Abstract
Ventilator-associated pneumonia (VAP) is a serious complication of mechanical ventilation leading to high morbidity and mortality among intubated neonates in neonatal intensive care units (NICUs). Endotracheal tube (ETT) biofilm flora were considered to be responsible for the occurrence of VAP as a reservoir of pathogens. However, regarding neonates with VAP, little is known about the complex microbial signatures in ETT biofilms. In the present study, a culture-independent approach based on next generation sequencing was performed as an initial survey to investigate the microbial communities in ETT biofilms of 49 intubated neonates with and without VAP. Our results revealed a far more complex microflora in ETT biofilms from intubated neonates compared to a previous culture-based study. The abundance of Streptococci in ETT biofilms was significantly related to the onset of VAP. By isolating Streptococci in ETT biofilms, we found that Streptococci enhanced biofilm formation of the common nosocomial pathogen Pseudomonas aeruginosa PAO1 and decreased IL-8 expression of airway epithelia cells exposed to the biofilm conditioned medium of PAO1. This study provides new insight into the pathogenesis of VAP among intubated neonates. More studies focusing on intubated neonates are warranted to develop strategies to address this important nosocomial disease in NICUs.
Similar content being viewed by others
Introduction
Ventilator-associated pneumonia (VAP) is a serious complication related to mechanical ventilation in neonatal intensive care units (NICUs), and it accounts for increased health care costs, prolonged hospital stays and hospital mortality1. It ranks as the second most common nosocomial infection in pediatric intensive care units2 and is the leading cause of death among nosocomial infections in ventilated patients3, 4. The burden of VAP in NICUs is much more serious in developing countries than in developed countries1, 5. According to a recent meta-analysis conducted in China, the incidence and case fatality rates of VAP in NICUs were 42.8% and 16.4%, respectively5. Therefore, VAP poses a major challenge to public health and has considerable health and economic consequences in China.
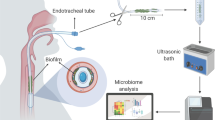
During ventilation, the endotracheal tube (ETT) provides a direct conduit for pathogenic bacteria to enter the normally sterile lower respiratory tract and lung parenchyma6. Remarkably, bacteria can adhere and form a biofilm on the surface of the ETT soon after intubation6,7,8. These biofilms are complex communities of microbes that produce a matrix of hydrated extracellular polymeric substances that mainly include polysaccharides, proteins, nucleic acids and lipids9. Biofilm cells can continuously access the lower airway through ventilator gas flow and aspiration8. Mounting evidence shows that biofilms on ETTs serve as significant and persistent reservoirs of pathogens to cause VAP7, 10, 11. Strategies involving modified ETTs to prevent or remove ETT biofilms were proven to reduce VAP occurrence in adults, including cuffed ETTs and silver or other nanoparticle coated ETTs12, 13. However, for intubated neonates, the CDC suggests that cuffed ETTs are not available and the safety of nanoparticle coated ETTs for neonates is uncertain1, 12; other strategies, therefore, are warranted to address ETT biofilms among ventilated neonates.
In addition to the ETT shape and material, bacterial factors should also be highlighted, considering that biofilms are generated by bacteria. The biofilm formation ability differs between bacterial species and is influenced by bacterial members both synergistically and antagonistically14. Therefore, identifying complex microbial members in biofilms is essential; yet, studies on biofilm bacterial populations among intubated neonates are limited in the literature. A previous study conducted among 29 intubated neonates demonstrated the predominance of Staphylococcus species and normal flora, such as Streptococcus viridans, in ETT biofilms based on a culture-dependent approach15. However, the culture-dependent approach showed a significant culturing bias due to the failure to recover and grow all of the bacteria in the ETT biofilm16. Culture-independent approaches are available to identify microorganisms, including fastidious or nonculturable organisms in ETT biofilms, on the basis of variations in the domains of the 16S ribosomal RNA (rRNA) gene16,17,18. For example, recent studies among adults undergoing mechanical ventilation demonstrated a much higher diversity of microflora in ETT biofilms via the culture-independent method compared with the culture-dependent method16,17,18. However, similar studies among intubated neonates are scarce.
To fill in these knowledge gaps described above, we conducted the present study using ETT samples collected from intubated neonates, and the patient data available at Children’s Hospital of Chongqing Medical University (CHCMU), China. The NICU in CHCMU is a 50-bed, level 3 NICU and serves as the referral base for the emergency medical treatment of critically ill and premature newborns from Chongqing. Using the culture-independent method based on Illumina Miseq platforms, we aimed to present a global view of the microbial communities in ETT biofilms from ventilated neonates with and without VAP. To further characterize the pathogenesis of the ETT biofilm flora in VAP, representative bacterial species were isolated from ETT biofilms of VAP patients, and their biofilm formation capacities were assessed in vitro. With a better understanding of the ETT biofilm flora among intubated neonates, corresponding prevention and treatment strategies can be developed to decrease the incidence of VAP among neonates.
