graded according to the guideline for HER2 testing in cancer cells, with 2+ or 3+ membranous staining in >10% of the tumor cells counted as positive47.
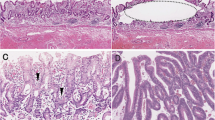
The mAb Das-1, which is highly specific against the colonic phenotype, is considered clinically useful in identifying small intestinal adenomas at “high risk” for malignancy41. Therefore, mAb Das-1 staining was also performed using sensitive immunoperoxidase assays, as described in previous reports41,48,49,50. Positive expression was defined as >10% of tumor cells staining for mAb Das-149,50 (Fig. 2C).
Staining of the serial sections of NADCs. (A) Hematoxylin and eosin (H&E) staining shows moderately differentiated-type adenocarcinoma (×200). NADC strongly reacted with MUC5AC (B, ×200) and the mAb Das-1 (C, ×200). (D) H&E staining revealed well differentiated-type adenocarcinoma. (E) Inset at the upper left: PD-L1 staining of (D) (×200). PD-L1 is expressed in cancer cells, as outlined in part by a red square (×400). (F) Immune cells were also positive for PD-L1 expression (×400).
For the PD-L1 staining, sections were retrieved in EDTA buffer (pH 8.0) at 98 °C for 20 min. The immunostaining used a mAb against the cytoplasmic domain of PD-L1 (clone E1L3N, dilution 1:400; Cell Signaling Technology, Danvers, MA), and the reactivity was evaluated separately for cancer cells and infiltrating immune cells. PD-L1 positivity was defined as positive cell staining of any intensity on ≥1% of the cell membrane. Cytoplasmic staining was not considered in this study24,33 (Fig. 2E,F).
The expression of MMR proteins such as MLH1 (ES05, 1:50), MSH2 (79H11, 1:50), MSH6 (PU29, 1:40) and PMS2 (M0R4G, 1:10; Leica Biosystems) was also examined. Regarding the criteria for MMR protein expression, if nuclear staining was identified in the cancerous cells, the lesion was defined as positive for that marker. Positive staining for all of these proteins was regarded as proficient MMR, and negative staining for any of these four proteins was regarded as deficient MMR51. In this study, four cases—i.e., two biopsy samples and two endoscopically resected specimens—could not be evaluated by the immunohistochemistry of MMR proteins because of an insufficient amount of material.
DNA extraction
Two or three 7-µm-thick tissue sections were cut for DNA extraction for a molecular analysis. DNA was extracted from the NADC and non-neoplastic normal mucosa using a QIAamp DNA Micro Kit (Qiagen, Hilden, Germany). The NADC and normal mucosa were isolated using a PALM MicroBeam LCM system (Microlaser Technologies, Munich, Germany) as described48,49,50.
Analysis of MSI by HRFMA
For the high-resolution fluorescent microsatellite analysis (HRFMA) following reported methods36,37,48,49,50,51, we examined the following five microsatellite loci on chromosomes for MSI based on the revised Bethesda panel52: BAT26, BAT25, D2S123, D5S346, and D17S250. The MSI status was judged as described previously36,37,49,50,51. There were two MSI patterns: (1) unequivocal extra peak bands in tumor DNA that differed by a multiple of two base pairs (bp) in dinucleotide markers or one bp in mononucleotide markers from DNA in normal DNA (minor pattern) (Suppl. Fig. S1A), and (2) the appearance of a large number of additional alleles in the tumor DNA (major pattern) (Suppl. Fig. S1B,C), as described previously36,37.
In cases in which MSI and loss of heterozygosity were indistinguishable53, we calculated the allelic imbalance (AI) ratio. We considered MSI as positive when the AI ratio (normal allele 1: normal allele 2 or tumor allele 1: tumor allele 2) was <0.67 or >1.35, as reported previously49,50,51 (Suppl. Fig. S1D). The lesions were defined as having MSI when unstable loci were observed in two or more of the five investigated markers37,49,50,51. One sample in the MSI test could not be analyzed due to an insufficient amount of DNA.