Results
Study patients
Neonates with sustained mechanical ventilation for more than 48 hours in CHCMU during the study period from January 31, 2014, to July 31, 2014, were considered in the study. Considering that the underlying disease prior to intubation may influence the microbiota in ETT biofilms, patients diagnosed with neonatal respiratory distress syndrome (NRDS) and pneumonia during the study period were consecutively involved and classified into NRDS and pneumonia groups. During the study period, we collected the data of 62 intubated neonates. However, the study included all of the available 49 (79.0%) patients, while the other 13 (21.0%) were excluded from the study due to sample contamination (n = 5) and PCR failure (n = 8). The five contaminated samples were polluted in the process of sample procession by non-sterile solutions due to improper operations. Considering the potential selection bias due to the 13 excluded cases, the distribution of the basic demographic (sex, gestational age and birth weight) and clinical characteristics (intubation duration, VAP or no VAP) between the 49 study patients and the 13 missing cases were compared (Table 1). No significant difference was identified in the distribution of the included demographic and clinical characteristics (P > 0.05, Table 1). Of the 49 study patients, 23 were diagnosed with NRDS, including 17 VAP cases; the remaining 26 patients were pneumonia cases, including 13 VAP cases. The baseline characteristics between VAP and non-VAP patients were compared among the NRDS and pneumonia groups (Tables 2 and 3). The included baseline characteristics were similarly distributed between patients with and without VAP, except for the intubation duration. The intubation duration of VAP cases was significantly longer than those without VAP among NRDS patients (P < 0.05, Table 2).
Miseq sequencing and initial data processing
We obtained all 49 ETT samples collected from the 49 study patients at extubation. All of the study samples were successfully processed for sequencing. After denoising and chimera removal, a total of 787, 916 reads remained and were de-multiplexed. The mean number of reads was 14, 591 per sample. The high coverage coefficients of sequencing (over 99.9%) showed that the magnitude of the sequencing was sufficient to capture the operational taxonomy unit (OTU) diversity in all of the study samples.
Microbial diversity in ETT biofilms
The Shannon-Wiener index in the ETT biofilm flora among pneumonia patients was significant higher than in those among NRDS subjects (3.36 ± 1.51 vs. 0.84 ± 0.18, P < 0.05, Table 4). Similar results were exhibited in the comparisons of the Ace and Chao index between pneumonia and NRDS patients (P < 0.05, Table 4). The Simpson’s index in pneumonia patients was significantly lower than that in NRDS subjects (P < 0.05, Table 4). All four biodiversity indexes above indicated a lower richness and diversity of microflora in the ETT biofilms of NRDS patients than of pneumonia patients.
Principal coordinates analysis (PCoA) to assess β diversity was also conducted between the NRDS and pneumonia subjects. PCoA was used to visualize the effects of the two major factors on the community composition. The distribution of the pneumonia samples was much more dispersed compared with the NRDS samples (Fig. 1). Additionally, the pneumonia samples were clearly separated from the NRDS samples, indicating the distinct bacterial patterns between pneumonia and NRDS subjects (Fig. 1). Adonis, one of the non-parametric multivariate statistical tests, also revealed that the microbial community structure was significantly different between the pneumonia and NRDS samples (F = 27.962, P = 0.001).
Principal coordinate analysis (PCoA) of the study samples based on the Bray-Curtis distance of bacteria communities. Samples collected from NRDS patients are represented by circles (red), including 17 with (V1–V17) and 6 without (R1–R6) ventilator-associated pneumonia (VAP) cases. Samples collected from pneumonia patients are represented by squares (blue), including 13 with (1V–13V) and 13 without (1R–13R) ventilator-associated pneumonia (VAP) cases.
Bacterial communities in ETT biofilms
Regarding the distinct bacterial signatures in ETT flora between neonates with NRDS and those with pneumonia, the bacterial communities of the two groups were analysed separately. For NRDS patients, a total of 56 OTUs were identified in ETT biofilms, with an average of 17.7 OTUs per sample. Proteobacteria (87.4%, 306, 140/350, 330 reads) was dominant, followed by Firmicutes (8.7%, 30,324/350,330 reads) (Fig. 2A). At the genus level, Pseudomonas (44.7%, 156,626/350,330) and Enterobacter (42.3%, 148,357/350,330) were overrepresented, followed by Streptococcus (5.1%, 17,775/350,330). The detection rates of the three dominant genera in ETT biofilms among NRDS patients are shown in Fig. 2B. From the PCoA above (Fig. 1), no difference was identified between the global structures of microflora in the ETT biofilms of subjects with and without VAP. At the species level, over 99% (212,490/212,657) of reads identified as Pseudomonas sp. belonged to Pseudomonas aeruginosa PAO1. All of the reads identified as Enterobacter sp. and Streptococcus sp. failed to be further assigned at the species level.
Bacterial communities in endotracheal tube biofilm. (A) Relative abundance of sequences belonging to different bacterial phyla in endotracheal tube biofilms of the 23 neonatal respiratory distress syndrome (NRDS) patients in the study. The y-axis indicates the relative abundance of phyla, and the x-axis indicates different samples from patients with concurrent ventilator-associated pneumonia (VAP) (V1–V17) and without VAP (R1–R6). (B) The detection rates of the three dominant genera in endotracheal tube biofilms of the 23 NRDS patients. (C) Relative abundance of sequences belonging to different bacterial phyla in endotracheal tube biofilms of the 26 patients with pneumonia in the study. The y-axis indicates the relative abundance of phyla, and the x-axis indicates different samples from patients with concurrent VAP (1V–13V) and without VAP (1R–13R). (D) The detection rates of the three dominant genera in endotracheal tube biofilms of the 26 pneumonia patients.
Among pneumonia patients, 1,557 OTUs were identified in all of the ETT biofilm samples, with an average of 257.6 OTUs per sample. Proteobacteria (48.0%, 182,922/381,068) and Firmicutes (32.1%, 122,336/381,068) were the most widely distributed phyla among pneumonia patients (Fig. 2C). The PCoA already showed the different microbial patterns between NRDS and pneumonia patients, and to further confirm if the compositions of the ETT biofilm flora were different, we compared the abundance of Proteobacteria and Firmicutes (the two most overrepresented phyla in both groups) in the above two groups. We found significant differences in the ratio of the two phyla (P < 0.05). At the genus level, Streptococcus (19.4%, 73,772/381,068), Pseudomonas (12.0%, 45,798/381,068) and Enterobacter (6.4%, 24,433/381,068) were overrepresented. The detection rates of the three dominant genera in the ETT biofilms among pneumonia patients are shown in Fig. 2D. The PCoA showed no difference between the global microbial communities in the ETT biofilms of subjects with and without VAP (Fig. 1). Among the three dominant genera, 99.9% of the reads (73,691/73,772) assigned to Streptococcus sp. and all of the reads assigned to Enterobacter sp. failed to be further identified at the species level. Sixty-six percent of the reads identified as Pseudomonas sp. were assigned as Pseudomonas aeruginosa PAO1.
Association between Streptococcus sp. in ETT biofilm and VAP
Considering the significantly distinct microbial signatures in ETT biofilms between NRDS and pneumonia subjects and their different proportions of VAP cases (17/23 vs. 13/26) in the study, the association between bacterial factors and VAP was analysed in NRDS and pneumonia patients. In NRDS patients, Streptococcus sp. OTUs were present (as at least 1% of sequencing reads) in ETT biofilms among eight patients, which included seven VAP patients and the remaining one non-VAP patient (Fig. 3A). However, the only non-VAP patient was diagnosed with culture-confirmed sepsis one week after extubation, which raised the concern that nosocomial infection may occur via mechanical ventilation. We further compared the demographic and clinical characteristics between VAP patients with (n = 6) and without Streptococcus sp. present (n = 11) in ETT biofilms. Although not statistically significant, the intubation duration was longer (13.0 [5.2–15.7] vs. 4.8 [4.2–10.4] days) and the white blood cell (WBC) counts were lower ([14.4 ± 5.1] × 109/L vs. [22.8 ± 10.5] × 109/L) in Streptococcus sp. present cases than the remaining Streptococcus sp. absent cases (P = 0.07 and 0.09, respectively, Table 5).
Ratio of Streptococcus sp. in endotracheal tube biofilm. (A) Relative abundance of sequences belonging to Streptococcus sp. in endotracheal tube biofilms of the 23 neonatal respiratory distress syndrome (NRDS) patients in the study. The y-axis indicates the relative abundance of Streptococcus sp. and the x-axis indicates different samples from patients with concurrent ventilator-associated pneumonia (VAP) (V1–V17) and without VAP (R1–R6) among the 23 NRDS patients in the study. (B) Relative abundance of sequences belonging to Streptococcus sp. in endotracheal tube biofilms of the 26 patients with pneumonia in the study. The y-axis indicates the relative abundance of Streptococcus sp., and the x-axis indicates different samples from patients with concurrent VAP (1V–13V) and without VAP (1R–13R) among the 26 patients with pneumonia in the study.
In pneumonia patients, Streptococcus sp. were present in nine VAP patients and seven non-VAP patients (Fig. 3B). The Spearman test showed a significant correlation between the abundance of Streptococcus sp. in ETT biofilms and the onset of VAP (R = 0.49, P = 0.011 < 0.05). Although not statistically significant, we found that the WBC counts of VAP patients with Streptococcus sp. (n = 9) were lower than the remaining (n = 4) VAP cases ([12.1 ± 1.9] × 109/L vs. [24.2 ± 8.8] × 109/L) (P = 0.07, Table 6).
Identification of Streptococcus sp. in ETT biofilms
In all of the 15 ETT biofilm samples in which Streptococcus was present among VAP patients, Streptococcus pneumonia (S. pneumonia) was not detected according to S. pneumonia specific PCR. Based on biofilm cultivation, only one Streptococcus sp. strain has been isolated. After performing multilocus sequence analysis (MLSA) of the seven housekeeping genes, the strain can be assigned to Streptococcus sanguinis (S. sanguinis) according to its phylogenetic tree with those established type stains (Fig. 4).