KRAS, BRAF, and GNAS mutation analysis
KRAS mutations in codons 12 and 13 were analyzed using a Mutector™ mutation detection kit (TrimGen, Sparks, MD) (Suppl. Fig. S2A), and the analysis of BRAF mutations in V600 was performed using a Mutector™ II (TrimGen) kit as reported previously51 (Suppl. Fig. S2B). These processes, involving a series of mutation analyses, were performed in accordance with the manufacturer’s instructions. GNAS mutation in exon 8 was also analyzed.
The polymerase chain reaction (PCR) products were electrophoresed on a 2% (w/v) agarose gel and recovered using a QIAquick Gel Extraction Kit (Qiagen). The isolated PCR products were sequenced using a Genetic Analyzer (3130xl; Applied Biosystems, Foster, CA) (Suppl. Fig. S2C). One GNAS mutation case could not be analyzed due to an insufficient amount of DNA.
Sodium bisulfite modification of DNA and CIMP markers
As in earlier studies48,49,50,51, purified DNA samples were chemically modified by sodium bisulfite with an EpiTect® Fast Bisulfite Kit (Qiagen). The bisulfite-modified DNA was amplified using primer pairs that specifically amplify the methylated or unmethylated sequences of several genes/loci related to carcinogenesis, including CDH1, CDKN2A, MLH1, MINT1, MINT31, MGMT, and RUNX3. These genes were used as CIMP markers.
Methylation-sensitive high-resolution melting analyses
We performed a methylation-sensitive high-resolution melting (MS-HRM) analysis as described previously48,49,50,51. Briefly, PCR amplification and the MS-HRM analysis were performed using a LightCycler® 480 System II (Roche, Mannheim, Germany). The primer sequences of all genes for the methylated and unmethylated forms and the PCR and MS-HRM conditions are summarized in Supplementary Tables S1 and S2. The percentages of methylation (0%, 10%, 50%, and 100%) were used to draw the standard curve (Suppl. Fig. S3). In this study, only samples with >10% methylation were considered methylated48,49,50,51. Samples in which ≥3 of the 7 CIMP panel markers were methylated were considered positive for CIMP49,50,51.
Statistical analyses
Categorical variables are presented as numbers and percentages and were compared by the chi-square test between groups or Fisher’s exact test when appropriate. Continuous variables are expressed as the median and interquartile range and were compared by the Kruskal-Wallis test or Mann-Whitney U-test between groups. The results of univariate analyses of the clinicopathological features, immunophenotype and genetic or epigenetic alterations (including CIMP) were evaluated using logistic regression models to calculate crude ORs and 95%CIs. We used multivariate logistic regression models with forward variable selections to calculate adjusted ORs for significant factors when a P-value < 0.1 was identified in the univariate analysis. Differences at P < 0.05 were considered significant.
We constructed OS curves using the Kaplan-Meier method, and we used the log-rank test to evaluate the statistical significance of differences and associations with each clinicopathological or molecular marker. The Cox proportional hazards regression model was used to assess the predictive effects of multiple covariates (including histology, tumor stage, immunohistochemical results, and molecular events) on the OS simultaneously. All statistical analyses were performed using JMP Pro 13 (SAS Institute, Cary, NC) and SPSS 22.0 (SPSS, Chicago, IL).
References
Schottenfeld, D., Beebe-Dimmer, J. L. & Vigneau, F. D. The epidemiology and pathogenesis of neoplasia in the small intestine. Ann. Epidemiol. 19, 58–69 (2009).
Cloyd, J. M., George, E. & Visser, B. C. Duodenal adenocarcinoma: Advances in diagnosis and surgical management. World J. Gastrointest. Surg. 8, 212–221 (2016).
Qubaiah, O., Devesa, S. S., Platz, C. E., Huycke, M. M. & Dores, G. M. Small intestinal cancer: A population-based study of incidence and survival patterns in the United States, 1992 to 2006. Cancer Epidemiol. Biomarkers Prev. 19, 1908–1918 (2010).
Raghav, K. & Overman, M. J. Small bowel adenocarcinomas–Existing evidence and evolving paradigms. Nat. Rev. Clin. Oncol. 10, 534–544 (2013).