Biofilm formation of the clinical S. sanguinis isolate
To gain insight into the role of the clinical S. sanguinis strain in the development of VAP, we investigated its capacity for biofilm formation. Crystal violet assays (Fig. 5A), plate counts (Fig. 5B), and confocal laser scanning micrographs (Fig. 5C) all showed that the clinical S. sanguinis strain did not form a significant biofilm. Pseudomonas aeruginosa PAO1, which was widely distributed in our study samples, was able to form biofilm. We further assessed whether the clinical S. sanguinis isolate could affect the biofilm formation of PAO1 in the in vitro co-culture model of PAO1 and S. sanguinis isolate. When co-cultured with the S. sanguinis isolate, the PAO1 biofilm was significantly thicker (33.16 ± 1.12 μm vs. 21.50 ± 1.33 μm) (Fig. 5C), cell viability of the biofilm was higher (Fig. 5B) and biofilm biomass (Fig. 5A) was larger than that of mono-PAO1 biofilms (P < 0.05). Quorum sensing (QS) of Pseudomonas aeruginosa is associated with biofilm formation19. To further explore the possible mechanisms for the enhanced biofilm mass with the concurrent clinical S. sanguinis isolate, we investigated gene expression of the four critical QS genes, lasI, rhlI, lasR, and rhlR. We found that all four investigated genes were significantly upregulated in the S. sanguinis and PAO1 co-cultured biofilms compared to the mono-PAO1 biofilm (P < 0.05, Fig. 6A,B,C and D).
Biofilm formation assessed by crystal violet assay (A), plate count (B) and confocal laser scanning microscopy (CLSM) (×400) (C). PAO1 represents the 3-day Pseudomonas aeruginosa PAO1 biofilm. PAO1 + ETB-1 represents the 3-day Pseudomonas aeruginosa PAO1 and the clinical S. sanguinis strain co-cultured biofilm. ETB-1 represents the 3-day clinical S. sanguinis strain biofilm. *Indicates a significant difference between the PAO1 and the PAO1 + ETB-1 group (P < 0.05). Control represents the brain heart infusion medium applied in the study for biofilm culture. (C) Representative confocal laser scanning micrographs of biofilm in PAO1 (a,d), PAO1 + ETB-1 (b,e) and ETB-1 (c,f); the scale bars = 50 μm.
(A,B,C,D) Expressions of Pseudomonas aeruginosa Quorum sensing genes lasI, lasR, rhlI and rhlR. PAO1 represents the 3-day Pseudomonas aeruginosa PAO1 biofilm. PAO1 + ETB-1 represents the 3-day Pseudomonas aeruginosa PAO1 and clinical S. sanguinis strain co-cultured biofilm. *Indicates a significant difference in the expression of lasI, lasR, rhlI and rhlR between the PAO1 and the PAO1 + ETB-1 group (P < 0.05). (E) IL-8 expression in different groups after stimulation with BCM. The x-axis indicates the 1 hour, 3 hour, 6 hour, 9 hour, 12 hour and 24 hour incubation of BEAS-2B cells with biofilm conditioned medium (BCM) of different groups. PAO1 represents BCM of 3-day Pseudomonas aeruginosa PAO1 biofilm. PAO1 + ETB-1 represents BCM of 3-day Pseudomonas aeruginosa PAO1 and the clinical S. sanguinis strain co-cultured biofilm. ETB-1 represents BCM of the 3-day clinical S. sanguinis strain biofilm. Control represents the brain heart infusion medium applied in the study of biofilm culture. *Indicates a significant difference in IL-8 expression between the PAO1 group and the PAO1 + ETB-1 group (P < 0.05).
IL-8 secretion in human lung/bronchus epithelia induced by BCM
After exposure to potential pathogens, airway epithelia cells produce IL-8 to initiate migration and activation of neutrophils and leukocytes against infection. Because IL-8 is critical in innate, and the thereafter, adaptive immune responses, its release by airway epithelia induced by the biofilm conditioned medium (BCM) was also investigated. The co-cultured S. sanguinis and PAO1 BCM induced a lower level of IL-8 compared with PAO1 BCM after intubation with human lung/bronchus epithelia BESA-2B cells for three hours (P < 0.05, Fig. 6E).
Discussion
Biofilm formation on ETTs is a universal phenomenon and provides a bacterial reservoir for VAP among mechanically ventilated patients. To our knowledge, this is the first study to characterize complex microbial communities in ETT biofilms among intubated neonates based on next generation sequencing. Microbial investigations based on next generation sequencing have broadened our ability to identify the complex microbial world. Our study showed that this non-cultural approach can be used to characterize microflora in ETT biofilms. The richness and diversity of ETT biofilm flora have been underestimated by the conventional culture-based approach. Our results revealed a far more complex microflora in ETT biofilms from intubated neonates compared to the previous culture-based study. Additionally, we found for the first time that patients with different underlying diseases before intubation had distinct microbial signatures in their ETT biofilms. This finding suggests the importance of well-categorized cases based on underlying diseases to characterize the ETT biofilm flora, particularly to investigate its association with outcome. Therefore, the diversity of pathogens involved in VAP is far more complex than the current literature suggests.
Previous studies have reported that Pseudomonas sp. and Enterobacter sp. were common pathogens that caused VAP20, 21, and we found that the two genera were overabundant in ETT biofilms in our study samples. This finding validates the fact that biofilms serve as the source of pathogens for VAP. However, the two genera were also identified in the ETT biofilms of patients without VAP, suggesting that the colonization of Pseudomonas sp. and Enterobacter sp. in ETT biofilms is necessary but not enough to cause VAP. The prevalence of Pseudomonas sp. and Enterobacter sp. in ETT biofilms in our study samples is alarming, and strategies are urgently needed to minimize or eliminate their colonization among intubated neonates to reduce nosocomial infections in NICUs.