Planck, M. et al. Microsatellite instability and expression of MLH1 and MSH2 in carcinomas of the small intestine. Cancer 97, 1551–1557 (2003).
Kim, S. G. et al. Epigenetic and genetic alterations in duodenal carcinomas are distinct from biliary and ampullary carcinomas. Gastroenterology 124, 1300–1310 (2003).
Overman, M. J. et al. Immunophenotype and molecular characterisation of adenocarcinoma of the small intestine. Br. J. Cancer 102, 144–150 (2010).
Warth, A., Kloor, M., Schirmacher, P. & Bläker, H. Genetics and epigenetics of small bowel adenocarcinoma: The interactions of CIN, MSI, and CIMP. Mod. Pathol. 24, 564–570 (2011).
Fu, T. et al. CpG island methylator phenotype-positive tumors in the absence of MLH1 methylation constitute a distinct subset of duodenal adenocarcinomas and are associated with poor prognosis. Clin. Cancer Res. 18, 4743–4752 (2012).
Fu, T. et al. KRAS G > A mutation favors poor tumor differentiation but may not be associated with prognosis in patients with curatively resected duodenal adenocarcinoma. Int. J. Cancer 132, 2502–2509 (2013).
Aparicio, T. et al. Small bowel adenocarcinoma phenotyping, a clinicobiological prognostic study. Br. J. Cancer 109, 3057–3066 (2013).
Sun, L. et al. CpG island methylator phenotype and its association with malignancy in sporadic duodenal adenomas. Epigenetics 9, 738–746 (2014).
Laforest, A. et al. ERBB2 gene as a potential therapeutic target in small bowel adenocarcinoma. Eur. J. Cancer 50, 1740–1746 (2014).
Kumagai, R. et al. Mucinous phenotype and CD10 expression of primary adenocarcinoma of the small intestine. World J. Gastroenterol. 21, 2700–2710 (2015).
Matsubara, A. et al. Activating GNAS and KRAS mutations in gastric foveolar metaplasia, gastric heterotopia, and adenocarcinoma of the duodenum. Br. J. Cancer 112, 1398–1404 (2015).
Fu, T. et al. Methylation of MGMT is associated with poor prognosis in patients with stage III duodenal adenocarcinoma. PLoS One 11, e0162929 (2016).
Hida, R. et al. Duodenal neoplasms of gastric phenotype: An immunohistochemical and genetic study with a practical approach to the classification. Am. J. Surg. Pathol. 41, 343–353 (2017).
Schrock, A. B. et al. Genomic profiling of small-bowel adenocarcinoma. JAMA Oncol. 3, 1546–1553 (2017).
Saraggi, D. et al. PD-L1 overexpression in ampulla of Vater carcinoma and its pre-invasive lesions. Histopathology 71, 470–474 (2017).
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Colon Cancer. ver. 3. 2018–August 7, 2018. NCCN.org.
Locher, C. et al. Small bowel adenocarcinoma: French intergroup clinical practice guidelines for diagnosis, treatments and follow-up (SNFGE, FFCD, GERCOR, UNICANCER, SFCD, SFED, SFRO). Dig. Liver Dis. 50, 15–19 (2018).
Le, D. T. et al. PD-1 blockade in tumors with mismatch-repair deficiency. N. Engl. J. Med. 372, 2509–2520 (2015).
Muro, K. et al. Pembrolizumab for patients with PD-L1-positive advanced gastric cancer (KEYNOTE-012): A multicentre, open-label, phase 1b trial. Lancet Oncol. 17, 717–726 (2016).
Overman, M. J. et al. Nivolumab in patients with metastatic DNA mismatch repair-deficient or microsatellite instability-high colorectal cancer (CheckMate 142): An open-label, multicentre, phase 2 study. Lancet Oncol. 18, 1182–1191 (2017).
Fuchs, C. S. et al. Safety and efficacy of pembrolizumab monotherapy in patients with previously treated advanced gastric and gastroesophageal junction cancer: Phase 2 Clinical KEYNOTE-059 Trial. JAMA Oncol. 4, e180013 (2018).
Shitara, K. et al. KEYNOTE-061 investigators. Pembrolizumab versus paclitaxel for previously treated, advanced gastric or gastro-oesophageal junction cancer (KEYNOTE-061): A randomised, open-label, controlled, phase 3 trial. Lancet 392, 123–133 (2018).
Pedersen, K., Smyrk, T. C., Harrington, S. & McWilliams, R. R. Programmed death-ligand 1 (PD-L1) expression in small bowel adenocarcinomas (SBA). J. Clin. Oncol. 33 (suppl; abstr 3619), 3619–3619 (2015).
Salem, M. E. et al. Landscape of tumor mutation load, mismatch repair deficiency, and PD-l1 expression in a large patient cohort of gastrointestinal cancers. Mol. Cancer Res. 16, 805–812 (2018).
Ushiku, T., Arnason, T., Fukayama, M. & Lauwers, G. Extra-ampullary duodenal adenocarcinoma. Am. J. Surg. Pathol. 38, 1484–1493 (2014).
Mitsuishi, T. et al. Clinicopathological characteristics of duodenal epithelial neoplasms: Focus on tumors with a gastric mucin phenotype (pyloric gland-type tumors). PLoS One 12, e0174985 (2017).
Hijikata, K., Nemoto, T., Igarashi, Y. & Shibuya, K. Extra-ampullary duodenal adenoma: A clinicopathological study. Histopathology 71, 200–207 (2017).
Halfdanarson, T. R., McWilliams, R. R., Donohue, J. H. & Quevedo, J. F. A single-institution experience with 491 cases of small bowel adenocarcinoma. Am. J. Surg. 199, 797–803 (2010).
Valentini, A. M. et al. PD-L1 expression in colorectal cancer defines three subsets of tumor immune microenvironments. Oncotarget 9, 8584–8596 (2018).
Kim, J. H., Park, H. E., Cho, N. Y., Lee, H. S. & Kang, G. H. Characterisation of PD-L1-positive subsets of microsatellite-unstable colorectal cancers. Br. J. Cancer 115, 490–496 (2016).
Cho, J. et al. Four distinct immune microenvironment subtypes in gastric adenocarcinoma with special reference to microsatellite instability. ESMO Open 3, e000326 (2018).
Tokunaga, E. et al. Frequency of microsatellite instability in breast cancer determined by high-resolution fluorescent microsatellite analysis. Oncology 59, 44–49 (2000).
Shibata, N. et al. Cell kinetics and genetic instabilities in differentiated type early gastric cancers with different mucin phenotype. Hum. Pathol. 34, 32–40 (2003).
Smyth, E. C. et al. Mismatch repair deficiency, microsatellite instability, and survival: An exploratory analysis of the Medical Research Council Adjuvant Gastric Infusional Chemotherapy (MAGIC) Trial. JAMA Oncol. 3, 1197–1203 (2017).
Llosa, N. J. et al. The vigorous immune microenvironment of microsatellite instable colon cancer is balanced by multiple counter-inhibitory checkpoints. Cancer Discov. 5, 43–51 (2015).
Lee, L. H. et al. Patterns and prognostic relevance of PD-1 and PD-L1 expression in colorectal carcinoma. Mod. Pathol. 29, 1433–1442 (2016).
Onuma, E. K. et al. A phenotypic change of small intestinal epithelium to colonocytes in small intestinal adenomas and adenocarcinomas. Am. J. Gastroenterol. 96, 2480–2485 (2001).
Das, K. M., Sakamaki, S., Vecchi, M. & Diamond, B. The production and characterization of monoclonal antibodies to a human colonic antigen associated with ulcerative colitis: Cellular localization of the antigen by using the monoclonal antibody. J. Immunol. 139, 77–84 (1987).
Popat, S., Hubner, R. & Houlston, R. S. Systematic review of microsatellite instability and colorectal cancer prognosis. J. Clin. Oncol. 23, 609–618 (2005).
Polom, K. et al. Meta-analysis of microsatellite instability in relation to clinicopathological characteristics and overall survival in gastric cancer. Br. J. Surg. 105, 159–167 (2018).