Oral bacterial species were considered to be the potential reservoir of pathogens involved in VAP22. Recent clinical trials have also shown that VAP incidence could be reduced by improving oral hygiene23, 24. In the current study, the abundance of Streptococci in ETT biofilms was significantly related to the onset of VAP, and Streptococci were reported to be the dominant members of normal flora in the oral cavity among infants25. This suggests an association between the oral flora in ETT biofilms and VAP. Previous non-cultural based studies among intubated adults also demonstrated the existence of oral flora in ETT biofilms, yet their role in VAP has not been addressed16,17,18. Our finding provides the first epidemiologic evidence of the role of Streptococci in ETT biofilms in VAP and therefore highlights the usefulness of addressing these commensal oral flora to prevent VAP among neonates.
The present study showed that the clinical S. sanguinis strain isolated from ETT biofilms could enhance the biofilm formation and viability of the common VAP pathogen, PAO1, in vitro. As one of the commensal oral species, S. sanguinis is pathogenic when entering the bloodstream to cause bacterial endocarditis or in some immune-comprised patients26. Multiple bacterial species in the microbiota are responsible for diseases rather than a single organism27. Bacterial members of the microbiota may interact with each other by competition and coordination28. A well-documented mechanism for the interactions among bacteria is cell-cell communication via QS cross talk. The QS system has been identified to regulate virulence factor expression, biofilm formation, and antibiotic production, all of which are associated with the pathogenesis of bacteria19. The communication is conducted via chemical signal molecules termed autoinducers (AI), which are produced and released by QS bacteria29. Genes involved in the QS system participate in the production of these AIs. For Pseudomonas aeruginosa, N-acyl homoserine lactone (AHL) is as an important AI as AI-1, which includes N-3-oxo-dodecanoyl-homoserine lactone (3O-C12-HSL) and N-butanoyl-homoserine lactone (C4-HSL). The QS Genes lasI and rhlI encode 3O-C12-HSL synthetase and C4-HSL synthetase, respectively. In addition, the las and rhl systems also consist of the transcriptional activator proteins lasR and rhlR. We found that the clinical S. sanguinis strain significantly upregulated expression of lasI, rhlI, lasR and rhlR of Pseudomonas aeruginosa PAO1 in the study. This finding might be a reason for the increased biofilm formation of PAO1 within the S. sanguinis isolate.
In addition, the co-culture S. sanguinis and PAO biofilm BCM induced a lower level of IL-8 release in airway epithelia cells than the mono-PAO biofilm in vitro. IL-8 can promote neutrophil recruitment and influence circulating leukocyte populations in vivo 30. Therefore, the IL-8 disparity in vitro in our study agrees with our finding of lower WBC counts among patients with Streptococcus sp. present in ETT biofilms. These findings imply that Streptococcus sp. in biofilms might alleviate the host immune response to putative pathogens, such as Pseudomonas aeruginosa. An impaired immune response results in the persistence of pathogens and infection31. It has been recently proposed that 3O-C12-HSL, mentioned earlier, could impair the function of nuclear factor-κB (NF-κB) and lead to the repression of NF-κB-responsive genes encoding inflammation cytokines, including IL-831. Thus, the alleviated immune response with S. sanguinis might partly be a result of the increased gene expression of lasI, which encodes 3O-C12-HSL synthetase.
This study has several limitations. First, the sample size in the present study is relatively small, which may result in inadequate statistical power. However, we provide a preliminary insight into the microflora in ETT biofilms among intubated neonates, especially in view of the total lack of this type of data among intubated neonates in NICUs in China and other parts of the world. Antimicrobial therapy might be one of the confounding factors influencing microbial communities. It has been confirmed that biofilm formation was not associated with systemic antibiotics among patients with mechanical ventilation11. However, the fact that various types of antibiotics were administered according to the patient condition in the present study made it impossible to assess the potential role of antibiotics in profiling the ET biofilm microflora. The reads identified as members of Streptococci have not been assigned at the species level due to the limited resolution of the 16S rRNA gene. The other limitation is that we did not have sequential ETT samples during intubation for every intubated neonate, so the dynamics of the microbiome during intubation for an individual patient has not been revealed. However, sequential sampling for intubated neonates is not allowed due to the concern for patient safety, and re-intubation was also reported to be a risk for VAP32.
Conclusions
This study extends the knowledgebase of microbiota in ETT biofilms from intubated neonates and provides new insight into the pathogenesis of VAP by suggesting that Streptococcus sp. may evolve in the onset of VAP by interacting with other nosocomial pathogens and regulating host immune responses. More epidemiological studies with larger sample sizes from intubated neonates are warranted based on which strategies can be developed to address this important nosocomial disease in NICUs.
Methods
Ethics statement
All experiments were performed following the relevant guidelines and regulations of CHCMU. The study protocol was reviewed and approved by the Medical Ethics Committee of CHCMU. Informed consent was obtained from the parents or legal guardians of the study patients. De-identified data were used in the data analysis.
Patient involvement and classification
Neonates who suffered from mechanical ventilation for more than 48 hours in CHCMU during the study period from January 31, 2014, to July 31, 2014, were considered in the study. Patients diagnosed with NRDS and pneumonia before mechanical ventilation during the study period were consecutively enrolled and classified into either the NRDS or pneumonia group. To ensure NRDS patients would have normal oral flora and a sterile lower respiratory tract prior to intubation, NRDS patients with the following conditions were excluded, as previously described33: (1) intrauterine infection; (2) diagnosed with concurrent infectious diseases prior to intubation; and (3) mothers with a history of infections or who had used antibiotics during the last month of pregnancy. VAP was considered according to the definition by the CDC for infants younger than 1 year, including radiological signs, clinical signs & symptoms and microbiological findings34, based on which all of the study patients in NRDS and pneumonia groups were further identified as VAP and non-VAP cases. Empirical antibiotics were applied in all the study patients according to patient condition as determined by clinicians (Tables 2 and 3). NRDS, pneumonia and VAP were diagnosed by at least three experienced clinicians according to patients’ symptoms and X-ray findings.
Sample collection and procession
ETT samples were collected immediately after extubation. The distal 2 cm of the tubes were cut off, transferred to 5 ml of phosphate buffered saline (PBS), and processed using a Vortex and sonication to release biofilms as previously described17.
PCR amplification and Illumina Miseq sequencing
Microbial DNA was extracted using the TIANamp Bacteria DNA Kit (Tiangen, China) from the PBS solution containing recovered biofilm following the manufactory’s instructions and stored at −20 °C for further use. The V3–V4 region of the bacteria 16S rRNA gene was amplified using the primers 338F (5′ACTCCTACGGGAGGCAGCA-3′) and 806R (5′GGACTACHVGGGTWTCTAAT-3′). A unique barcode was added to the primer for the identification of each sample. Amplification was initiated by initial denaturation at 95 °C for 2 min, followed by 25 cycles at 95 °C for 30 s, 55 °C for 30 s, 72 °C for 30 s, and a final extension at 72 °C for 5 min. The PCR products were confirmed by 2% agarose gel electrophoresis. The DNA in the target band was recovered using an AxyPrep DNA Gel Extraction Kit (Axygen Biosciences, Union City, CA, U.S.), and amplicon sequencing was performed on the Illumina MiSeq platforms at Majorbio Bio-Pharm Technology Co., Ltd (Shanghai, China). Complete data sets are submitted to the NCBI Short Read Archive under accession no. SRP057949.
Processing of sequencing data
Pairs of reads of the original DNA fragments were quality-filtered using QIIME (version 1.17) and merged using FLASH35. Sequence reads were assigned to each sample according to the unique barcode of each sample. QIIME and the UPARSE pipeline were used to analyse the sequences, and sequences were assigned to the OTU with 97% similarity as previously described36, 37. One representative sequence per OTU was selected and assigned to certain taxonomic data using RDP classifier (version 2.2) and was identified in the RDP database (Release 11.1 http://rdp.cme.msu.edu/)38. To evaluate the coverage of sequencing, coverage coefficients for each sample were calculated using Mothur software (version v.1.30.1)39. The Ace and Chao indices were calculated by Mothur to assess the richness of the microbial communities, and the Shannon Wiener index and the Simpson’s index were to evaluate the community diversity. β diversity of microbial environments visualized by PCoA and Adonis based on Bray-Curtis distances were performed in R (http://www.r-project.org/). The histogram of microbial communities for each sample was generated based on the relative abundance of each OTU. Bacterial species with a relative ratio of more than 1% were considered to be present in the sample.
Isolation and identification of Streptococcus sp. strains
According to our preliminary data, Streptococcus sp. OTU was overabundant in ETT biofilms of VAP patients. To investigate the existence of S. pneumonia, the common causative agent of pneumonia, the S. pneumonia specific PCR was performed as previously described40. The released biofilm cell suspensions were inoculated onto Columbia blood agar plates and cultured for 48 h at 37 °C. Bacterial isolates were identified as Streptococcus sp. according to haemolysis, gram positive reaction, coccus morphology arranged in chains, and negative catalase test results41. Bacterial DNA was extracted as mentioned earlier. The concatenated sequences of the seven housekeeping genes were amplified and sequenced as previously described42. The phylogenetic tree was constructed via MEGA5 (http://www.megasoftware.net/). Strains were assigned to species on the basis of their distance from species type strains41, 42.
Biofilm culture
Bacterial biofilms were cultured in brain heart infusion (BHI) (Nissui, China) medium at 37 °C 5% CO2 for three days43. BCM was collected as previously described44. Plate count was conducted to evaluate the viability of bacteria in biofilms. Crystal violet assay was performed to compare the biofilm biomass as described43. Biofilms were stained with a LIVE/DEAD BacLight kit (Invitrogen, U.S.) and observed using confocal laser scanning microscopy (CLSM). The CLSM image was analysed using ImageJ (http://imagej.nih.gov/ij/)43. Gene expressions of the critical QS genes including lasI, rhlI, lasR, and rhlR were investigated via Real-time PCR as previously described45.
IL-8 detection
Human lung/bronchus epithelia (BEAS-2B, ATCC-9609) were cultured in DMEM: F12 medium (Invitrogen, U.S.) with 10% fetal bovine serum (Hyclone, U.S.), 100 U/ml penicillin, and 100 mg/ml streptomycin at 37 °C in 5% CO2. The supernatants were collected after stimulation with BCM for 1 h, 3 h, 6 h, 9 h, 12 h and 24 h. The secretion of IL-8 was quantified using enzyme-linked immunosorbent assay (ELISA) following the manufacturer’s instructions (4A Biotech, China).