Thompson, E. D. et al. Patterns of PD-L1 expression and CD8 T cell infiltration in gastric adenocarcinomas and associated immune stroma. Gut 66, 794–801 (2017).
Brierley, J. D., Gospodarowicz, M. K. & Wittekind, C. (eds). TNM Classification of Malignant Tumours. 8th edn. (Wiley Blackwell, 2017).
Wolff, A. C. et al. Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. Arch. Pathol. Lab. Med. 138, 241–256 (2014).
Kawanaka, M. et al. Effects of Helicobacter pylori eradication on the development of metachronous gastric cancer after endoscopic treatment: Analysis of molecular alterations by a randomised controlled trial. Br. J. Cancer 114, 21–29 (2016).
Michigami, Y. et al. Effects of long-term aspirin use on molecular alterations in precancerous gastric mucosa in patients with and without gastric cancer. Sci. Rep. 7, 13384 (2017).
Michigami, Y. et al. Long-term effects of H. pylori eradication on epigenetic alterations related to gastric carcinogenesis. Sci. Rep. 8, 14369 (2018).
Nando, Y. et al. Genetic instability, CpG island methylator phenotype and proliferative activity are distinct differences between diminutive and small tubular adenoma of the colorectum. Hum. Pathol. 60, 37–45 (2016).
Umar, A. et al. Revised Bethesda Guidelines for hereditary nonpolyposis colorectal cancer (Lynch syndrome) and microsatellite instability. J. Natl. Cancer Inst. 96, 261–268 (2004).
Bryś, M. et al. Diagnostic value of DNA alteration: Loss of heterozygosity or allelic imbalance-promising for molecular staging of prostate cancers. Med. Oncol. 30, 391 (2013).
Acknowledgements
We thank Ms. Mayumi Yamada for the excellent technical assistance. This study was supported by funding from Ohtsuka Pharm, Astellas Academic Support (Astellas Pharm. Inc.), EA Pharma Research Support (EA Pharma Co., Ltd.) and a research grant (U.S. National Institute of Diabetes and Digestive and Kidney Disease, RO1DK63618 to K.M.D.) from the U.S. National Institutes of Health (Bethesda, MD).
Author information
Authors and Affiliations
Contributions
J.W. and S.M. designed the study and analyzed the data; J.W. and C.I. recruited the patients, performed the DNA extraction, molecular analysis, and immunohistochemistry and analyzed the data; J.W., S.M., C.I., K.T., T.T., T.O., H.F., S.K., S.N., Y.S., M.T., K.H., Y.Y., Y.S., K.M. and S.H. collected the materials; J.W., C.I., and T.M. performed the statistical analyses; S.H. judged the immunohistochemical staining. K.M.D. provided the mAb Das-1 and revised the manuscript; J.W. wrote the manuscript; S.H., T.M., K.M.D. and H.M. revised the manuscript. All authors read and approved the final version of the paper.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Watari, J., Mitani, S., Ito, C. et al. Molecular alterations and PD-L1 expression in non-ampullary duodenal adenocarcinoma: Associations among clinicopathological, immunophenotypic and molecular features. Sci Rep 9, 10526 (2019). https://doi.org/10.1038/s41598-019-46167-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-46167-y
- Springer Nature Limited
This article is cited by
-
Microsatellite instability in the high-grade dysplasia component of duodenal adenoma is associated with progression to adenocarcinoma
Surgery Today (2023)
-
Mixed adenoneuroendocrine carcinoma of the non-ampullary duodenum with mismatch repair deficiency: a rare case report
Medical Molecular Morphology (2022)
-
Clinicopathological features of tumor mutation burden, Epstein-Barr virus infection, microsatellite instability and PD-L1 status in Chinese patients with gastric cancer
Diagnostic Pathology (2021)
-
APC mutations are common in adenomas but infrequent in adenocarcinomas of the non-ampullary duodenum
Journal of Gastroenterology (2021)
-
Beyond bevacizumab: a review of targeted agents in metastatic small bowel adenocarcinoma
Medical Oncology (2020)