Statistical analysis
Continuous variables were expressed as the mean ± SEM or median (P25–P75) according to whether they followed a normal distribution. Dichotomous variables were presented as number (percentage). The comparisons among continuous data were analysed by Student’s t test, adjusted t-test, ANOVA, and Mann-Whitney U test as appropriate. The Chi-square test or Fisher’s exact test was used for comparisons among dichotomous variables. The association between OTU count and VAP was investigated using the Spearman test. All the statistical analyses were performed using SPSS (version 17.0). P value less than 0.05 was considered to be statistically significant.
References
Cernada, M., Brugada, M., Golombek, S. & Vento, M. Ventilator-associated pneumonia in neonatal patients: an update. Neonatology 105, 98–107 (2014).
System, N. N. I. S. National Nosocomial Infections Surveillance (NNIS) System Report, data summary from January 1992 through June 2004, issued October 2004. Am J Infect Control 32, 470 (2004).
Aly, H., Badawy, M., El-Kholy, A., Nabil, R. & Mohamed, A. Randomized, controlled trial on tracheal colonization of ventilated infants: can gravity prevent ventilator-associated pneumonia? Pediatrics 122, 770–774 (2008).
Polin, R. A. et al. Strategies for prevention of health care–associated infections in the NICU. Pediatrics 129, e1085–e1093 (2012).
Tan, B. et al. Epidemiology of pathogens and drug resistance of ventilator-associated pneumonia in Chinese neonatal intensive care units: A meta-analysis. Am J Infect Control 42, 902–910 (2014).
Pneumatikos, I. A., Dragoumanis, C. K. & Bouros, D. E. Ventilator-associated pneumonia or endotracheal tube-associated pneumonia? an approach to the pathogenesis and preventive strategies emphasizing the importance of endotracheal tube. Anesthesiology 110, 673–680 (2009).
Adair, C. et al. Implications of endotracheal tube biofilm for ventilator-associated pneumonia. Intens Care Med 25, 1072–1076 (1999).
Inglis, T., Millar, M., Jones, J. & Robinson, D. Tracheal tube biofilm as a source of bacterial colonization of the lung. J Clin Microbiol 27, 2014–2018 (1989).
Flemming, H.-C. & Wingender, J. The biofilm matrix. Nat Rev Microbiol 8, 623–633 (2010).
Feldman, C. et al. The presence and sequence of endotracheal tube colonization in patients undergoing mechanical ventilation. Eur Respir J 13, 546–551 (1999).
Gil-Perotin, S. et al. Implications of endotracheal tube biofilm in ventilator-associated pneumonia response: a state of concept. Crit Care 16, R93 (2012).
Garland, J. S. Strategies to prevent ventilator-associated pneumonia in neonates. Clin Perinatol 37, 629–643 (2010).
Tablan, O., Anderson, L., Besser, R., Bridges, C. & Hajjeh, R. CDC; Healthcare Infection Control Practices Advisory Committee. Guidelines for preventing health-care-associated pneumonia, 2003: recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee. MMWR Recomm Rep 53, 1–36 (2004).
Burmølle, M. et al. Enhanced biofilm formation and increased resistance to antimicrobial agents and bacterial invasion are caused by synergistic interactions in multispecies biofilms. Appl Environ Microbiol 72, 3916–3923 (2006).
Friedland, D. R., Rothschild, M. A., Delgado, M., Isenberg, H. & Holzman, I. Bacterial colonization of endotracheal tubes in intubated neonates. Arch Otolaryngol Head Neck Surg 127, 525–528 (2001).
Vandecandelaere, I. et al. Assessment of microbial diversity in biofilms recovered from endotracheal tubes using culture dependent and independent approaches. PloS one 7, e38401 (2012).
Cairns, S. et al. Molecular analysis of microbial communities in endotracheal tube biofilms. PLoS One 6, e14759 (2011).
Perkins, S. D., Woeltje, K. F. & Angenent, L. T. Endotracheal tube biofilm inoculation of oral flora and subsequent colonization of opportunistic pathogens. Int J Med Microbiol 300, 503–511 (2010).
Geske, G. D., O’Neill, J. C. & Blackwell, H. E. Expanding dialogues: from natural autoinducers to non-natural analogues that modulate quorum sensing in Gram-negative bacteria. Chem Soc Rev 37, 1432–1447 (2008).
Klompas, M., Kleinman, K. & Murphy, M. V. Descriptive epidemiology and attributable morbidity of ventilator-associated events. Infect Cont Hosp Ep 35, 502–510 (2014).
Jones, R. N. Microbial etiologies of hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia. Clin Infect Dis 51, S81–S87 (2010).
Bahrani-Mougeot, F. K. et al. Molecular analysis of oral and respiratory bacterial species associated with ventilator-associated pneumonia. J Clin Microbiol 45, 1588–1593 (2007).
Mori, H. et al. Oral care reduces incidence of ventilator-associated pneumonia in ICU populations. Intens Care Med 32, 230–236 (2006).
Berry, A. M., Davidson, P. M., Masters, J. & Rolls, K. Systematic literature review of oral hygiene practices for intensive care patients receiving mechanical ventilation. Am J Crit Care 16, 552–562 (2007).
Pearce, C. et al. Identification of pioneer viridans streptococci in the oral cavity of human neonates. J Med Microbiol 42, 67–72 (1995).
Caufield, P. W. et al. Natural history of Streptococcus sanguinis in the oral cavity of infants: evidence for a discrete window of infectivity. Infect Immun 68, 4018–4023 (2000).
Zhao, L. The gut microbiota and obesity: from correlation to causality. Nat Rev Microbiol 11, 639–647 (2013).
Peters, B. M., Jabra-Rizk, M. A., Graeme, A., Costerton, J. W. & Shirtliff, M. E. Polymicrobial interactions: impact on pathogenesis and human disease. Clin Microbiol Rev 25, 193–213 (2012).
Waters, C. M. & Bassler, B. L. Quorum sensing: cell-to-cell communication in bacteria. Annu. Rev. Cell Dev. Biol. 21, 319–346 (2005).
Van Zee, K. J. et al. IL-8 in septic shock, endotoxemia, and after IL-1 administration. J Immunol 146, 3478–3482 (1991).
Kravchenko, V. V. et al. Modulation of gene expression via disruption of NF-κB signaling by a bacterial small molecule. Science 321, 259–263 (2008).
Yuan, T.-M., Chen, L.-H. & Yu, H.-M. Risk factors and outcomes for ventilator-associated pneumonia in neonatal intensive care unit patients. J Perinat Med 35, 334–338 (2007).
Lu, W. et al. Increased constituent ratios of Klebsiella sp., Acinetobacter sp., and Streptococcus sp. and a decrease in microflora diversity may be indicators of ventilator-associated pneumonia: a prospective study in the respiratory Tracts of neonates. PloS one 9, e87504 (2014).
Centers for Disease Control and Prevention. Criteria for defining nosocomial pneumonia. http://www.cdc.gov/ncidod/hip/NNIS/members/penumonia/Final/PneuCriteriaFinal.pdf (Accessed January 30, 2009).
Magoč, T. & Salzberg, S. L. FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27, 2957–2963 (2011).
Edgar, R. C. UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10, 996–998 (2013).
Wang, Q., Garrity, G. M., Tiedje, J. M. & Cole, J. R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73, 5261–5267 (2007).
Cole, J. R. et al. The Ribosomal Database Project: improved alignments and new tools for rRNA analysis. Nucleic Acids Res 37, D141–D145 (2009).
Schloss, P. D., Gevers, D. & Westcott, S. L. Reducing the effects of PCR amplification and sequencing artifacts on 16S rRNA-based studies. PloS one 6, e27310 (2011).
Kim, W., Park, H. K., Hwang, W.-J. & Shin, H.-S. Simultaneous detection of Streptococcus pneumoniae, S. mitis, and S. oralis by a novel multiplex PCR assay targeting the gyrB gene. J Clin Microbiol 51, 835–840.
Shelburne, S. A. et al. Streptococcus mitis strains causing severe clinical disease in cancer patients. Emerg Infect Dis 20, 762–771 (2014).
Bishop, C. J. et al. Assigning strains to bacterial species via the internet. BMC Biol 7, 1 (2009).
Song, S. et al. Does Streptococcus mitis, a neonatal oropharyngeal bacterium, influence the pathogenicity of Pseudomonas aeruginosa? Microb Infect 17, 710–716 (2015).
Kirker, K. R., James, G. A., Fleckman, P., Olerud, J. E. & Stewart, P. S. Differential effects of planktonic and biofilm MRSA on human fibroblasts. Wound Repair and Regen 20, 253–261 (2012).
Li, H. et al. Autoinducer-2 regulates Pseudomonas aeruginosa PAO1 biofilm formation and virulence production in a dose-dependent manner. BMC microbiol 15, 1 (2015).
Acknowledgements
We thank Xianhong Zhang in the Department of Neonatology, Children’s Hospital of Chongqing Medical University, for her excellent help in sample collection. This work was supported by National Natural Science Foundation of China (No. 81370744), Doctoral Degree Funding from Chinese Ministry of Education (No. 20135503110009), State key clinic discipline project (No. 2011-873), the subproject of National Science & Technology Pillar Program during the 12th Five-year Plan Period in China (No. 2012BAI04B05), and the Clinical Research Program of Children’s Hospital of Chongqing Medical University (lcyj2014-11). The funding sources had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Contributions
Y.J. conceived and supervised the study, P.Y., S.S., A.Q. and Z.D. conducted the experiments, P.Y., T.X. and L.Z. collected the study samples, L.Z. collected the clinical data, P.Y. and S.S. analyzed the results, P.Y. drafted the manuscript. All the above authors reviewed and approved the final version of the manuscript for submission.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pan, Y., Song, S., Tang, X. et al. Streptococcus sp. in neonatal endotracheal tube biofilms is associated with ventilator-associated pneumonia and enhanced biofilm formation of Pseudomonas aeruginosa PAO1. Sci Rep 7, 3423 (2017). https://doi.org/10.1038/s41598-017-03656-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-03656-2
- Springer Nature Limited
This article is cited by
-
The spectrum of pneumonia among intubated neonates in the neonatal intensive care unit
Journal of Perinatology (2024)
-
Effect of invasive mechanical ventilation on the diversity of the pulmonary microbiota
Critical Care (2022)
-
Real-time monitoring of Pseudomonas aeruginosa biofilm formation on endotracheal tubes in vitro
BMC Microbiology (2018)